Health Canada approves Neffy, a needle-free nasal spray for severe allergies. Experts weigh in on its impact for anaphylaxis patients. Andrew Johnson has more.
VANCOUVER – Canadians living with life-threatening allergies will soon have a needle-free option to treat anaphylaxis, which experts are calling a “game changer.”
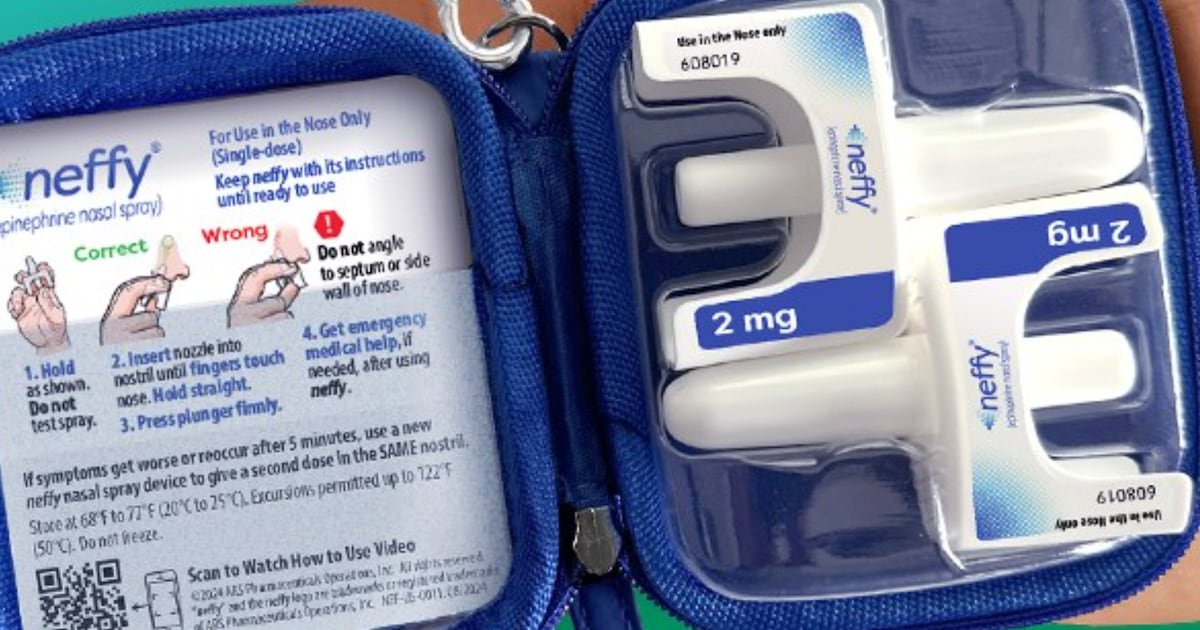
Health Canada has approved Neffy, a pocket-sized nasal spray that delivers epinephrine, the same life-saving medication found in auto-injectors commonly known by the brand name EpiPen, but without the need for a needle.
One doctor says the new device is “huge” for patients and caregivers.
“This is the ability to have a life-saving device available on you that’s easy to administer, and almost challenging to get wrong,” Dr. Mariam Hanna, a pediatric allergist, told CTV News.
 Neffy Neffy A new option for a life-threatening condition
Neffy Neffy A new option for a life-threatening condition
Anaphylaxis is a severe and potentially deadly allergic reaction with millions of Canadians at risk. It can be triggered by foods like peanuts and shellfish, insect stings or other allergens. Symptoms can include difficulty breathing, swelling of the throat or tongue, and a dangerous drop in blood pressure. Immediate access to epinephrine is critical, and Hanna says the nasal spray is a step forward.
“You want to be able to easily help in the event of an emergency,” she said. “We’ve seen an example of this quite a number of years back now with naloxone. Now police departments even carry and administer that as an antidote for those who have overdosed.”
For decades, Canadians have relied on injectable devices, which are single-use and must be jabbed into the thigh.
“Carrying a needle-based epinephrine or having this as the only route of delivery causes barriers for people who one: don’t like needles; two: don’t want to use a needle; or three: don’t want to carry the size that is required,” Hanna said.
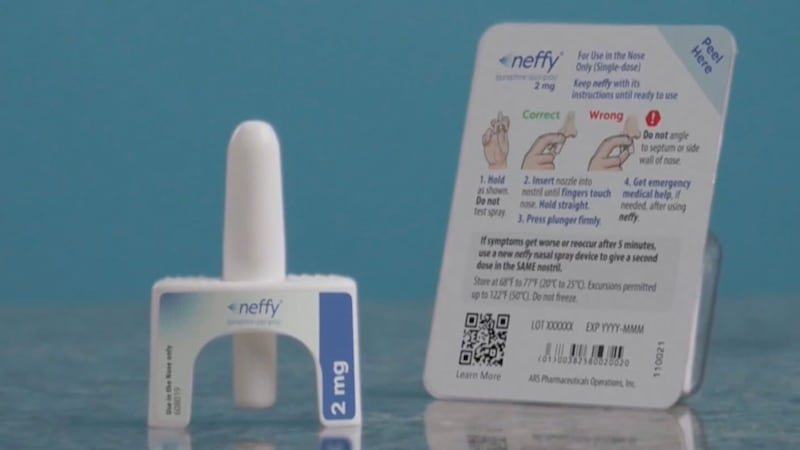 Needle-free allergy treatment Neffy approved in Canada Who can use it and when it’s coming
Needle-free allergy treatment Neffy approved in Canada Who can use it and when it’s coming
The newly approved two-milligram dose of Neffy is authorized for adults and children who weigh at least 30 kg (about 66 lbs).
According to ALK Canada, which holds the Canadian distribution rights, the spray could begin appearing in pharmacies as early as this summer.
A lower, one-milligram dose already approved in the United States for children between 15 and 30 kg is not yet authorized in Canada. The manufacturer says it plans to seek approval for that version later this year.
With files from CTV News’ Spencer Van Dyk