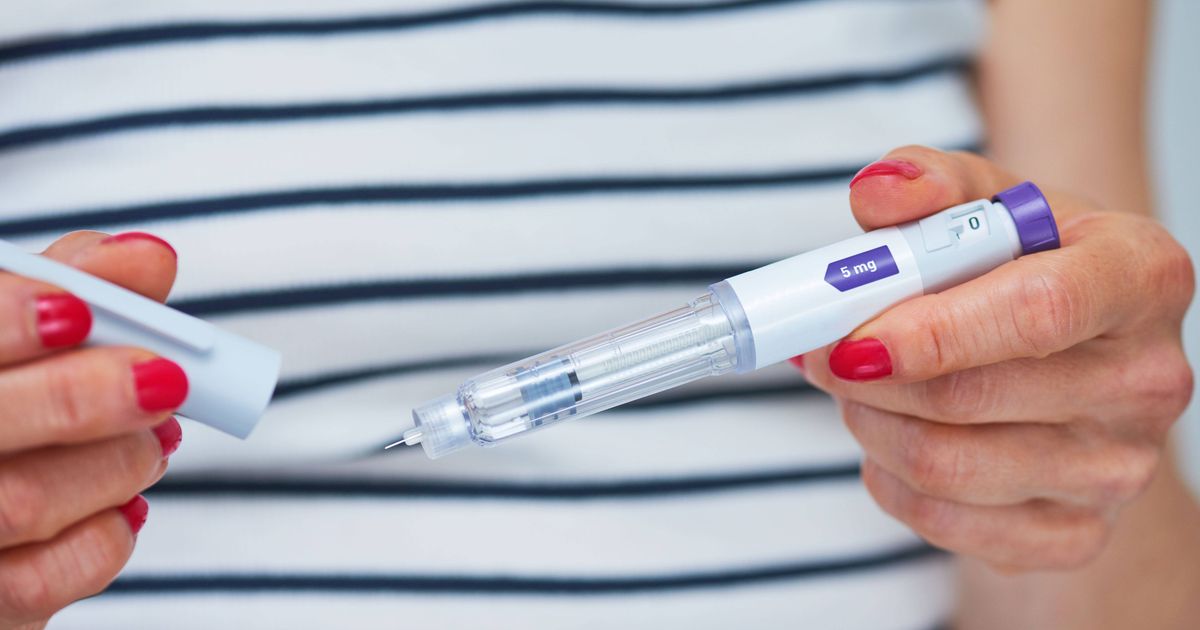
A safety alert has been issued after falsified Mounjaro pens were found.
A safety alert has been issued after falsified Mounjaro pens were supplied by an online pharmacy, with the MHRA warning users to check batch numbers immediately(Image: Peter Dazeley via Getty Images)
A safety warning has been issued for anyone taking Mounjaro after a counterfeit version of the medication was distributed to the public. Specific batches of the pen, which is an injectable prescription medicine approved for weight management, have been recalled, announced by the Medicines and Healthcare products Regulatory Agency (MHRA).
An online pharmacy was discovered to be supplying a version that didn’t meet safety standards to customers. The safety warning was released on Tuesday, February 24, and relates to the solution for injection in pre-filled pen only.
The Mounjaro KwikPen 7.5mg pens with this batch code are not affected. Five of the affected pens have been identified as being distributed through the Private Pharmacy Clinic in Birmingham.
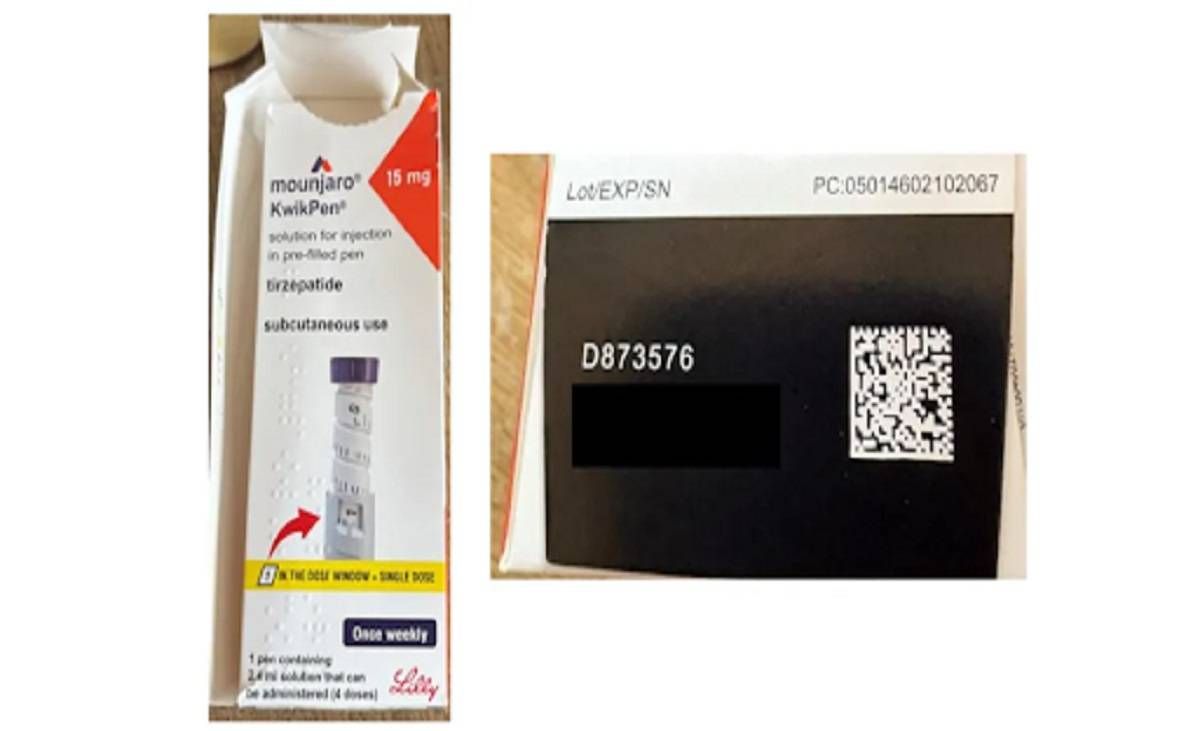
The affected Mounjaro (tirzepatide) KwikPen 15mg has the batch code D873576(Image: Which?)
The Mounjaro product affected by this alert include:
Mounjaro (tirzepatide) KwikPen 15mg – batch D873576
Testing of the medications confirmed that they do contain tirzepatide, however, their manufacturing process is unknown, therefore “sterility cannot be guaranteed”. As the product is injectable and its contents are not sterile, there is a potential risk of infection and allergic reactions, reports the Express.
Potential signs of infection can include redness, swelling or warmth around the site of the injection; pain or discomfort around the site of injection; fluid leakage from the site of injection; and mild fever, chills, fatigue, and sore throat. Signs of an allergic reaction might include a rash, itching or breathing difficulties.
Anyone experiencing symptoms of infection or an allergic reaction should seek urgent medical attention straight away.
The MHRA confirmed it hasn’t received any reports of patients needing treatment following use of these counterfeit medications. The problem came to light through defects with the pens, with the dose knob detaching during use in all instances bar one.
The MHRA was alerted to these fake products by the genuine manufacturer, Eli Lilly and Company Limited, which has logged five reports of faulty pens.
Dr Alison Cave, Chief Safety Officer at the MHRA said: “Check the batch number and strength of your KwikPen. If you have one of the affected pens, stop using it straight away. Please report it to the MHRA by emailing info@mhra.gov.uk. Please include “Mounjaro Pens” in the subject line. Keep the pen in a safe place.
“If you have administered injections using the pen already, please be reassured that, based on the cases reviewed to date, the risk to you is low. Testing has confirmed that the identified fake pens do contain tirzepatide, the active substance in Mounjaro, but because the manufacturing conditions are unknown, we cannot confirm that the contents are sterile.
“If you are unsure of how to identify the pen, or are unsure on whether you have administered an affected product and have any questions, please speak to a healthcare professional.”
Anyone who suspects they’ve been given a counterfeit medication or has spotted any defect with a weight-loss pen should flag it directly to the Yellow Card scheme via the website www.yellowcard.mhra.gov.uk. The scheme is also accessible through both Google Play and the Apple App Store.
Ensure our latest headlines always appear at the top of your Google Search by making us a Preferred Source. Click here to activate or add us as your Preferred Source in your Google search settings.