Wanwan Qiu, Qin and Müller’s former doctoral student, developed the connecting molecule specifically for this application. “It enables rapid structuring of hydrogels in the sub-micrometre range,” she says. The polymer chains are linked as soon as laser pulses of a certain wavelength hit the hydrogel. The irradiated areas immediately become solid, while the non-irradiated parts can be washed out later.
Jelly can be set at world-record speed
In this way, the researchers can use the laser beam to print any shapes and structures into the hydrogel with very fine resolution and extreme precision. The structures can be as small as 500 nanometres.
“Hydrogels resemble jelly, making them difficult to shape,” says ETH Professor Qin. “With our newly developed connecting molecule, we can now not only structure the hydrogel in a stable and extremely fine manner but also produce it at high writing speeds of up to 400 millimetres per second. That’s a new world record.”
Structures in the nanometre range
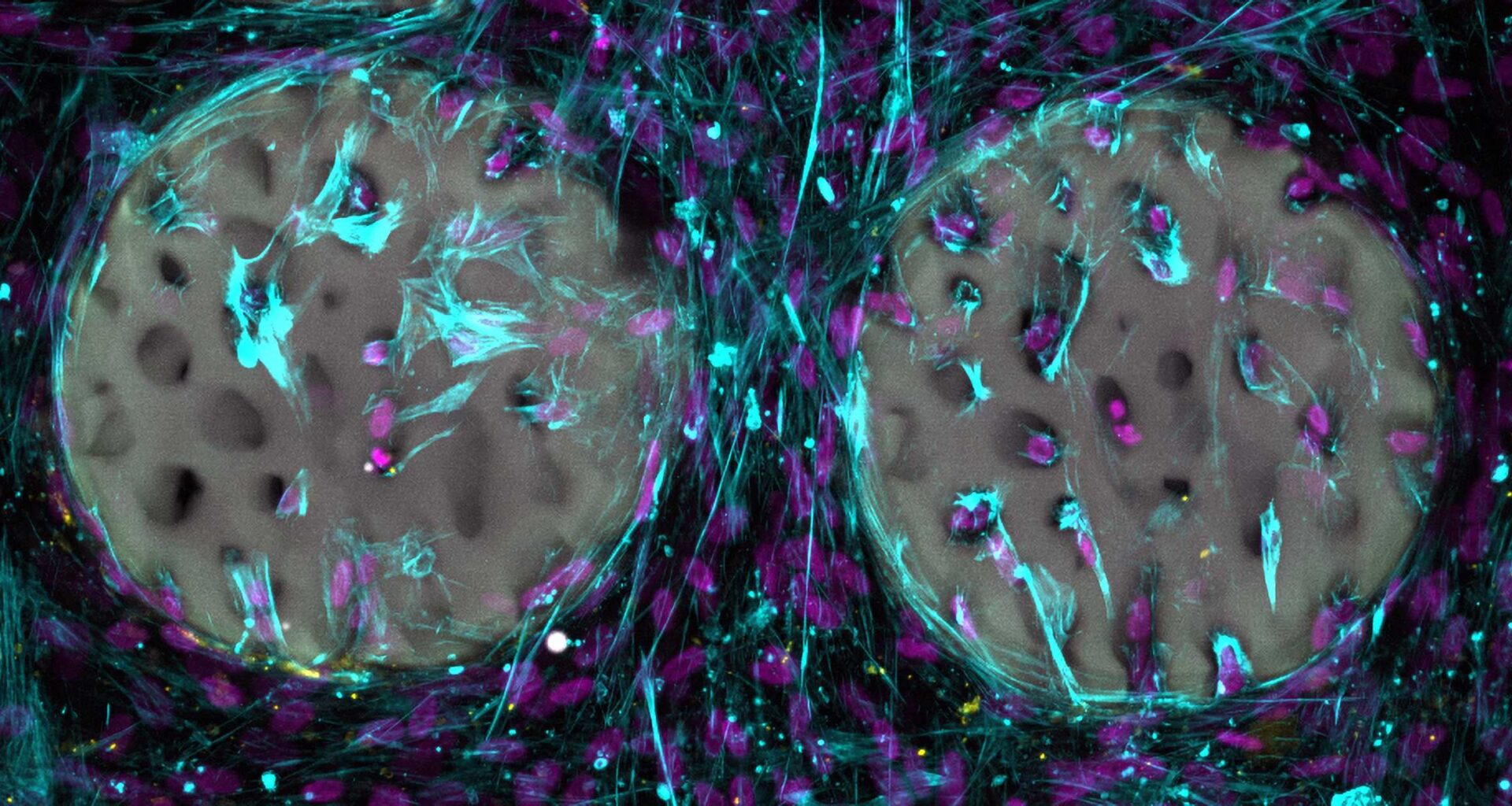
In their study, the researchers created complex, structured hydrogels that resemble real bone and feature a fine network of bone trabeculae. They used medical imaging as a template.
Even healthy natural bone is criss-crossed by a fine network of channels that are only nanometres thick and filled with fluid. “A piece of bone the size of a dice contains 74 kilometres of tunnels,” says Qin. By way of comparison, the longest railway tunnel in the world, the Gotthard Base Tunnel, measures 54 kilometres.
Material is biocompatible
So far, the researchers have tested the material only in a test tube. Results showed that bone-forming cells rapidly colonise the structured hydrogel and begin forming collagen, a vital component of bone. The tests also confirmed that the material is biocompatible and does not damage the bone-forming cells. The researchers have patented the base material and plan to make it available to the medical industry.
The researcher’s declared goal is for the hydrogel-based implant to one day be used in clinics to repair broken bones. However, more work is needed. Qin is preparing to conduct animal tests in collaboration with the AO Research Institute Davos. The team aims to determine whether their new bone repair material promotes the migration of bone-forming cells in living organisms and whether it restores bone strength over time.