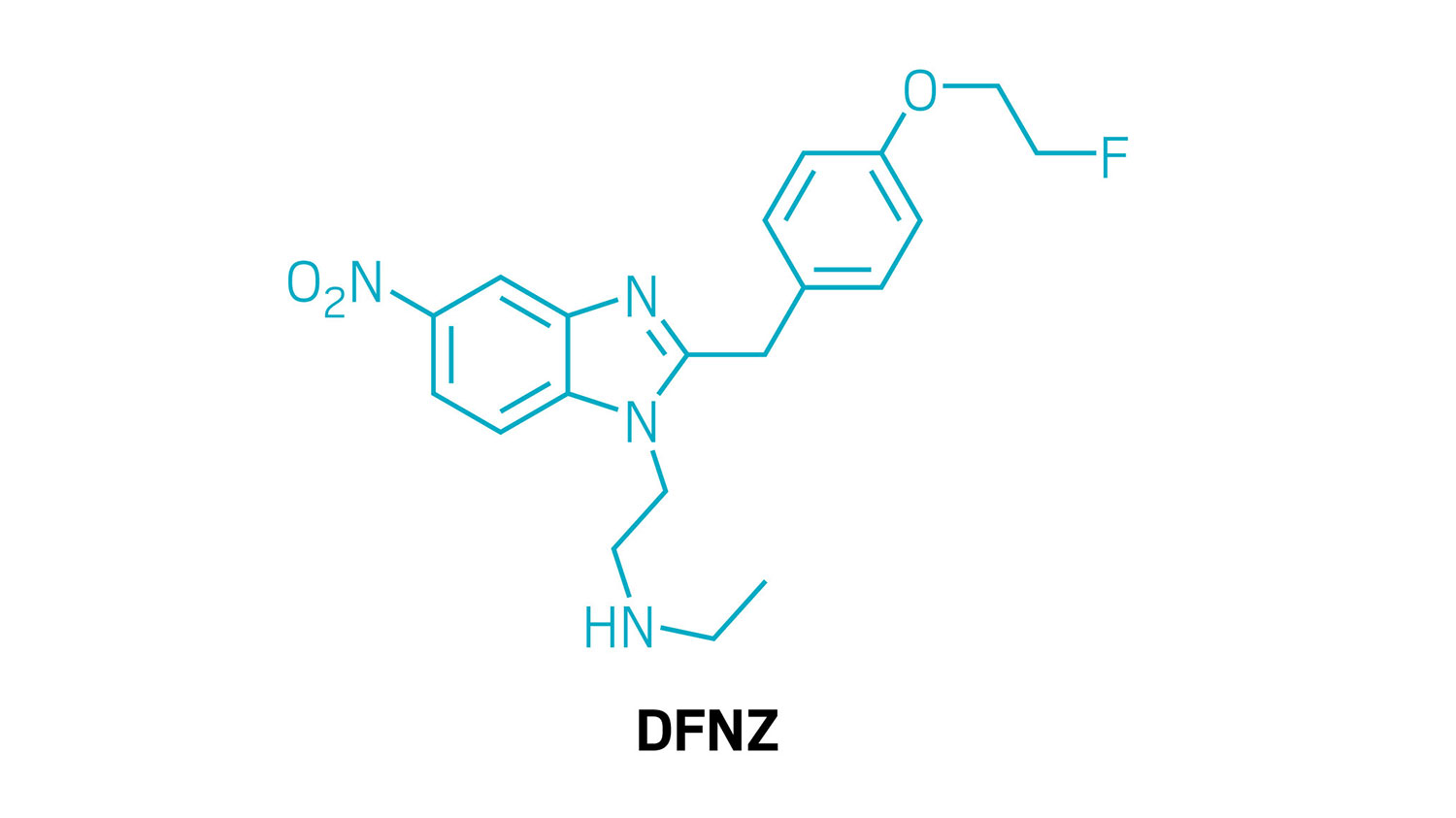
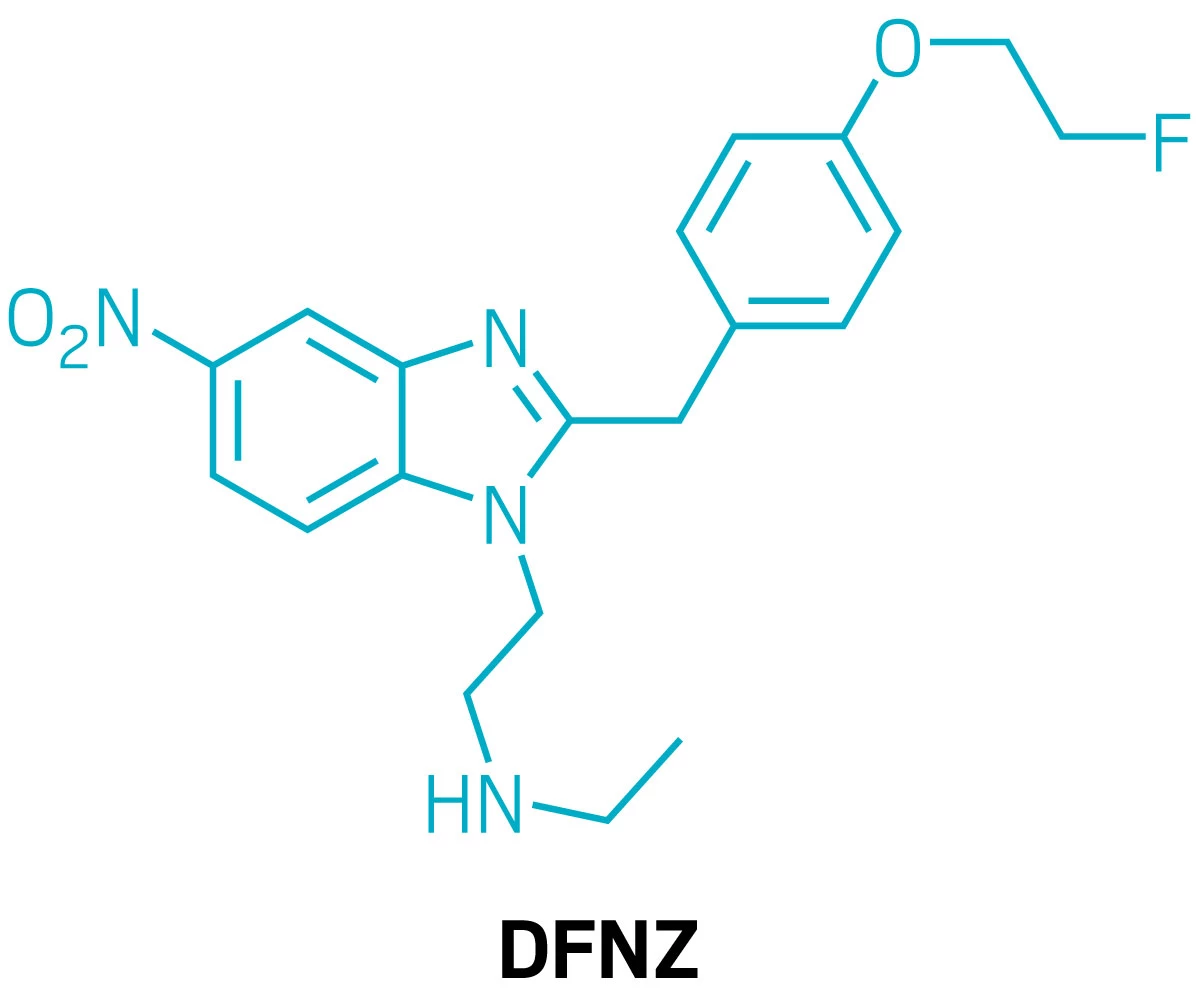
Structure of N-desethyl-fluornitrazene, or DFNZ.
Using a long-abandoned class of opioids as their starting point, scientists have discovered an opioid painkiller that doesn’t have the side effects typical of this class of drugs (Nature 2026, DOI: 10.1038/s41586-026-10299-9). In tests with rodents, the molecule—known as N-desethyl-fluornitrazene, or DFNZ—had a low potential for addiction and didn’t lead to respiratory depression, which is what causes most opioid overdose deaths. Although the compound has many hurdles to clear to become an approved drug, it could find use as an analgesic and as a replacement for the opioid-use disorder treatment methadone.
DFNZ is a member of the nitazene family of opioids. Because of their high potency and overdose risk, nitazenes were shelved not long after they were first reported in the 1950s. Etonitazene, the most potent member of the nitazene family, is about 1,000 times as powerful as morphine.
Researchers led by Michael Michaelides and Kenner C. Rice at the US National Institute on Drug Abuse and Georgios Skiniotis at St. Jude Children’s Research Hospital wanted to make a radiolabeled version of etonitazene so that they could use positron emission tomography (PET) to study its ability to get into the brain. They tacked a fluorine atom to the end of etonitazene’s ethoxy group, which also decreased the compound’s potency, and made a version with fluorine-18.
The researchers gave the radiolabeled compound to rats and used PET to observe its brain penetration, but they were in for a surprise. “We thought that it would stay in the brain longer and lead to more adverse effects,” Michaelides says. Instead, the team saw the radiolabeled compound going in and then coming out of the brain quickly.
That “led us to speculate that any metabolite would not be in the brain,” Michaelides says. The primary metabolite turned out to be DFNZ.
The researchers used medicinal chemistry, structural biology, pharmacology, and animal studies to study DFNZ. They describe the compound as a μ-opioid receptor superagonist—a molecule that elicits a greater effect than full agonists such as morphine.
DFNZ “has a very interesting profile overall that seems to be different than any other existing opioid,” Michaelides says. It can treat pain even though it doesn’t seem to get into the brain, and it appears to affect dopamine release in the brain in a way that’s distinct from other opioids—which means it’s less likely to be abused. It also doesn’t produce the tolerance or withdrawal symptoms associated with other opioids. Michaelides says the researchers are now pursuing an investigational new drug application for DFNZ.
Victor Mathis and Emmanuel Darcq, neuroscientists studying addictive disorders at the University of Strasbourg, write in a commentary accompanying the paper that the work is “a promising approach to opioid analgesics that dissociates pain relief from side effects such as tolerance, hyperalgesia, respiratory depression and misuse potential,” (Nature 2026, DOI: 10.1038/d41586-026-00806-3).
But Mathis and Darcq note that there are important limitations to address to avoid repeating mistakes that led to the opioid crisis. They write that prematurely and overoptimistically calling formulations of oxycodone safer analgesics “stands as a stark reminder that early claims of reduced misuse liability require rigorous, independent and long-term evaluation.”
Chemical & Engineering News
ISSN 0009-2347
Copyright ©
2026 American Chemical Society