LEARNING OBJECTIVES
At the completion of this article, the reader should… Identify clinical signs of the interventional window in glaucoma, including ocular surface issues, IOP fluctuations, and steroid responses, to guide timely referral.
Understand the effects of chronic topical therapy on the ocular surface and how early MIGS can protect conjunctival health and future surgical outcomes.
Apply evidence-based criteria for IOL selection in glaucoma, balancing contrast sensitivity, disease progression, and patient visual needs.
Dr Nikhil L. Kumar
BMED GradDipMed(RefSurg) MPH FRANZCO FWCRS
Medical Director and Principal Ophthalmic Surgeon
Vision Clinic Sydney and Narellan Eye Specialists, NSW
Dr Ilesh Patel
MBBS MPH FRANZCO
ophthalmologist
Hornsby Eye Specialists and Chatswood Eye Specialists
Dr Nikhil L. Kumar and Dr Ilesh Patel outline practical strategies – from minimally invasive glaucoma surgery (MIGS) to laser trabeculoplasty – to protect the ocular surface, optimise IOP, and set patients up for sharper vision and more reliable cataract outcomes.
Glaucoma management today looks markedly different from a decade ago. A move away from the traditional ‘drops-first’ approach towards a more proactive, interventional model is occurring. This paradigm shift is fundamentally changing what eye health professionals offer our patients.
Central to this evolution is what we term ‘interventional glaucoma’ – the ability to achieve stable intraocular pressure (IOP) control while minimising the patient’s daily treatment burden.
Traditionally, elevated IOP meant reaching for the drop bottle. However, mounting evidence shows the significant hidden costs of the ‘drop-first’ approach.
By recognising the interventional window earlier, optometrists can play a pivotal role in preserving the ocular surface, optimising refractive outcomes, and guiding patients toward long-term, sustainable glaucoma care.
The true cost of topical therapy
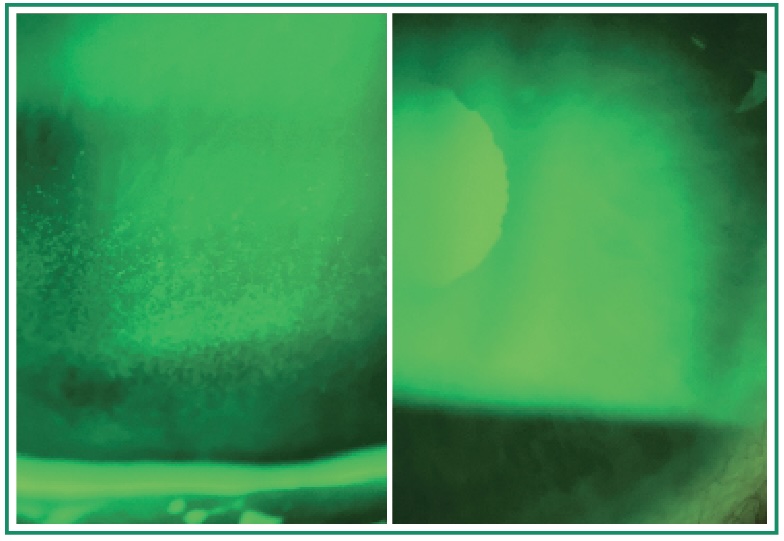
Chronic topical hypotensive therapy directly causes substantial ocular surface adverse effects. Current estimates suggest up to 60% of glaucoma patients develop concomitant ocular surface disease (OSD), with prevalence increasing in direct proportion to the number of medications prescribed.1
The primary culprit remains benzalkonium chloride (BAK), a quaternary ammonium compound that functions as a detergent, disrupting the tear film lipid layer and triggering evaporative dry eye.
Beyond this mechanical effect, BAK induces a pro-inflammatory cascade, upregulating cytokines such as IL-6 and IL-8 while promoting infiltration of T-lymphocytes and macrophages into the conjunctiva.2
This chronic subclinical inflammation leads to goblet cell loss and squamous metaplasia of the ocular surface epithelium.
Implications of OSD
The consequences extend beyond patient comfort. There are direct implications for corneal and lenticular surgery, as well as future, more invasive glaucoma surgeries.
OSD can adversely alter biometric accuracy for cataract surgery and also contribute to refractive inaccuracies post cataract surgery due to a chronically unstable tear film.
A chronically inflamed conjunctiva predicts higher failure rates in future subconjunctival filtration surgeries due to enhanced fibrotic responses.3
Long-term topical therapy-induced OSD is associated with increased conjunctival fibroblast activation, inflammatory cell infiltration, and extracellular matrix deposition, all of which compromise bleb formation and longevity. These pre-existing surface changes reduce the success of trabeculectomy and other subconjunctival procedures by accelerating post-operative scarring despite the use of antimetabolites.
By shifting toward interventional glaucoma through technologies like selective laser trabeculoplasty and trabecular micro-bypass stents, we are not merely lowering pressure, we are reclaiming the ocular surface from chemical assault.
Our goal is to break the ‘preservative cycle’ before irreversible damage occurs.4
Balancing vsual expectations with progressive neuropathy
Discussing the selection of appropriate intraocular lenses (IOL) for glaucoma patients requires careful navigation.
When patients request complete ‘spectacle-free’ vision, trifocal IOLs frequently top their wish list. However, in glaucoma patients, these lenses are rarely, if ever, appropriate. The fundamental problem lies in how these lenses work: they split incoming light into multiple foci which reduces contrast sensitivity.
This is critically important because glaucoma primarily erodes contrast sensitivity, often well before affecting Snellen acuity.5
So, layering a trifocal lens atop a compromised optic nerve will significantly degrade visual quality, particularly in low-light conditions.6
Moreover, the unpredictable nature of glaucoma progression adds another layer of complexity. An IOL that performs adequately today may become a visual liability as the disease advances.
If a patient develops an arcuate scotoma years after surgery, the split-light optics of a trifocal may exacerbate functional deficits, creating a ‘double-jeopardy’ of visual loss.7
The EDOF solution: Expanding the safe zone
Patients with mild-to-moderate, stable glaucoma may be excellent candidates for extended depth of focus (EDOF) IOLs. These lenses provide a continuous range of vision with substantially lower impact on contrast sensitivity compared to diffractive multifocals.8
Key advantages of EDOF lenses in glaucoma include:
• Preserved contrast sensitivity: Multiple studies show EDOF lenses maintain contrast sensitivity closer to monofocal levels, unlike trifocals which show significant degradation9,10
• No impact on visual field testing: Unlike trifocal IOLs, EDOF lenses demonstrate no difference in visual field sensitivity on standard automated perimetry11
• Reduced dysphotopsias: Refractive EDOF designs like the Tecnis PureSee minimise glare and halos that can be particularly troublesome for glaucoma patients12
• Superior low-light performance: Especially important given that glaucoma patients already struggle with reduced contrast
Contemporary EDOF options
The current Australian market offers several viable EDOF choices for glaucoma patients:
Non-diffractive EDOF (Vivity-type): These use wavefront-shaping technology rather than diffractive rings, providing extended range while minimising photic phenomena. They excel for distance and intermediate vision, though near vision may require occasional reading glasses.13
Next-generation refractive EDOF (TECNIS PureSee): Designed to provide smooth extended vision with visual side effects comparable to monofocal IOLs. Recent comparative studies show superior intermediate and near vision versus enhanced monofocals while maintaining excellent distance vision.14
Enhanced monofocal IOLs (Eyhance, RayOne EMV): While not true EDOF lenses, these offer mild depth of focus extension with minimal compromise to visual quality, making them suitable for patients with moderate glaucoma or those particularly concerned about visual disturbances.15
Special considerations for pseudoexfoliation
As discussed at AAO 2025, pseudoexfoliation patients require particularly individualised approaches.7
While advanced lenses can benefit these patients, considerations must include:
• Capsular bag and zonular stability (critical for centration and toric correction)
• Disease severity and progression risk
• Pupil dynamics (essential for diffractive optics)
• Long-term visual function, not just short-term acuity
The critical role of ocular surface health
Success with any premium IOL – particularly EDOF technology – depends entirely on a pristine ocular surface. Accurate biometry and keratometry become impossible with BAK-induced keratopathy. Irregular astigmatism from a poor tear film can cause significant IOL power calculation errors.
This is where preoperative early intervention such as selective laser trabeculoplasty or MIGS becomes transformative.
By eliminating or reducing topical medications, we stabilise the tear film, ensure accurate IOL calculations, and ultimately deliver the refractive outcomes our patients expect.
When cataract surgery is combined with procedures like iStent infinite, we can offer a ‘refractive-plus’ outcome: stabilising IOP, reducing medication burden, and providing high-quality functional vision.16
The power of multi-stent MIGS
The traditional treatment pyramid has been flattened. We now view minimally invasive glaucoma surgery (MIGS) not as a last resort but as a foundational pillar of care. Among these technologies, trabecular micro-bypass systems have established new benchmarks for safety and efficacy.
The three-stent advantage
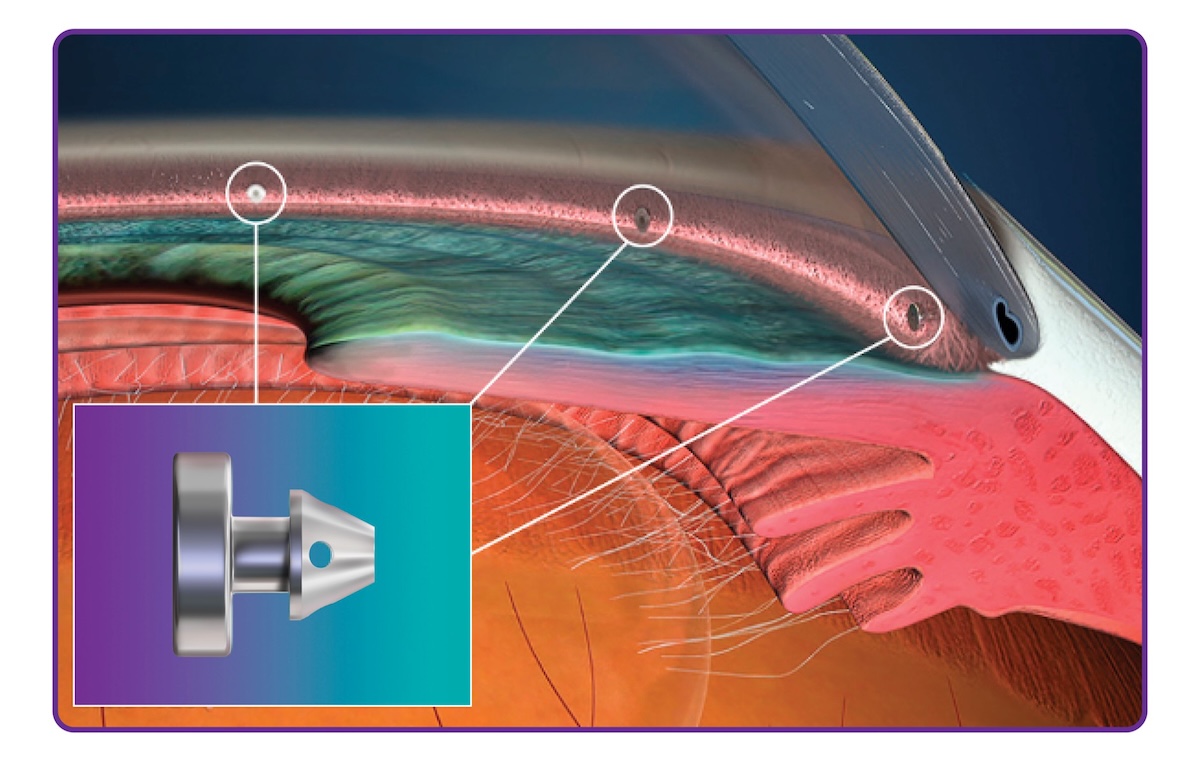
While early MIGS focused on single-point drainage, current evidence supports a more comprehensive approach to the trabecular meshwork. The iStent infinite system utilises three heparin-coated titanium stents placed across a wider arc of the canal (approximately 240° or 8 clock hours), addressing the natural segmental flow of the eye’s drainage system.17
Standalone versatility
Perhaps the most significant shift for optometric practice is that MIGS is no longer strictly utilised solely in conjunction with cataract surgery. The iStent infinite system carries standalone indication in Australia, meaning we no longer must wait for cataract development before offering surgical solutions to patients struggling with IOP control or medication intolerance.
This fundamentally changes our referral paradigm.
A 55-year-old professional with mild glaucoma, suffering from drop-induced OSD but still phakic, now has an interventional option that can eliminate medications while preserving their ocular surface for future refractive cataract surgery.
Recognising the interventional window
As primary providers, optometrists are well placed to identify the “interventional window” – that critical moment when a patient’s current therapy no longer serves their best interests.
Clinical signals warranting referral
• The “fluctuator”
Patients with significant visit-to-visit IOP variations, often signalling poor adherence or “white coat” adherence. These patients may benefit more from 24/7 mechanical pressure control than additional medications they do not use consistently.
• Ocular surface distress
Punctate keratitis, follicular conjunctivitis, or reduced tear break-up time suggests the “drop-induced” pathology may be causing more harm than the glaucoma itself. Early intervention preserves the conjunctiva for both current quality of life and future glaucoma surgical success.
• The refractive seeker
Patients planning cataract surgery who desire EDOF outcomes require IOP stabilisation and ocular surface optimisation to ensure accurate biometry and long-term success. Addressing glaucoma before cataract surgery allows for proper surface healing and informed IOL selection.
• The steroid responder
Patients developing IOP spikes secondary to necessary steroid therapy (intravitreal injections, post-keratoplasty, inflammatory conditions) represent ideal candidates for trabecular bypass to re-establish physiologic outflow.18
Case vignettes: Real-world applications
Case 1: The refractive standalone success
Patient: 55-year-old software architect with mild-to-moderate controlled open-angle glaucoma.
Presentation: Bilateral IOP 22 mmHg on latanoprost monotherapy. Complained of debilitating dry eye symptoms interfering with 10+ hour daily screen work. Desired premium refractive outcome for anticipated cataract surgery but currently phakic with minimal lens changes.
Intervention: Bilateral staged standalone iStent infinite procedures.
Outcome: Post-operatively achieved bilateral IOP of 15 mmHg on zero medications. Ocular surface recovered dramatically within 8 weeks, with TBUT improving from <5 seconds to 9 seconds. Stable biometry now established for future EDOF IOL planning when cataract progresses. Patient reports the procedure “gave me my life back” in terms of comfort and work productivity.
Case 2: The steroid-induced spike
Patient: 62-year-old with persistent diabetic macular oedema and moderate glaucoma on topical dual therapy.
Presentation: Required regular intravitreal dexamethasone implants for vision-threatening macular oedema. Developed severe steroid-induced IOP spikes reaching 34 mmHg despite maximum tolerated medical therapy. Facing choice between vision loss from oedema or optic nerve damage from pressure.
Intervention: iStent infinite trabecular micro-bypass to re-establish physiologic outflow capacity.
Outcome: IOP stabilised at 14-16 mmHg for 18+ months, allowing continued steroid treatment without optic nerve damage. Patient maintained visual acuity gains from oedema treatment without glaucoma progression.
Case 3: The surface-damaged multi-drop patient
Patient: 68-year-old retired teacher with moderate POAG.
Presentation: On three topical medications (latanoprost, dorzolamide/timolol, brimonidine), IOP marginally controlled at 18-19 mmHg. Severe ocular surface disease with Grade 3 punctate keratitis, severe discomfort, and fluctuating vision affecting reading – her primary hobby.
Intervention: Combined cataract surgery with iStent infinite, EDOF IOL.
Outcome: Twelve months post-operatively: IOP 13 mmHg on zero medications, complete resolution of OSD, stable TBUT of 8 seconds, uncorrected distance vision 6/6, requiring only +1.50 readers for sustained near work. Patient describes the outcome as “life-changing – I can finally enjoy reading again without constant discomfort.”
Conclusion: Shaping the future of care
The future of glaucoma management is micro-invasive, targeted, and genuinely patient-centric.
As Australian ophthalmologists and optometrists, we serve as architects of our patients’ visual journeys. By recognising both the limitations of medical therapy – particularly its impact on ocular surface health and future refractive choices – and embracing evidence-based interventional technologies, we can offer a meaningfully higher standard of care.
The key is identifying that critical window when intervention serves the patient better than another prescription. For many of our patients, that window is far earlier than traditional paradigms suggested.
Dr Nikhil L. Kumar is a cataract, refractive and cornea subspecialist practicing at Vision Clinic Sydney and Narellan Eye Specialists. Dr Ilesh Patel is a cataract and glaucoma specialist at Hornsby Eye Specialists. Both authors are passionate about advancing patient-centred approaches to combined cataract and glaucoma management in Australia.
References:
Micheletti JM, Shultz M, Singh IP, Samuelson TW. An emerging multi-mechanism and multi-modal approach in interventional glaucoma therapy. Ophthalmol Ther. 2024;13(1):13–22.
Zhang X, Vadoothker S, Gottlieb JL, Liu Y. The impact of benzalkonium chloride on the ocular surface: a systematic review. J Ocul Pharmacol Ther. 2024;40(8):456–68.
Broadway DC, Grierson I, O’Brien C, Hitchings RA. Adverse effects of topical antiglaucoma medication. II. The outcome of filtration surgery. Archives of Ophthalmology (Chicago, Ill. : 1960). 1994 Nov;112(11):1446-1454. DOI: 10.1001/archopht.1994.01090230060021. PMID: 7980134.
Gomes JAP, Azar DT, Baudouin C, et al. TFOS DEWS II iatrogenic report. Ocul Surf. 2024;22:511–38.
Hong ASY, Ang BCH, Dorairaj E, Dorairaj S. Premium intraocular lenses in glaucoma: a systematic review. Bioengineering. 2023;10(9):993.
Harasymowycz P. Multifocal, EDOF, and light adjustable IOLs in glaucoma. AGS 2024 interview. American Academy of Ophthalmology Education. February 2025.
Berdahl J. Advanced IOL selection for pseudoexfoliation patients. Presentation at AAO 2025. Ophthalmology Times. February 2025.
Peterson CL, Wong TT, Perera S. Editorial: insights in glaucoma: 2023. Front Ophthalmol. 2025;4:1519088.
Kohnen T, Suryakumar R. Extended depth-of-focus technology in intraocular lenses. J Cataract Refract Surg. 2020;46:298–304.
Lee S, Kim J, Park SH, et al. Comparative outcomes of the next-generation extended depth-of-focus intraocular lens and enhanced monofocal intraocular lens in cataract surgery. J Clin Med. 2025;14(14):4967.
Takahashi M, Yamashiro C, Yoshimoto T, et al. Influence of extended depth of focus intraocular lenses on visual field sensitivity. Sci Rep. 2024;14:8856.
Asena L, Kırcı Dogan İ, Oto S, Dursun Altınors D. Comparison of visual performance and quality of life with a new nondiffractive EDOF intraocular lens and a trifocal intraocular lens. J Cataract Refract Surg. 2024;50:847–54.
Rocha KM. Extended depth of focus IOLs: the next chapter in refractive technology? J Refract Surg. 2017;33:146–9.
Johnson & Johnson Vision. Tecnis PureSee EDOF IOL clinical data. 2024–2025.
Sharpe D. Comprehensive comparison of multifocal and EDOF IOLs available in the USA (2024–2026). Sharpe Vision Blog. March 2025.
Singh IP, Shultz M. Refractive outcomes in glaucoma: the role of EDOF and the danger of multifocality. J Cataract Refract Surg. 2025;51(4):442–50.
Glaukos Corporation. iStent infinite Trabecular Micro-Bypass System: Instructions for Use. 2024.
Donnenfeld ED, Solomon KD, Voskanyan L, et al. Third-generation trabecular micro-bypass implantation with phacoemulsification for glaucoma. Ophthalmol Ther. 2025;14:257–71.