Fly wings are exquisite, versatile biological actuators. During flight, they sweep back and forth through the air hundreds of times per second to keep the fly aloft. On the ground, flies extend their wings to groom, and males vibrate a wing to attract females during courtship. To accomplish these myriad functions, wing motor control relies on temporally and spatially precise feedback from diverse sensory neurons distributed throughout the wing (Figure 1A). Proprioceptive mechanosensory neurons play a particularly important role in flight control (Pringle, 1957), as mechanosensory feedback has a shorter latency than visual signals and can, therefore, be used to rapidly adjust wing motion (Dickerson, 2020). Wings experience dynamic forces during flight, and proprioceptors encode features of these forces, such as wing bending, twisting, and load (Dickerson et al., 2021). Drosophila typically beat their wings at 200–250 Hz and can adjust wing kinematics from one stroke to the next (Dickinson et al., 1993; Heide and Götz, 1996). Thus, muscle contraction must be temporally precise enough to act at these short time scales (Dickinson and Tu, 1997). Consistent with the need for rapid feedback, some motor neurons that control wing steering muscles receive monosynaptic input from wing sensory neurons (Fayyazuddin and Dickinson, 1999). However, the peripheral location and identity of the wing sensory neurons that provide feedback to the wing motor system remain largely unknown.
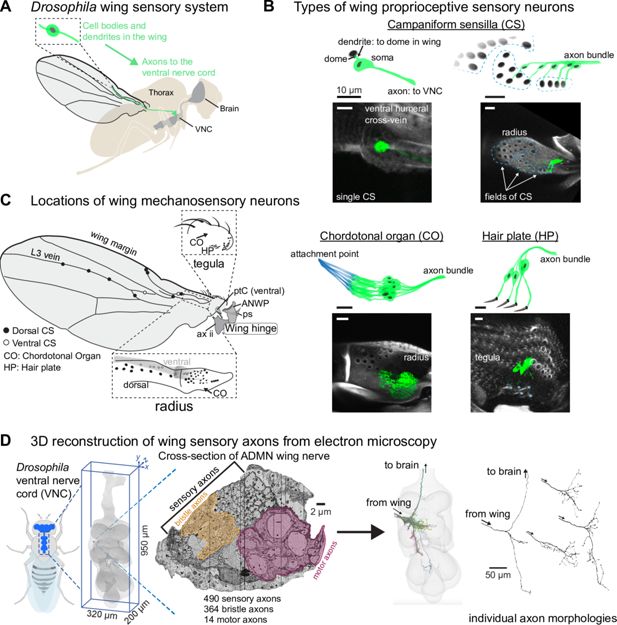
Proprioceptive neurons on the Drosophila wing.
(A) The cell bodies and dendrites of sensory neurons are in the periphery, on the wing and wing hinge, and their axons project to the ventral nerve cord (VNC). Before entering the VNC, the sensory axons fasciculate together and enter through the Anterior Dorsal Mesothoracic Nerve (ADMN). (B) Proprioceptors on the wing include campaniform sensilla (CS), chordotonal organs (CO), and a hair plate (HP). Each campaniform sensillum dome is innervated by a single sensory neuron, as is each hair in a HP. A CO is made up of a group of sensory neurons with supporting cells that fix them to the underside of the cuticle (shown in blue). Blue asterisks (bottom right) indicate a single external hair in the HP. Images show the membrane-bound fluorescent label mCD8::GFP to highlight each proprioceptor type. See Appendix 1—table 3 for details on which proprioceptors are labeled by which driver lines, driver lines for representative images in this panel are: single CS (12C07-GAL4); field CS (10G03-GAL4); CO (15F10-GAL4); HP (16C09-GAL4). Scale bars are 10 μm. (C) Location of sensory neurons on the wing and wing hinge. The location of sensory neurons and the number of CS in each field are based on confocal images and a prior study (Dinges et al., 2021). A subset of sclerites and other structures that make up the wing hinge are included as landmarks: pterale C (ptC), the anterior nodal wing process (ANWP, which also features three CS), the parascutal shelf (ps), and the second axillary (ax ii). (D) We reconstructed each sensory axon in the ADMN wing nerve to visualize its full morphology and analyze downstream connectivity in the VNC. More information on each of these steps is in Azevedo et al., 2024. In the nerve cross-section, the motor domain and margin bristle domains are highlighted by outlined yellow and mauve masks.
Sensory neurons on the Drosophila wing can be grouped into different classes based on their end-organ morphology (Figure 1B). The most numerous are the bristles along the wing margin, which include both mechanosensory and chemosensory sensilla (Hartenstein and Posakony, 1989; Palka et al., 1979). Wing chemosensory neurons can detect external odors and pheromones (Stocker, 1994), while mechanosensory bristles can detect the presence of dust particles or mites (Hampel et al., 2017; Li et al., 2016). Bristles also line the tegula, a cuticular protuberance at the proximal edge of the wing. Apart from the tactile and chemosensory bristles, other sensory neuron classes are presumed to be proprioceptive, in that they monitor the movement and strain of the wing itself. These include campaniform sensilla, chordotonal organs, and hair plates, all of which occur at other locations across the adult fly body, including the legs (Dinges et al., 2021; Field and Matheson, 1998). Each campaniform sensillum (CS) consists of a single neuron with a dendrite that contacts a cuticular cap, or dome, on the surface of the wing; the CS neuron fires action potentials when the dome deforms (Chapman et al., 1973; Moran et al., 1971; Pringle, 1938a). CS can be found alone or in fields of domes that have similar sizes and orientations (Cole and Palka, 1982; Dinges et al., 2021). A chordotonal organ (CO) is a cluster of neurons with cap cells that anchor the dendrites to an internal structure, such as a tendon (Field and Matheson, 1998). In the wing, they are anchored to inner extensions of the cuticle; for example, the wall of the tegula and the inner wall of the radius (sometimes called the radial vein). A hair plate (HP) is a small, tightly packed cluster of sensory hairs, each of which is innervated by a single mechanosensory neuron (Pringle, 1938b). Proprioceptive neurons (CS, CO, and HP) are concentrated proximally, especially along the radius and the tegula (Figure 1C). The axons of wing sensory neurons project into the fly’s ventral nerve cord (VNC), the invertebrate analog of the spinal cord. Previous work has described the activity of fly leg proprioceptors during walking (Dallmann et al., 2024; Pratt et al., 2026), but it has been prohibitively challenging to record activity of wing sensory neurons during flight.
Much of what we know about wing sensory neurons comes from developmental studies that used the fly wing as a model to investigate whether axonal morphology is intrinsically determined or extrinsically directed. Some studies used mosaic mutant flies with hindwings in place of halteres to test whether sensory axons would follow haltere-like morphologies or wing-like morphologies once they entered the developing central nervous system (Ghysen, 1978; Palka et al., 1979). These studies measured morphological similarities between wild-type and mutant axons to uncover their intrinsic developmental programs. Their findings showed that the degree of intrinsic programming was different for single CS and field CS, in that axons from field CS on the mutant hindwings followed similar paths in the VNC to the field CS on wild-type halteres, while the axons of single CS on mutant hindwings retained the morphological characteristics of the wild-type forewing single CS axons (Palka et al., 1979). This difference suggests that the field and single CS are endowed with different axon guidance instructions, connect to different postsynaptic partners, and thus may serve distinct functions.
Understanding how central circuits integrate information from wing sensory neurons is key to understanding their function. Connectomics, or dense reconstruction of neurons and synapses from electron microscopy, offers new opportunities for mapping peripheral sensory feedback to the CNS (Galili et al., 2022). In this study, we bridge the gap between a VNC connectome and the wing by mapping central axon morphologies to the peripheral structures from which they originate (Meinertzhagen et al., 2009). We reconstructed all 490 afferents in the left wing nerve (Anterior Dorsal Mesothoracic nerve, ADMN) in the FANC electron microscopy dataset (Azevedo et al., 2024; Phelps et al., 2021). Many axon morphologies and their corresponding peripheral end-organs were previously undescribed. We identified genetic driver lines for a subset of these unknown wing sensory neurons and elucidated their peripheral location and anatomy. For example, we identified novel classes of peripheral sensory neurons near the wing hinge and found that CS on the tegula synapse onto the tonic wing b1 motor neuron, suggesting a specialized role in feedback control of flight steering. We also confirmed a long-standing prediction that individual CS from the same field can have distinct axon morphologies (Palka et al., 1986). A companion paper that reconstructed haltere CS axons in the connectome identified a similar organization (Dhawan et al., 2026). Overall, knowing the relationships between peripheral neuroanatomy, axon morphology, and downstream connectivity to wing motor neurons provides a foundation for investigating proprioceptive sensing and motor control of the fly wing.
