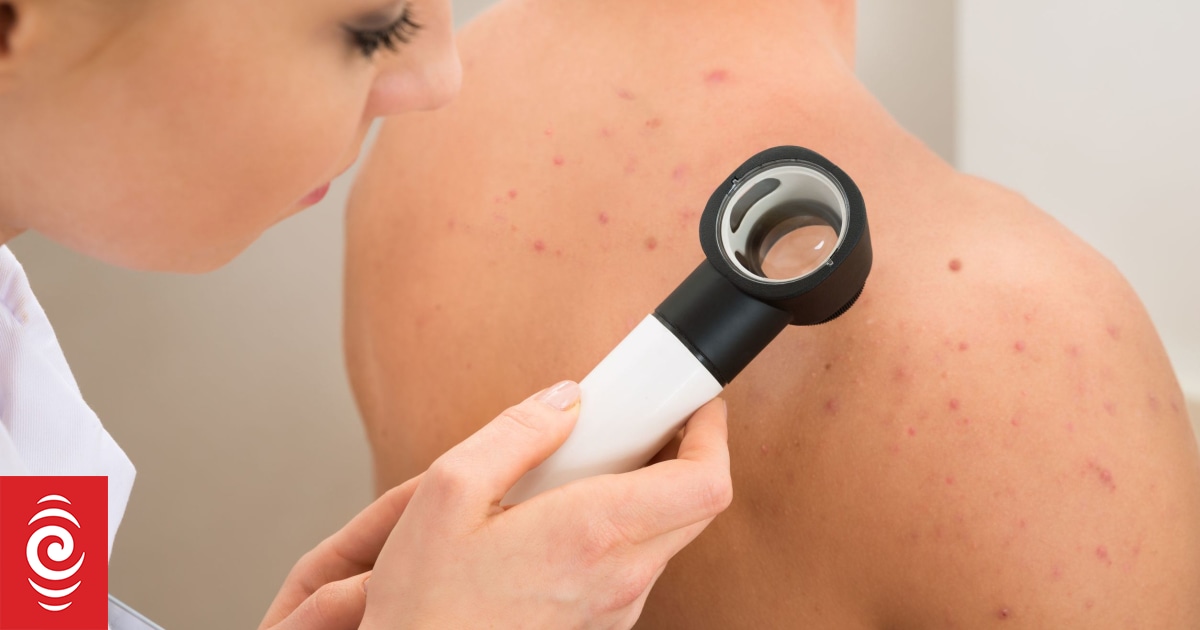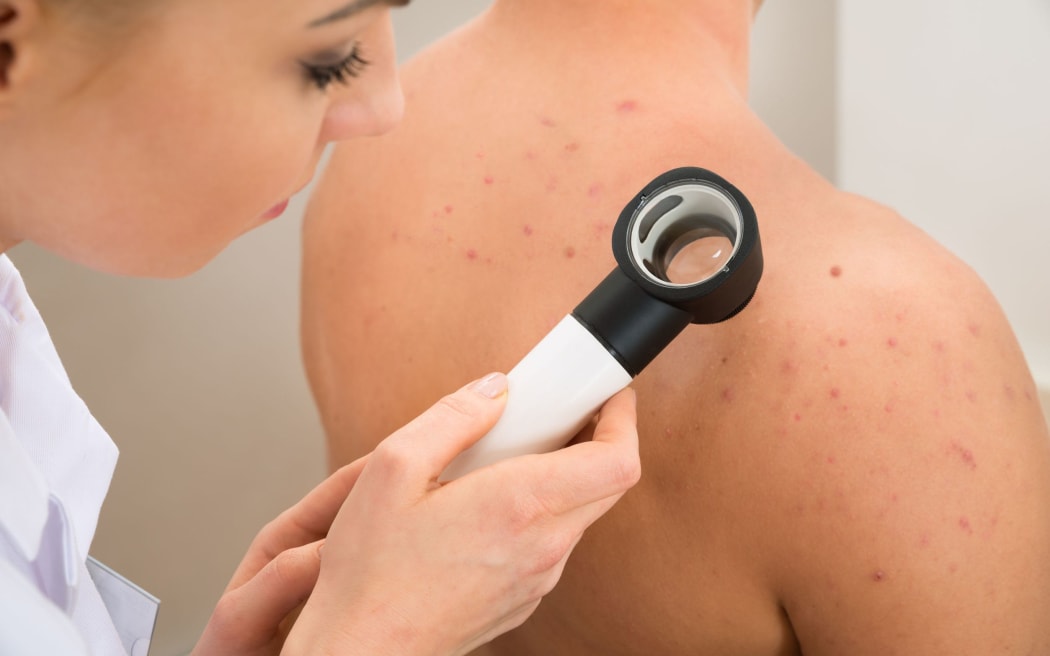
From 1 May, people with stage 3-B to stage 4 melanoma that can be removed with surgery will be eligible for funded nivolumab and ipilimumab before their operation.
Photo: 123rf.com
Pharmac is widening access to two melanoma medicines, giving cancer patients more funded immunotherapy options.
From 1 May, people with stage 3-B to stage 4 melanoma that can be removed with surgery will be eligible for funded nivolumab and ipilimumab before their operation.
Pharmac manager of pharmaceutical funding, Claire Pouwels, said these were immunotherapy medicines that helped the immune system fight melanoma.
“Funding these medicines gives people another effective treatment option.”
Nivolumab and ipilimumab were already approved for use in New Zealand and funded for some other cancers.
Clinical advice provided to Pharmac had shown these treatments could reduce the risk of melanoma coming back.
Around two in three people responded “strongly” to treatment by the time their tumour was removed, with significant tumour shrinkage or very little active disease remaining.
“For many people, that response means they may not need further treatment after surgery,” Pouwels said. “For patients and their whānau, that can mean fewer infusions, fewer hospital visits, and less time spent in treatment.”
This meant reduced demand on hospital oncology infusion services.
Pharmac expected the change to save around 1000 infusion hours each year, reducing pressure on hospital oncology infusion centres and freeing up capacity for others.
Pharmac currently funds pembrolizumab (Keytruda) as the only immunotherapy option for melanoma that can be removed with surgery. Funding nivolumab and ipilimumab would provide another funded immunotherapy choice for people.
Around 155 people would receive this treatment in the first year, increasing to about 166 people per year after five years. Pharmac said most people who receive this treatment would otherwise have received funded pembrolizumab.
During consultation “some people asked if access could be widened further to include different types of melanoma”, Pouwels said.
“While we weren’t able to do that as part of this decision, we will seek further clinical advice to understand the potential impact of wider access in the future.”
Sign up for Ngā Pitopito Kōrero, a daily newsletter curated by our editors and delivered straight to your inbox every weekday.