Maria Carolina Florian explores the complexities of aging and research on the hematopoietic system, particularly focusing on hematopoietic stem cells (HSCs), as well as how rejuvenating strategies targeting the hematopoietic system could enhance healthspan and lifespan
Aging is a complex and fascinating biological process, which broadly reflects the decline of organismal and cellular function over time. While the community is building a consensus over the hallmarks of aging(1), the identification of druggable targets that would not only correct the specific dysregulated hallmark, but also more holistically improve healthspan and lifespan of the organism is still highly controversial.
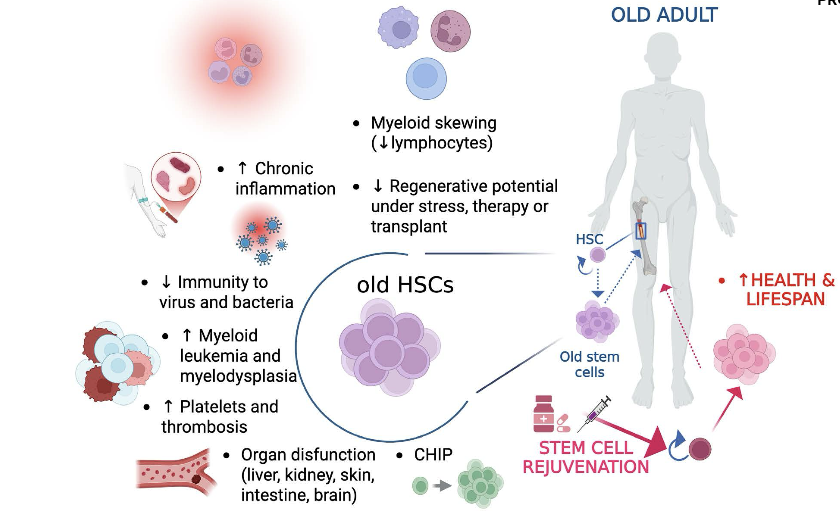
Alterations of the hematopoietic system over time
Among other tissues, aging of the hematopoietic system is characterized by phenotypic and functional impairments that are driven by alterations of hematopoietic stem cells (HSCs) and of their bone marrow (BM) niche(2). HSCs are responsible for the production of all the different cell types that constitute the blood, and their maintenance and differentiation must be tightly regulated during the whole life of an organism. In adult mammals, HSCs mainly reside in the BM, which not only supports HSC activity but also adjusts blood cell production to meet physiological demands in response to insults over time(3–10). The BM presents with a complex anatomical architecture, which is characterised by discrete microanatomical production sites that confer plasticity to hematopoiesis and support function of the different hematopoietic stem and progenitor cells(6,11). A microanatomical site that supports HSC function is generally identified as HSC niche and, similarly to HSCs themselves, it persists through life and it is affected by aging.
Recent findings that characterize alterations in HSCs and their bone marrow (BM) niche over time(7,10,12–14), immunosenescence and inflammaging(15), as well as emerging data regarding clonal hematopoiesis of indeterminate potential (CHIP)(16), that originates as a consequence of somatic mutations in HSCs and leads to clonally restricted hematopoiesis, together support a central role of the blood for organismal aging and the notion that the hematopoietic tissue represents an elective target for rejuvenating strategies.
During life, HSCs and their niche may be subjected to changes that affect their own function, as well as the maintenance and interactions with other hematopoietic and non- hematopoietic cells. Alterations of the BM niche and systemic changes, such as inflammation, disruption of the circadian rhythm or other secreted signalling factors and hormones that impact on HSC function, are broadly known as extrinsic causes of aging(17–20) , whereas intrinsic HSC aging is independent from the (micro-) environment and refers to all the alterations that affect the homeostasis of HSCs that are only dependent on the HSC itself(21–24). Some examples of these intrinsic alterations, among others, are at the level of DNA damage(25–27), epigenetics (DNA hypermethylation and histone post-translational modifications(28–32), H4K16ac polarity(33–36), increased overall chromatin accessibility(22,37) and increased nuclear envelope tension(38)), metabolic alterations (high ROS levels(39), mitochondrial stress(40)), disruption of protein homeostasis (proteostasis; low levels of autophagy(41)) and of signalling factors (TGFβ(42), Notch(12,21,43)).
Clonal hematopoiesis of indeterminate potential and age-related conditions
Among the different alterations of the aged hematopoietic system(44,45), CHIP has gained considerable attention by the scientific community in the last decade. CHIP is an aging-related phenomenon, in which HSCs or other early blood cell progenitors contribute to the formation of a genetically distinct subpopulation (clone) of blood cells with a variant allele frequency equal or more than 2%(16,46). The somatic mutations in hematopoietic cells that lead to clonal expansion are commonly acquired during human aging, and clonally restricted hematopoiesis is associated with an increased risk of several aging-related diseases and increases all-cause mortality(47). The expansion of clones carrying mutations in genes connected to specific leukemic malignancies in otherwise hematologically healthy individuals was described first in 2014(48), with a further link between CHIP and cardiovascular disease (CVD) described just shortly after(49,50). Since these pioneering discoveries, CHIP has been associated with many other non-hematopoietic diseases. For example, there are data linking CHIP to the development of cancer in the digestive system(51,52), to inflammatory diseases such as ulcerative colitis(53), to kidney disease(54) and to adverse outcomes after COVID- 19-infections(55). Moreover, CHIP not only has an increased prevalence in the elderly, it is also linked to accelerated organismal aging as measured by using DNA-methylation based epigenetic clocks(56,57).
Research on the aged hematopoietic system
The idea that the hematopoietic system drives aging, as well as healthspan and lifespan of the whole organism dates back to several centuries and was first empirically proven in the parabiosis experi-ment described in the late 1950s, when parabiotic pairs between young and old rats were generated(58). Later on, heterochronic parabiosis has been shown to rejuvenate aged skeletal muscle, liver, heart, pancreas, bone, spinal cord and brain, ultimately leading to an extension of life and health- span(59–64). Supporting these findings, it was also demonstrated that young bone-marrow transplanta-tion attenuates cognitive decline in old mice, with preservation of hippocampal synapses and re-duced microglial reactivity(65). However, several decades after the very first experiments, the effect of heterochronic parabiosis is still highly controversial(66,67). Indeed, it has been recently demonstrated that aged HSCs are resistant to bloodborne systemic rejuvenation approaches(67).
Aged HSCs appear to be refractory also to other systemic rejuvenation strategies, such as exercise(67). Intriguingly, life-long caloric restriction (CR), which represents the most reproducible and robust intervention so far known to effectively delay the onset of age-related diseases and extend lifespan in multiple species, was extensively investigated in a recent study68. In contrast to the expected health and metabolic traits, which strikingly were not found to be significantly associated with lifespan extension, the study revealed that instead many immunological and hematological traits were strongly associated with lifespan. For example, the percentage of circulating lymphocytes with-in peripheral blood cells was positively linked to lifespan, with percentage of circulating CD4+ and CD8+ naïve T cells and immature NK cells positively correlated with lifespan, whereas CD4+ and CD8+ effector T cells and CD11+ memory B cells are associated with a shortened lifespan(68). Haemo-globin levels were improved (increased) under CR and some of the erythroid traits exhibited signifi-cant positive associations (haemoglobin, hematocrit, red blood cell count) while other showed nega-tive associations (RDW, HDW) with lifespan(68). Supporting the notion that improvements to the aged hematopoietic system might impact on healthspan and lifespan, we have shown that a 4-day long systemic treatment of aged (75-80 week-old) female mice with CASIN, the inhibitor of the small Rho GTPase Cdc42 that has been shown to rejuvenate aged HSCs(33,35), significantly increases murine median and maximum lifespan(69,70). Further, it was shown that upon transplantation, rejuvenated blood stem cells are sufficient to increase murine healthspan, as well as median and maximum lifespan of aged immunocompromised mice, providing additional support for a central role of the hematopoietic system in aging and proof-of-principle evidence that rejuvenating HSCs could indeed extend lifespan(71–73). Moreover, we have recently identified another possibility of intervention to rejuvenate aged HSCs by targeting the alteration in nuclear mechanosignalling due to overactivated RhoA, via ex vivo treatment with the small molecule inhibitor RHOSIN(38). In addition, we have iden-tified targetable aging-associated alterations in the BM niche, with a shift in Notch signalling driven by endothelial cells impairing function of aged HSCs(12).
Refining and defining aging research
Aging represents a fascinating biological process that despite many years of research remains a challenge to explain and treat. Similar to other biological processes, aging can now be explained and possibly targeted to counteract its effects, such as somatic stem cell aging and eventually tissue and organismal functional decay, over time. Rigorous clinical investigations are needed in order to validate and refine current findings and define therapeutic interventions for the elderly population. To this end, the scientific community should be call into action also in the direction of revisiting standard clinical trials to open at including new specific parameters of aging for clinical trial evaluation and as clinical endpoint criteria. For example, CHIP, epigenetic clocks, frailty, response to vaccination, RBC, relative percentage in blood of B and T lymphocytes type/levels, number of platelets could all be implemented for clinical estimation of hematopoietic aging or organism aging and also as endpoint criteria in clinical trial settings for testing rejuvenating interventions. Considering the predicted increase of people over the age of 65 year in the near future, surpassing already more than 30% of the total populations in many European countries and in Japan, it is now timely to move research forward into translation and to switch our mindset from considering aging as a natural process, to treating aging as the major risk factor for several diseases that affect most of the population, both at present and in the future. As discussed above, we have identified a few interventions which by targeting the aged hematopoietic system hold great promise to decrease our risk of developing many aging- related diseases, as well as to improve our healthspan and possibly lifespan.

Acknowledgments
We acknowledge the following funding sources: European Research Council (ERC) grant 101002453 (MCF), Spanish Ministry of Science, Innovation, and University grants CNS2023-144908 – CONSOLIDACIÓN INVEST. We thank the CERCA Program/Generalitat de Catalunya for institutional support.
Competing interests
M. Carolina Florian discloses financial interest in MoglingBio (https://www.moglingbio.com/). The other authors declare no competing interests.
CLICK HERE for references