An implant is transforming the lives of spinal cord injury patients, addressing a common yet serious complication—unstable blood pressure.
Research led by teams at several universities worldwide, including the University of Calgary, shows that a new implantable system on patients’ spines can restore blood pressure balance after a spinal cord injury.
The findings, published in both Nature and Nature Medicine, describe a targeted therapy to address blood pressure regulation in 14 patients across four clinical studies conducted at three medical centres in Canada, Switzerland, and the Netherlands.
“Blood pressure is a profound issue after a spinal cord injury, both highs and lows,” said Aaron Phillips, an associate dean at the University of Calgary Cumming School of Medicine, who is involved in the research.
“This is because the spinal cord is disconnected from the brain, which is responsible for controlling blood pressure.”
Phillips explains that low blood pressure can lead to fainting and reduced energy, while high blood pressure can increase the risk of a stroke.
“We also know that this blood pressure instability that happens after a spinal cord injury can lead to cardiovascular disease over the long term,” he said.
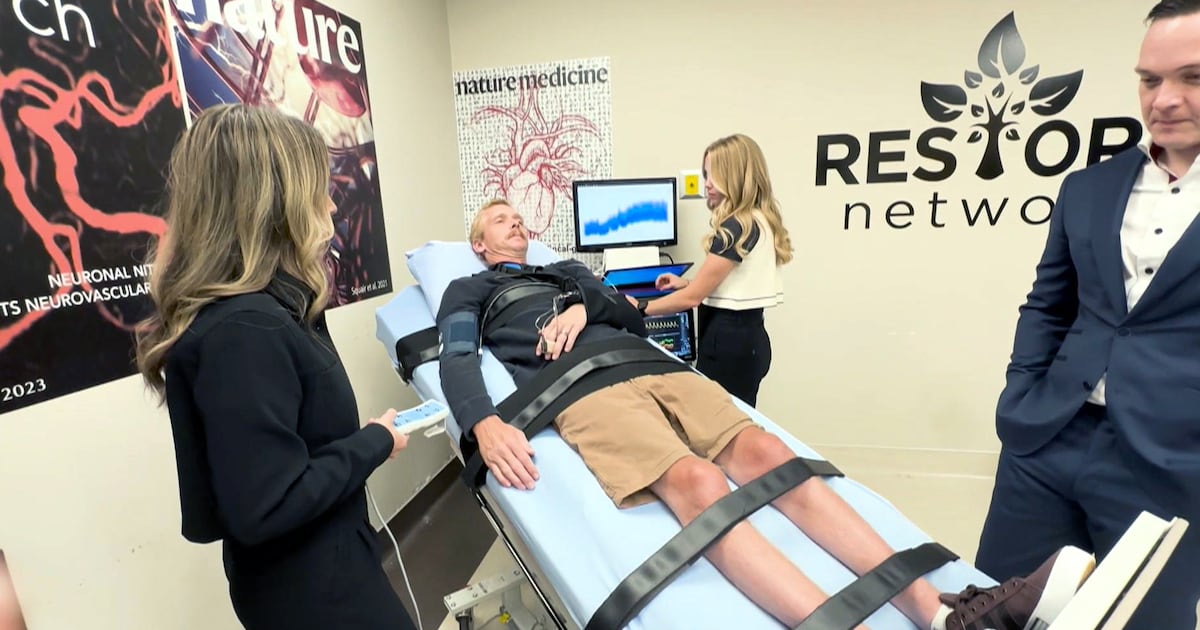
One of the 14 participants in the clinical trial is Cody Krebs from Alberta.
The 32-year-old says a semi-truck blew through a stop sign and t-boned his vehicle in 2022.
“That resulted in me breaking my neck — C6 and C7 level,” said Krebs.
Krebs is now in a wheelchair and the damage to his spinal cord means he can’t regulate his blood pressure.
“I get really lightheaded and dizzy, and my ears can start ringing,” he said.
As part of the trial, Krebs had one of the implantable systems surgically placed in his spine.
“This is a device that is implanted around the spinal cord. It delivers a low electrical current to basically replace the signal the brain would normally give to control blood pressure,” said Dr. Fady Girgis, a neurosurgeon and associate professor at the University of Calgary.
“Surgery, of course, carries some risk, but these devices have been around for a long time and are generally very safe. They can prevent patients from needing to take blood pressure medications.”
The electrical currents in Krebs’ device are controlled externally with a remote. A new prototype has been developed that allows the currents to be delivered without a remote.
Krebs says his quality of life has improved immensely with the implant.
“It’s helped me get back into work because I’m not exhausted all the time,” said Krebs.
“If I have it on throughout the day, I find I can spend more time in the evenings without being tired and having to go to bed early.”
The company that developed the implantable neurostimulation system that were used in the studies has received FDA approval to initiate a pivotal trial of the therapy. The trial is expected to involve about 20 neurorehabilitation and neurosurgical research centers across Canada, Europe and the United States.