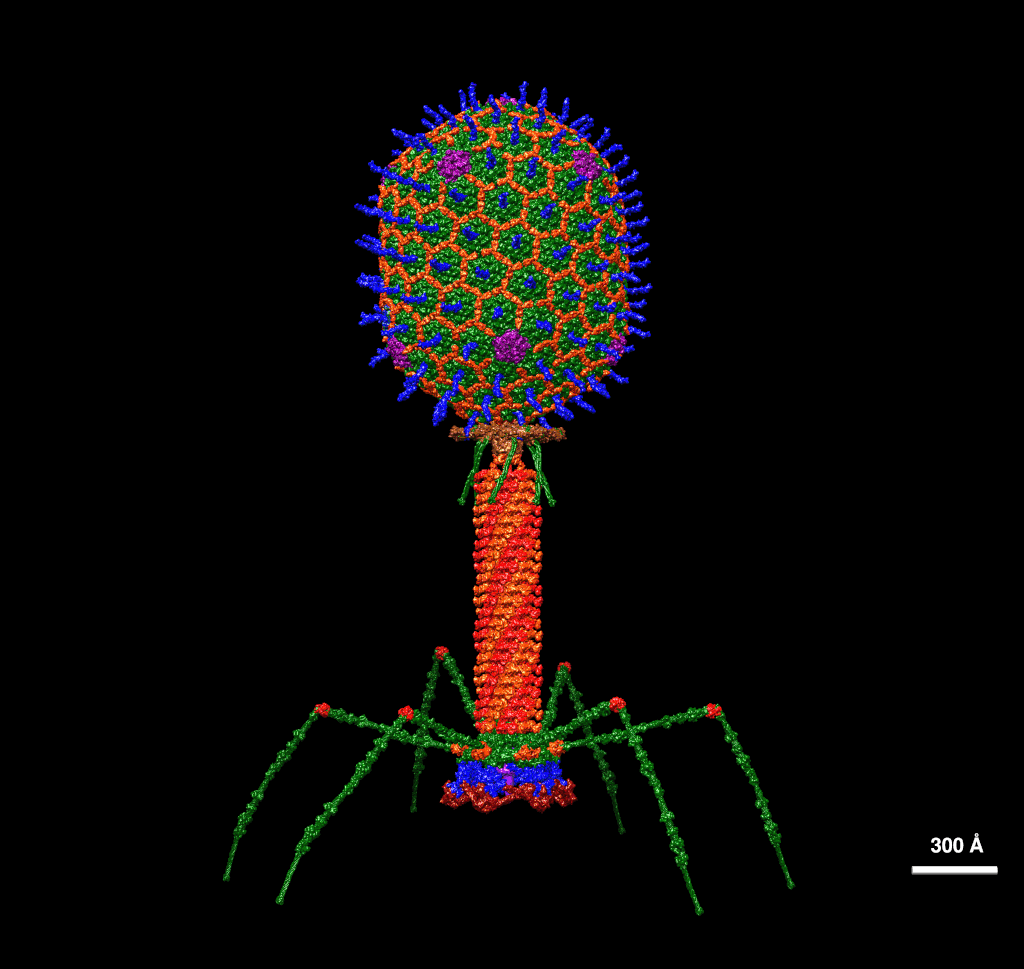
A bacteriophage is a virus that infects and replicates within bacteria. Image by Dr. Victor Padilla-Sanchez, PhD – Own work (CC BY-SA 4.0)
The first fully synthetic bacteriophage engineering system for Pseudomonas aeruginosa, an antibiotic-resistant bacterium of global concern, has been created at Yale University.
The system was enabled by New England Biolabs’ High-Complexity Golden Gate Assembly (HC-GGA) platform. With this method, researchers engineer bacteriophages synthetically using sequence data rather than bacteriophage isolates.
This system divides the 41S1 genome into DNA fragments small enough to be synthesised and to avoid toxicity during DNA propagation, with all but one maintained in Escherichia coli.
These fragments are next readily assembled into a one-pot reaction. After this, phages can be rescued by direct transformation into P. aeruginosa PAO1 or E. coli 10-beta cells.
Bacteriophages, viruses that infect bacteria, have been used as medical treatments for bacterial infections for more than 100 years.
Most phage-based research has remained focused on naturally occurring viruses, largely because traditional methods for modifying phages are slow, complex, and difficult to scale.
Using the new system, the team constructed a P. aeruginosa phage from 28 synthetic DNA fragments. They then programmed the virus with new capabilities by introducing point mutations as well as DNA insertions and deletions. These changes allowed the researchers to swap tail fibre genes to change which bacteria the phage could infect and to add fluorescent markers that made infections visible in real time.
Bio-technological leap
Lead scientist Andy Sikkema, as reported by Pharmaceutical Microbiology Resources, says: “Even in the best of cases, bacteriophage engineering has been extremely labour-intensive. Researchers spent entire careers developing processes to engineer specific model bacteriophages in host bacteria.”
 Mycobacterium tuberculosis (Public Health Image Library, NIAID, Image ID: 18139)
Mycobacterium tuberculosis (Public Health Image Library, NIAID, Image ID: 18139)
Sikkema adds: “This synthetic method offers technological leaps in simplicity, safety and speed, paving the way for biological discoveries and therapeutic development.”
Digital DNA
The platform enables scientists to assemble an entire phage genome outside the cell using synthetic DNA, incorporating all planned genetic changes during construction. Once assembled, the genome is introduced into a safe laboratory strain where it becomes an active bacteriophage.
This strategy avoids many long-standing obstacles in phage research. Traditional approaches depend on maintaining physical phage samples and using specialized host bacteria, which can be especially challenging when working with viruses that infect dangerous human pathogens. The new method also eliminates the need for repeated rounds of screening or step-by-step genetic edits inside living cells.
In contrast with other DNA assembly techniques that combine fewer but longer fragments, the new platform uses shorter DNA segments. These shorter pieces are easier to produce, less toxic to host cells, and less likely to contain errors. The method also works well with phage genomes that contain repeated sequences or extreme GC content, both of which often complicate DNA assembly.
GC content refers to the percentage of guanine (G) and cytosine (C) bases in a DNA or RNA molecule.
By simplifying the process and expanding what is technically possible, this approach significantly broadens the potential for developing bacteriophages as targeted therapies against antibiotic-resistant infections.
Collaboration turns the abstract into therapies
NEB researchers had spent years refining Golden Gate Assembly so it could reliably handle large DNA targets made from many fragments. Yale researchers recognized that these tools could unlock new possibilities in phage biology and reached out to explore more ambitious applications.
 Vaccination – Copyright AFP MANDEL NGAN
Vaccination – Copyright AFP MANDEL NGAN
The scientists first optimized the method using a well-studied model virus, Escherichia coli phage T7. From there, collaborative teams expanded the technique to non-model phages that target some of the most antibiotic-resistant bacteria known.
An earlier study using the same Golden Gate approach to build high-GC content Mycobacterium phages was reported in November 2025 in collaboration with the Hatfull Lab at the University of Pittsburgh and Ansa Biotechnologies.
In another example, researchers from Cornell University partnered with NEB to create synthetically engineered T7 phages that function as biosensors to detect E. coli in drinking water.
Research paper
The findings appear in the journal Proceedings of the National Academy of Sciences, titled “A fully synthetic Golden Gate assembly system for engineering a Pseudomonas aeruginosa phiKMV-like phage.”