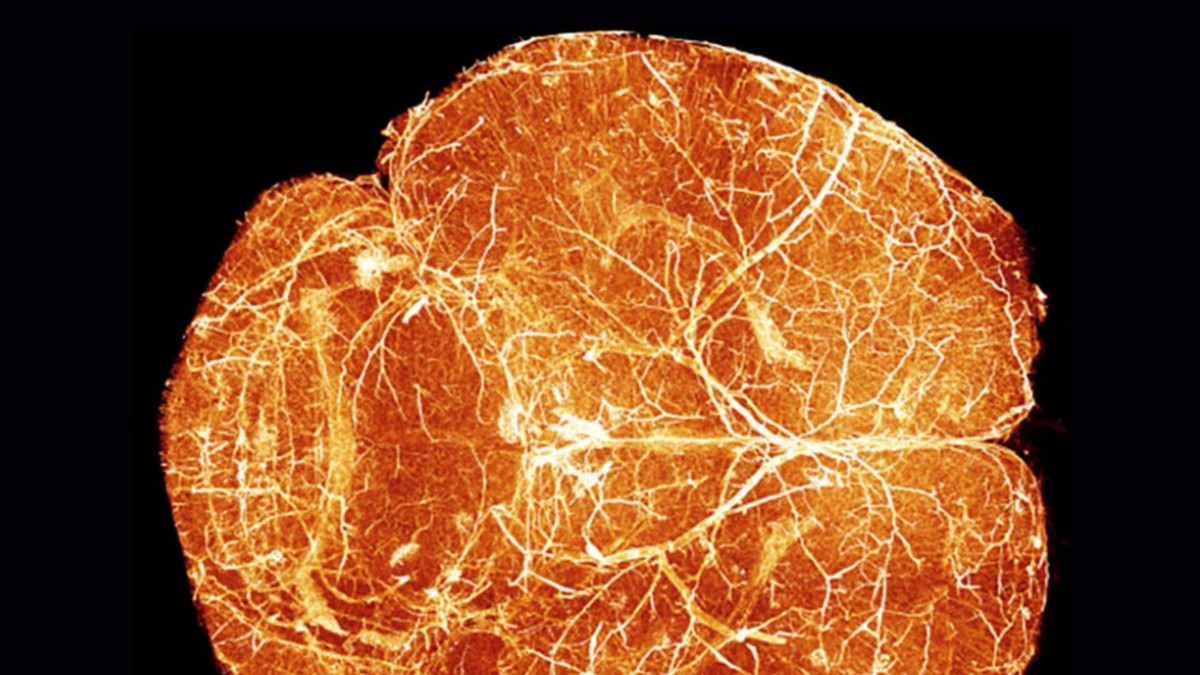
Although it represents only a small fraction of body weight, the brain alone consumes about 20 per cent of the body’s available oxygen and energy. Meeting this high demand depends on a dense and finely organized network of blood vessels responsible for delivering oxygen and nutrients to neurons.
At birth, this vascular network is still immature. Yet it is precisely after birth that the brain undergoes major transformations: it grows rapidly, neural circuits are refined, and certain regions specialize based on sensory experience and environmental input.
Until now, researchers had very limited tools to track—over time and across the entire brain—how blood vessels adapt to these changes.
“We had very detailed maps of the adult brain, but far less information on how the vascular network is established after birth,” said Dubrac. “It was a bit like trying to understand how a city functions without access to its road map.”
To address this gap, Renier’s team developed a three‑dimensional atlas based on a mouse model, enabling them to track—with unprecedented spatiotemporal precision—the development of the vascular network from birth to adulthood.
For their part, Dubrac’s team generated and integrated spatiotemporal transcriptomic data, making it possible to link vascular architecture to dynamic molecular programs.
By combining these complementary areas of expertise, the study reconstructs the brain’s entire vascular network and analyzes its evolution over time, both structurally and molecularly.