 Credit: Illustration by ZME Science.
Credit: Illustration by ZME Science.
Heating accounts for nearly half of our global energy demand. Right now, we meet two-thirds of that demand by burning fossil fuels.
Solar power offers a cleaner path. We install panels and convert sunlight into electricity, which can power radiators. But the sun always sets. When night falls or winter arrives, solar panels stop working. We can stash that solar electricity in batteries, but relying on electrical batteries to meet massive global heating demands is inefficient. Batteries are bulky, expensive, and slowly lose their charge over time. Converting their stored electricity back into heat adds another layer of energy loss.
If we want to decarbonize the globe, we need a better way to trap the summer sun and save it for a dark winter night.
A team of chemists at the University of California, Santa Barbara (UCSB) and UCLA just offered a compelling solution. In their new study, they showed how to bypass traditional batteries entirely using a molecular machine.
By mimicking the genetic damage caused by sunburn, they created a liquid that acts like a rechargeable thermal battery. It swallows sunlight, locks the energy in chemical bonds, and releases it as intense heat months later on demand.
The Promise of Molecular Solar Thermal Storage
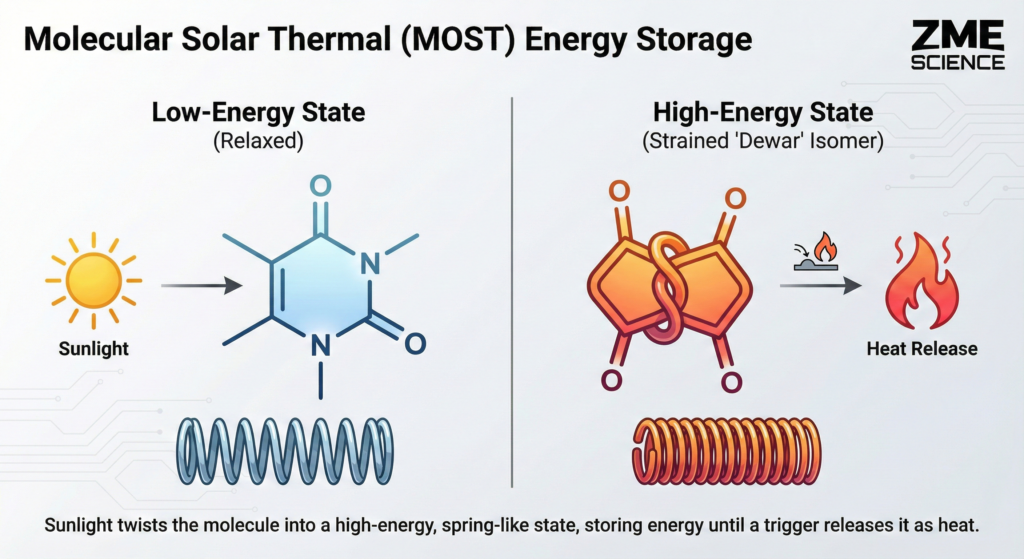
The concept is known as molecular solar thermal (MOST) energy storage. Scientists have experimented with this technology for quite some time. The idea is simple: find a molecule that absorbs light and twists into a strained, high-energy shape.
Think of winding up a tiny mechanical spring. The molecule holds that tension indefinitely. Later, you apply a trigger—like a catalyst or a little heat—and the molecule snaps back to its original shape. That snap releases the stored energy as pure heat.
“With solar panels, you need an additional battery system to store the energy,” said co-author Benjamin Baker, a doctoral student in the Han Lab at the University of California, Santa Barbara. “With molecular solar thermal energy storage, the material itself is able to store that energy from sunlight.”
×
Thank you! One more thing…
Please check your inbox and confirm your subscription.
Historically, MOST systems failed to leave the laboratory. There are many problems with them. They degraded rapidly and typically required toxic solvents. Worst of all, they simply could not hold enough energy to compete with modern batteries. Older MOST candidates topped out at a meager 0.97 megajoules per kilogram (MJ/kg).
You cannot heat a home with that kind of output. To solve this, UCSB Associate Professor Grace Han and her team looked away from traditional engineering. They looked at human biology.
A Battery Built from Sunburn
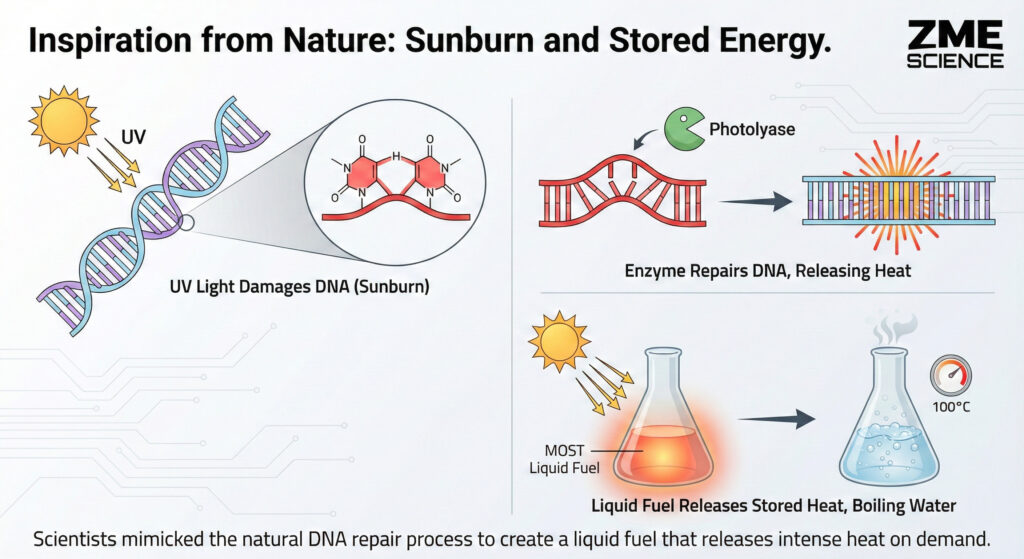
When you spend too much time on the beach without sunscreen, high-energy ultraviolet (UV) light attacks your DNA. It causes adjacent thymine bases in your genetic code to fuse together.
This fusion creates a structure called a (6-4) lesion. If the UV assault continues, this lesion twists into an even more bizarre, strained shape known as a “Dewar” isomer.
In biology, Dewar isomers are terrible news. They create physical kinks in your DNA’s double-helix spiral. This disrupts genetic copying and can lead to skin cancer.
However, nature evolved a defense mechanism. An enzyme called photolyase hunts down these lesions and forcefully snaps them back into their safe, relaxed forms. This biological snap-back releases a significant burst of heat.
Han’s team realized this dangerous genetic kink was actually a perfect molecular battery. They synthesized a chemical cousin to DNA’s thymine, called a 2-pyrimidone.
“The concept is reusable and recyclable,” said Han Nguyen, the paper’s lead author.
They engineered this synthetic pyrimidone to reliably fold into a high-energy Dewar isomer under sunlight. When triggered, it unfolds on command.
Compounded Strain and Boiling Water
The UCSB team packed enormous power into their new molecule. They achieved this through a process called compounded strain.
When the synthetic pyrimidone absorbs sunlight, it twists into a fused structure containing two distinct, four-membered rings.
Four-membered chemical rings exist under immense structural tension. Fusing two of them together creates a molecule absolutely desperate to snap back to a relaxed state.
To maximize this power, the researchers trimmed the fat. “We prioritized a lightweight, compact molecule design,” Nguyen said. “For this project, we cut everything we didn’t need. Anything that was unnecessary, we removed to make the molecule as compact as possible.”
The result blew previous records out of the water. The new pyrimidone-based system boasts an energy density of 1.65 MJ/kg. That is nearly double the capacity of a standard lithium-ion battery.
In their experiments, the researchers used the released heat to boil water under ambient conditions.
“Boiling water is an energy-intensive process,” Nguyen said. “The fact that we can boil water under ambient conditions is a big achievement.”
Ditching Toxic Solvents for Liquid Fuel
But achieving high energy density on paper means nothing if the fuel requires massive dilution. Previous MOST systems relied on solid molecules. Engineers had to dissolve these solids in toxic chemical solvents to make them flow through pipes.
Solvents destroy efficiency. If you dilute your solar fuel to a 10% concentration, you lose 90% of your energy density. You also introduce toxic hazards into the system.
Nguyen and her team solved this by designing a version of their molecule that remains liquid at room temperature. It requires zero solvent. Furthermore, the molecule gets along perfectly well with water. If a pipe leaks in a future residential heating system, it will not spew toxic chemicals into your basement.
“Think of photochromic sunglasses. When you’re inside, they’re just clear lenses. You walk out into the sun, and they darken on their own. Come back inside, and the lenses become clear again,” Nguyen continued in the UCSB press release.
She expanded on the ultimate goal of the project. “That kind of reversible change is what we’re interested in. Only instead of changing color, we want to use the same idea to store energy, release it when we need it, and then reuse the material over and over,” Nguyen said in the press release.
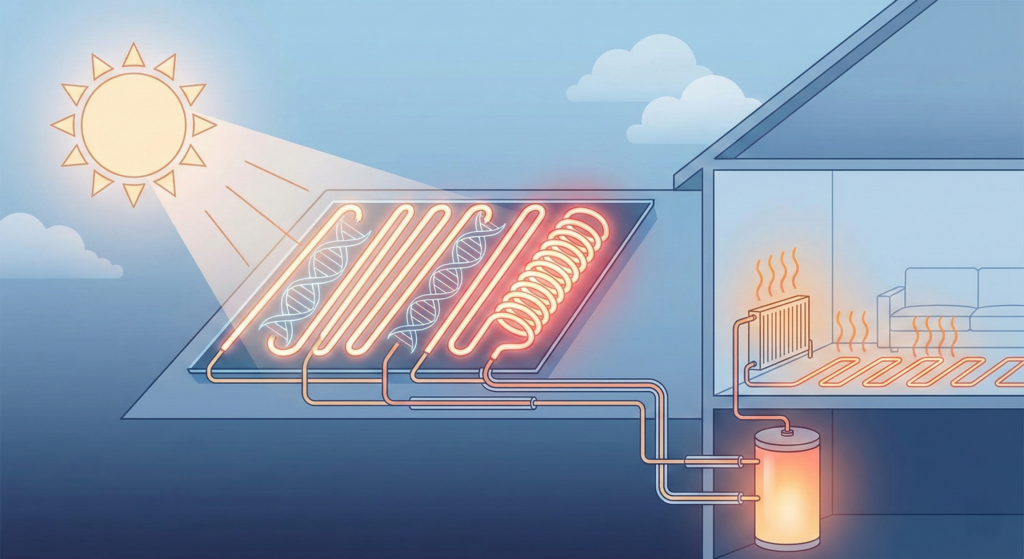
The team envisions a closed-loop system. A home solar collector circulates the liquid fuel. The sun charges it. An insulated tank stores it. When the thermostat drops, the liquid flows through a reactor. An acid catalyst triggers the heat release. The spent liquid then loops back to the roof.
The Catch: Leaks and UV Limits
Before we start pumping liquid sunburn through our roofs, we have to address the system’s current flaws. The research team is highly transparent about the hurdles ahead.
First, the sun produces a vast spectrum of light. A perfect solar collector would harvest all of it. Unfortunately, the pyrimidone molecules only absorb light in a narrow UV-A and UV-B range.
This narrow band represents a mere five percent of the total solar spectrum. The vast majority of the sun’s energy—visible and infrared light—passes right through the liquid without charging it at all.
Second, the system suffers from a low “quantum yield.” This metric asks a simple question: for every 100 photons of light that hit the fluid, how many successfully force the molecule to twist into its storage state?
Currently, the answer is in the single digits. The researchers suspect the molecule has a “fast leak.” Instead of twisting into the high-energy Dewar isomer, the excited molecule frequently shakes off the energy as useless ambient heat. Because of this low efficiency, the fluid requires prolonged exposure to sunlight to reach a full charge.
Bottling the Summer Sun for Winter
Finally, the current prototype relies on an acid catalyst mixed directly into the fuel to trigger the heat release. In a real-world system, engineers would need to add a neutralization step to remove the acid after the heat drops.
The researchers propose a clever workaround in their paper. Future designs might flow the fuel over a solid surface coated in acid. This would trigger the reaction without contaminating the liquid itself.
Despite these engineering challenges, this system looks stable. The biggest fear with chemical storage is thermal reversion—your fuel spontaneously discharging all its heat because the storage tank got a little warm in August. But this pyrimidone molecule is incredibly robust. The researchers calculated a half-life of up to 481 days at room temperature. You could charge this fluid on a sweltering July afternoon, leave it in a tank, and use it to boil water in the dead of January.
“We typically describe it as a rechargeable solar battery,” Nguyen said. “It stores sunlight, and it can be recharged.”
We are not quite ready to heat our homes with MOST systems tomorrow. Scientists must tweak the molecules to absorb a much wider spectrum of light. But by looking at how our own biology reacts to the sun, these chemists have proven that capturing lightning—or rather, sunlight—in a bottle is no longer just a figure of speech.
The new findings appeared in the journal Science.