There’s a new entry in the growing body of evidence that main-group elements like phosphorous, which are often relegated to supporting ligand roles in transition-metal-catalyzed reactions, can step up as the main catalytic event.
A team led by Abigail Doyle at the University of California, Los Angeles, found that phosphines can show transition- metal-like reactivity in a light-driven reaction between amine-containing rings and carbon-carbon double bonds (Nature 2026, DOI: 10.1038/s41586-026-10263-7).
“This project adds to the growing body of literature that showcases the unique reactivity of main group compounds as catalysts and the resulting opportunities for organic synthesis,” Doyle says in an email to C&EN.
“It’s kind of just rethinking what these compounds can do and their role in chemistry,” says Flora Fan, a fourth-year graduate student in Doyle’s lab who did much of the hands-on work.
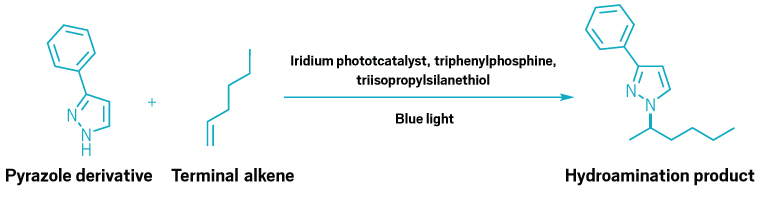
A reaction scheme showing a pyrazole derivative adding to a terminal alkene.
Phosphine catalysts can help chemists access reactivity that transition metals may struggle with, such as hydroamination with nitrogen-rich rings and terminal alkenes.
The new paper builds on a previous study the group published describing a way to use a phosphine and an iridium photocatalyst to facilitate an addition reaction between alkenes and azoles (J. Am. Chem. Soc. 2024, DOI: 10.1021/jacs.4c05881). Kassandra Sedillo, who worked on the project as a graduate student, noticed that when she used a triphenylphosphine derivative instead of tricyclohexylphosphine, the amine attached to the more substituted alkene carbon rather than the less substituted one. That selectivity switch indicated that the mechanism had changed from the expected nitrogen-radical route to something else.
“We couldn’t explain through our proposed mechanism how we were obtaining that product,” Fan says, so she started investigating how it might have formed.
From competition experiments and density functional theory computations, Fan figured out that the phosphine radical generated by the photocatalyst was coordinating to the alkene—much like a metal would—to create a spatially separated radical cation intermediate. This intermediate then adds to the amine through one of two potential pathways; Fan says both could be occurring at once. A thiol furnishes the molecule with a nitrogen to give the final product.
“It’s a very atom-economical reaction. You’re not generating any by-products. Everything is catalytic. So all the atoms are used,” Fan says. She and her lab mates found that the reaction can handle a variety of aromatic and nonaromatic heterocycles, and it even works if other potentially reactive functional groups, such as alcohols, are present.
Sami Lakhdar, who researches photocatalysis at the University of Toulouse and was not involved in the work, calls it “really spectacular” and “a breakthrough in the field of main-group catalysis.” The overall transformation is something that isn’t accessible with transition metals, he adds, so it’s practical as well as conceptually interesting.
John Slattery of the University of York, another chemist who was not involved in the work, says the thoroughness with which the researchers investigated unexpected results to discover something new “just sums up great science and great scientists. It’s a really nice sort of story, and it shows that serendipity is alive and well in science.”
Fan says she and her lab mates will continue investigating the mechanism and try to expand it. For example, they would like to see if they can use chiral phosphines to make the reaction stereoselective or perhaps intercept the carbon radical intermediate with other functional groups.
Seeing the project go from “Wow, this is a curious finding” to publishing in Nature was a surprising and fulfilling journey, Fan says. “You never really know where your PhD takes you.”
Chemical & Engineering News
ISSN 0009-2347
Copyright ©
2026 American Chemical Society