Track your investments for FREE with Simply Wall St, the portfolio command center trusted by over 7 million individual investors worldwide.
Johnson & Johnson (NYSE:JNJ) has received U.S. FDA approval for ICOTYDE, an oral IL-23 receptor inhibitor for moderate-to-severe plaque psoriasis.
ICOTYDE is the first targeted oral IL-23 inhibitor and offers an alternative to injectable biologics for eligible adult and adolescent patients.
The therapy is supported by Phase 3 efficacy and safety data and is part of a broader clinical program in psoriatic arthritis and inflammatory bowel diseases.
For investors watching NYSE:JNJ, ICOTYDE adds a new asset to the company’s immunology portfolio at a time when autoimmune disease treatments are a focus for large healthcare groups. Johnson & Johnson is aiming to address patients who cycle through topical therapies and may prefer an oral option instead of injections.
The broader clinical program in psoriatic arthritis and inflammatory bowel diseases gives ICOTYDE potential relevance beyond plaque psoriasis, depending on future trial outcomes and regulatory decisions. Readers tracking Johnson & Johnson may want to follow how quickly the product is adopted in psoriasis and how management updates the market on progress in these additional indications.
Stay updated on the most important news stories for Johnson & Johnson by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on Johnson & Johnson.
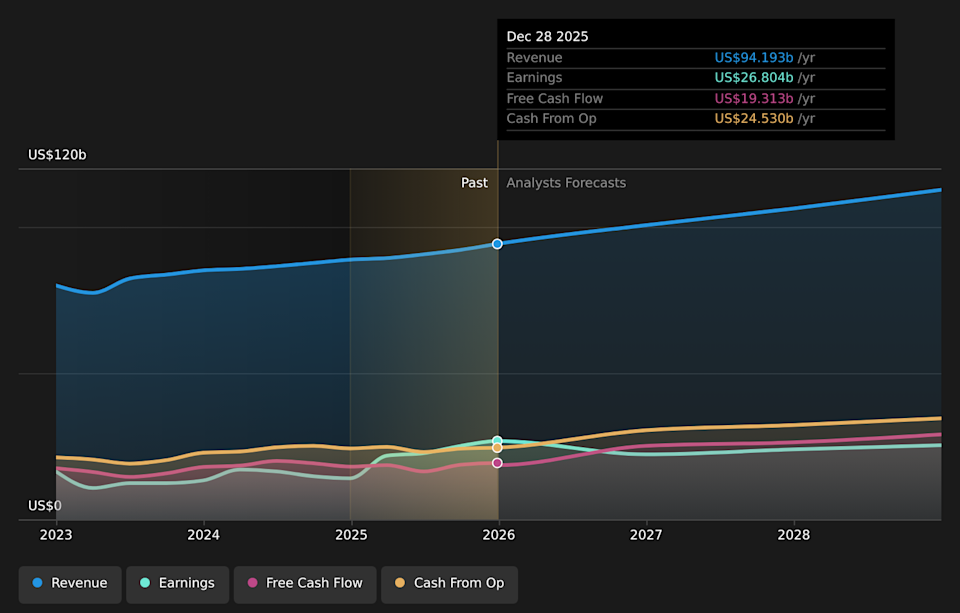 NYSE:JNJ Earnings & Revenue Growth as at Mar 2026
NYSE:JNJ Earnings & Revenue Growth as at Mar 2026
We’ve flagged 2 risks for Johnson & Johnson. See which could impact your investment.
⚖️ Price vs Analyst Target: At US$238.11, JNJ trades about 1% below the US$240.38 analyst target, which sits comfortably within the typical range of opinions.
✅ Simply Wall St Valuation: Simply Wall St estimates the shares are trading about 37.8% below fair value, flagging the stock as undervalued on that model.
❌ Recent Momentum: The 30 day return of roughly 2.2% decline shows recent sentiment has been soft despite the ICOTYDE approval.
There is only one way to know the right time to buy, sell or hold Johnson & Johnson. Head to Simply Wall St’s company report for the latest analysis of Johnson & Johnson’s Fair Value.
📊 ICOTYDE gives JNJ a differentiated oral option in plaque psoriasis, adding another branded product to its immunology portfolio.
📊 Watch uptake trends in psoriasis, progress of trials in psoriatic arthritis and inflammatory bowel disease, and any revenue commentary in upcoming results calls.
⚠️ Key risks include execution on launch, competition from existing biologics and orals, and the two flagged business risks already identified for the company.
For the full picture including more risks and rewards, check out the complete Johnson & Johnson analysis. Alternatively, you can check out the community page for Johnson & Johnson to see how other investors believe this latest news will impact the company’s narrative.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.
Companies discussed in this article include JNJ.
Have feedback on this article? Concerned about the content? Get in touch with us directly. Alternatively, email editorial-team@simplywallst.com

