Quitting cocaine does not always silence the urge to use it again. Even after long periods of abstinence, memories tied to the drug can suddenly reignite powerful cravings.
New research shows that repeated cocaine exposure may rewire a memory-linked brain pathway, quietly locking drug cues and reward together.
In mice, scientists discovered that the drug dampens activity in a circuit that normally helps regulate how memories influence motivation.
The findings reveal how long-lasting molecular changes inside this pathway can leave the brain more vulnerable to relapse long after cocaine use stops.
Cocaine reshapes brain signaling
Inside a memory circuit that links emotional context to reward, activity fell sharply after repeated cocaine exposure.
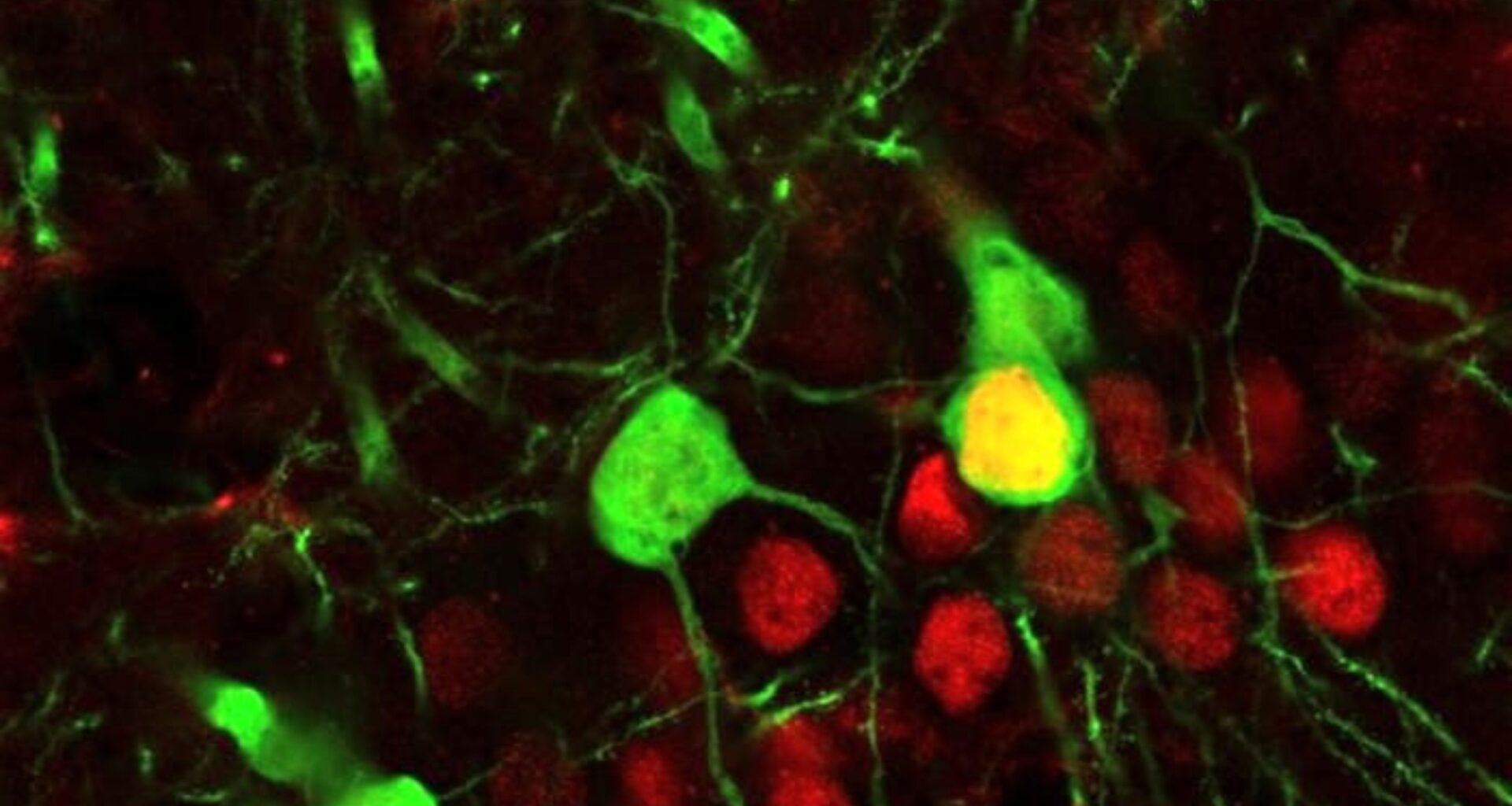
Tracing those changes in mice, Dr. Andrew Eagle and colleagues at Michigan State University (MSU), demonstrated that cocaine rewires this pathway in ways that promote later drug seeking.
As cocaine exposure continued, a molecular signal accumulated inside the same neurons and steadily altered how strongly the circuit responded to the drug.
Understanding how this circuit becomes locked into that altered state is essential for explaining why the urge to seek cocaine can return long after the drug disappears.
Brain links memory and reward
Deep in the brain, the new paper tracked a circuit that links memory cues to drug reward. Signals began in the ventral hippocampus, the emotion-linked part of the memory system, and then moved toward a reward hub.
From there, neurons sent excitatory messages toward reward circuits, and repeated cocaine made that signal harder to trigger.
Because this brain region stores memories of places and experiences, its signals to the brain’s reward center can trigger powerful urges to use cocaine again.
Gene switch reshapes brain circuit
After repeated cocaine exposure, researchers found that a control protein built up inside the same brain cells in this memory-to-reward pathway.
As the protein accumulated, it began turning other genes on and off, gradually changing how those cells responded to cocaine.
“This protein isn’t just associated with these changes, it is necessary for them,” said Dr. Eagle. When scientists removed the protein from that circuit, cocaine no longer pushed the pathway into the same relapse-prone state.
One gene that became far more active produced a protein called calreticulin, which helps regulate calcium inside cells. Higher levels of calreticulin altered the internal calcium balance that controls how nerve cells send signals.
With more calreticulin present, the neurons became harder to activate, and fewer signals traveled from memory circuits into the brain’s reward system.
Because that shift lasted for weeks in mice, the findings indicate that the brain changes driving relapse can persist long after cocaine use stops.
Cocaine dampens neuron signals
Instead of firing easily, the pathway’s neurons showed lower excitability – meaning they were less able to fire electrical signals – after repeated cocaine exposure.
Slices of brain tissue taken one day after cocaine exposure showed fewer spikes, and removing DeltaFosB prevented that slowdown.
Similar effects appeared when mice self-administered cocaine, which linked the cellular change to behavior that resembles addiction.
A weaker signal from the hippocampus may leave the reward system easier to steer by cues, even when a person wants to stop.
Brain stimulation reduces preference
To test whether this pathway truly drives drug-seeking behavior, the researchers tried boosting its activity.
They stimulated the circuit for several days while mice were still exposed to cues linked to cocaine. As the neurons became more active again, the animals gradually stopped preferring the chamber associated with the drug.
Short bursts of stimulation did not change the mice’s behavior, suggesting the circuit needs sustained activity to recover after repeated cocaine exposure.
Importantly, reducing the pathway’s activity did not increase reward responses at the dose tested. That finding suggests any future therapy targeting this circuit would require careful timing and dosing.
Cocaine relapse remains common
The findings also help explain why cocaine relapse remains a major challenge. Across the United States, an estimated 1.3 million people were living with cocaine use disorder in 2023.
Follow-up studies show how difficult recovery can be. Within a year, about 24 percent of people returned to weekly cocaine use, while another 18 percent entered treatment again.
Cocaine works by boosting dopamine in the brain’s reward system. It blocks the normal reuptake of this chemical signal, strengthening the memories and cues linked to drug use.
“Addiction is a disease in the same sense as cancer,” said study co-author Dr. A.J. Robison, a professor of neuroscience and physiology at MSU.
Scientists pursue new therapies
Today no U.S. Food and Drug Administration-approved medicine treats cocaine addiction, and quitting rarely brings the intense physical withdrawal seen with opioids.
Instead of aiming at withdrawal symptoms, MSU scientists want compounds that stop DeltaFosB from binding DNA in those neurons.
“If we could find the right kind of compound that works in the right way, that could potentially be a treatment for cocaine addiction,” said Dr. Robison.
Robison noted that turning that idea into a real treatment will likely take many years, but it remains the long-term goal.
Hormones may alter brain signals
Only male mice went through these experiments, so the results leave open how cocaine changes the same circuit in females.
Hormones can tune how strongly neurons respond to stress and reward, which may alter how DeltaFosB accumulates over time.
At MSU, the lab plans to test sex hormones in this pathway and watch for different patterns after cocaine exposure.
Human brains share many genes with mice, but turning a circuit discovery into a medicine will require careful studies in people.
By tracing relapse-like seeking to a gene-controlled drop in a memory-to-reward circuit, the study also provides a clearer map for future therapy.
Scientists suggest that compounds designed to block DeltaFosB in the right cells could one day reduce cravings without dulling normal motivation, although translating these findings into treatments will take time.
The study is published in the journal Science Advances.
Image Credit: Michigan State University Robison Lab
—–
Like what you read? Subscribe to our newsletter for engaging articles, exclusive content, and the latest updates.
Check us out on EarthSnap, a free app brought to you by Eric Ralls and Earth.com.
—–