A new ultrasound-based technique developed by researchers at Duke University uses microbubbles to deliver cancer drugs directly into tumour cells, showing promise for highly targeted treatments that cause minimal damage to healthy tissue.

Engineers at Duke University have developed a promising new method that uses microbubbles and ultrasound to deliver large cancer drugs directly into tumour cells, triggering them to self-destruct while sparing healthy tissue.
The technique, known as Sonoporation-assisted Precise Intracellular Nanodelivery (SonoPIN), achieved powerful results in early laboratory experiments. Researchers reported that 50 percent of targeted cancer cells were destroyed, while 99 percent of non-targeted cells remained healthy.
The findings highlight the potential for a more precise approach to cancer treatment with minimal side effects.
Unlocking the potential of PROTACs
The research focuses on a new class of therapeutics called proteolysis-targeting chimeras (PROTACs) which are designed to break down harmful proteins inside cells. These molecules work by binding to a target protein and recruiting an enzyme that marks it for destruction by the cell’s natural waste disposal system.
In cancer therapy, PROTACs have been used to degrade a protein called BRD4, which plays a key role in tumour growth.
In cancer therapy, PROTACs have been used to degrade a protein called BRD4, which plays a key role in tumour growth. Removing this protein disrupts the cancer cells’ ability to multiply and survive, forcing them to self-destruct.
Despite their promise, PROTACs face two major challenges: they are too large to easily enter cells and they can also affect healthy cells if delivered indiscriminately.
“PROTAC molecules are too big to get into cells in the first place,” said Yuqi Wu, a doctoral student working in the laboratory of Tony Jun Huang, the William Bevan Distinguished Professor of Mechanical Engineering and Materials Science at Duke. “But with our SonoPIN platform, the PROTACs can enter into targeted cancer cells while almost completely ignoring non-targeted cells.”
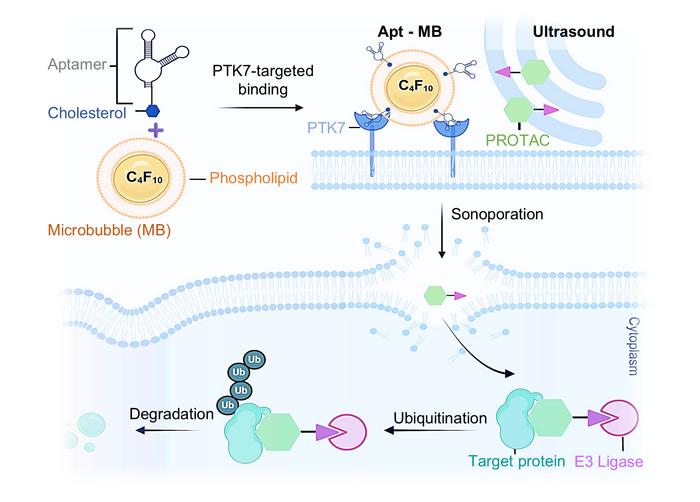
A graphical chart demonstrating how the SonoPIN platform works. Microbubbles targeted to cancer cells are burst by ultrasound waves, creating small pores in nearby cell membranes large enough for pharmaceuticals like PROTACs to enter. Credit: Duke University.
How SonoPIN works
SonoPIN uses microscopic bubbles that are usually employed in medical imaging to enhance ultrasound scans. When exposed to controlled ultrasound waves, these bubbles collapse in a way that creates tiny temporary openings in nearby cell membranes.
SonoPIN uses microscopic bubbles that are usually employed in medical imaging to enhance ultrasound scans.
“This process is less like an explosion and more like a temporary, controlled mechanical opening,” said Huang. “While it involves physical force, because cell membranes are fluid and dynamic, they naturally self-heal and close these pores within minutes if not seconds.”
These fleeting openings allow large molecules such as PROTACs to pass inside the cell. To ensure precision, the researchers coated the microbubbles with synthetic nucleic acid strands that bind specifically to cancer cell receptors.
Promising early results
In testing, the team fine-tuned ultrasound settings to maximise delivery efficiency. By attaching fluorescent markers to the PROTACs they were able to track how effectively the drugs entered cells.
After just one minute of ultrasound exposure, treated cancer cells showed fluorescence levels seven times higher than those receiving conventional delivery methods. This confirmed that significantly more PROTAC molecules had entered the cells, leading to the destruction of half the cancer cells while leaving most healthy cells unharmed.
Next steps for cancer treatment
The researchers now plan to move into animal studies and have already filed a patent for the technology. They believe that injecting both PROTACs and targeted microbubbles into the bloodstream, combined with focused ultrasound, could offer a powerful and precise cancer therapy.
“Because SonoPIN relies on a mechanical delivery approach rather than biological engulfment, it could theoretically deliver therapeutics of almost any size,” said Huang. “We would also be excited to see how it performs with therapeutics such as large gene-editing complexes.”
Related topics
Cancer research, Cell Cultures, Chemotherapy, Drug Delivery, Drug Discovery Processes, Immuno-oncology therapeutics, In Vitro, Nanomedicine, Nanoparticles, Oncology, Precision Medicine

