New findings and functional significance of the argonaut eggcase microstructure
The argonaut eggcase exhibits a three-layered structure, with a middle organic layer sandwiched between outer and inner spherulitic-fibrous prismatic layers14,15,21,22 (Figs. 3, 7, S3). The presence of two sheets separated by a gap at the keel tubercles (Figs. 3B, F, S3C1, C2, C) suggests the middle organic layer is a single structural unit composed of two organic sheets, serving as a scaffold for crystal nucleation. Meanwhile, the continuous prismatic layers at the spiral center (Figs. 3B–E, S3B) suggest that the inner and outer prismatic layers are not only identical in composition and microstructure, but also follow a similar growth pattern in which crystal layers can grow omnidirectionally when an organic layer is present (Fig. 3). These local structural variations indicate that the eggcase adjusts its mechanical strength by altering the shape of the middle organic layer.
Fig. 7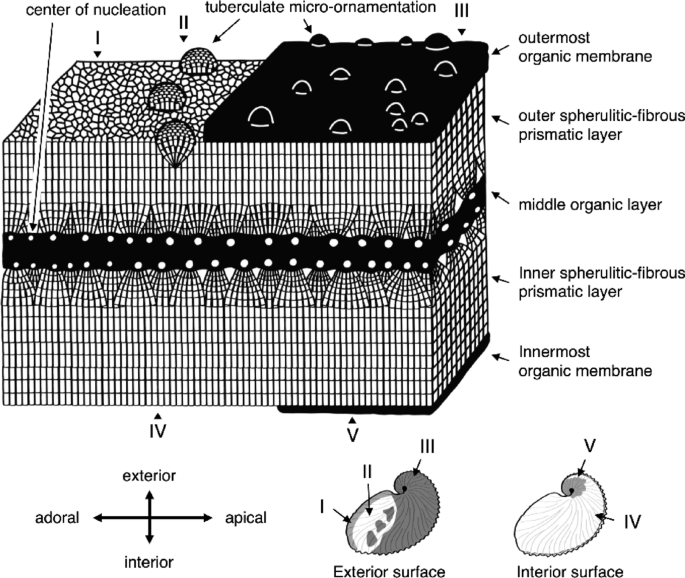 The alternative text for this image may have been generated using AI.
The alternative text for this image may have been generated using AI.
Schematic diagram of the argonauts’ eggcase microstructure. The distribution of the organic membrane and the tuberculate micro-ornamentation is shown at the bottom right. The exterior surface is classified into three types: (I) without the organic membrane and micro-ornamentation, (II) with micro-ornamentation only, and (III) with both the organic membrane and micro-ornamentation. The interior surface is categorized into two types: (IV) without the organic membrane and (V) with the organic membrane.
Surface observations showed structural contrasts between the inner and outer layers, including differences in organic membrane distribution and tuberculate ornamentation (Figs. 4, 7, S4). The outermost membrane, composed of stacked organic sheets, was thicker and covered nearly the entire exterior except at the adoral margin, while the thinner innermost membrane was confined to the central interior. We suggest that this innermost organic membrane acted as the termination point for crystal growth, because it resembles the boundary region (the margin) where crystal growth ends, which is visible as organic membranes within the crystals (Figs. 4A1, 4C2, S4A2, C2, S7). Structural differences among the organic membranes likely reflect adaptations to isolate the mineralized layer from the surrounding environment, perhaps related to their functions29,30. For example, the localized distribution of the innermost organic membrane and the function of the surrounding region in egg attachment to the surface of the eggcase31 suggest that this membrane may serve as an adhesive surface for eggs.
Our observation indicates that the outermost organic membrane covers the entire eggcase and is specifically thick in the posterior areas inaccessible to the first dorsal arm. The areas where the covering organic membrane is damaged and the underlying crystal layers are exposed display lath-like crystals (Figs. 4F2, S4E2), which is interpreted as indicative of partial crystal dissolution23,28. This suggests that the organic membrane may function to prevent calcium carbonate crystals from dissolving into seawater, as commonly observed in other biominerals32,33,34. In fresh, hydrated samples, the eggcase dissipates mechanical energy through its viscoelastic response that varies with relative humidity and load application rate35. In aged, dried, and deteriorated specimens, the outermost organic membrane peels off, causing the eggcase to become brittle. The thick outermost organic membrane likely maintains the eggcase’s flexibility, mechanical strength, and provides protection from dissolution and physical impacts. Known functions of surface-coating organic membranes, such as degradation prevention, adhesion, and mechanical properties enhancement, are also observed in analogous calcareous structures of other organisms, such as bivalves, coccoliths and eggshells34,35,36,37,38,39,40.
In addition to the innermost and outermost organic membranes, the eggcase also differs structurally between the inner and outer surfaces. Hemispherical structures 30–150 µm in diameter, known as tuberculate micro-ornamentation, were observed only on the exterior surface of the eggcase and were absent on the interior surface of the eggcase. We suggest that these tubercles may play a role in protection against the impacts from waves and rocks. Therefore, the outer layer of the eggcase forms such a complex architecture to enhance structural strength to prevent damage.
Comparative microstructural analysis of the argonaut eggcase and other conchiferan shells
The general appearance of the argonaut eggcase superficially resembles that of typical molluscan shells, appearing nearly indistinguishable at first glance. Compared with other cephalopod external shells, the argonaut eggcase lacks key architectural features such as septa and a siphuncle. Differences in shell architecture therefore represent one aspect supporting the interpretation of the argonaut eggcase as a non-homologous shell-like structure. Nevertheless, caution is required when using the absence of septa and a siphuncle as criteria for non-homology, because the gladius of squids and the stylets of octopuses also lack these features but are generally regarded as homologous derivatives of the cephalopod shell41,42.
The microstructural characteristics reveal clear differences in their biomineralization processes. Molluscan shells are secreted by mantle tissue, with mineral deposition proceeding inward from an organic sheet called the periostracum43,44. In contrast, the argonaut eggcase is formed probably by secretions from the first dorsal arm pairs (sensu Villepreux-Power), with crystals growing bidirectionally from a middle organic layer. This growth pattern differs markedly from that of typical molluscan shells, in which a calcified layer grows unidirectionally inward from the periostracum22.
Among modern cephalopods, the argonaut eggcase may be interpreted as a non-homologous shell-like structure, differing in shell architecture and crystal polymorph rather than microstructure, since modern cephalopod shells also grow bidirectionally from an organic layer45,46,47,48. Unlike primitive cephalopods (e.g., nautilus and ammonite shells), modern cephalopods have modified their shells into internal or non-calcified structures (e.g., cuttlebone, internal shell of Spirula). Despite variation in shell macro- and microstructure, all modern cephalopod shells contain aragonite as their carbonate crystal polymorph49,50,51. In contrast, the argonaut eggcase consists entirely of calcite, as previously reported15,22,23. The selection of crystal polymorphs in biominerals is thought to reflect paleoenvironmental ocean compositions (Mg/Ca ratio) when the biomineral structures were acquired52. The origin of cephalopod shells can be traced back to a single origin acquired prior to the Ordovician period, when the Mg/Ca ratio indicated an aragonite sea. Meanwhile, argonauts appeared during the Early Tertiary11,53, when the Mg/Ca ratio indicated a calcite sea and ocean chemical compositions predominantly favored calcite54. These contrasting crystal polymorphs suggest that the biomineralized structures in cephalopods and argonauts arose independently within the cephalopod lineage.
Although fundamentally different in origin and organization, the argonaut eggcase shares several microstructural features with other cephalopod shells, including microstructural architecture, bidirectional growth, and tuberculate micro-ornamentation. The outer calcified layers of the internalized shells in cuttlefish and Spirula display a microstructural architecture characterized by the presence of spherulitic prismatic layers47,48,55,56, superficially similar to that of the argonaut eggcase. In these species, the spherulitic prismatic layer grows outward from an organic layer (probably periostracum41,57) forming a microstructure with a middle organic layer sandwiched between two calcified layers. This organization is also seen in embryonic/juvenile shells of Mesozoic ammonites47,48,56,58. However, despite the resemblance between spherulitic prismatic and layered structures, the microstructure of other cephalopod shells is fundamentally different (i.e., not homologous) from that of the argonaut eggcase. This is because the inner and outer calcified layers in their shells are not continuous and therefore structurally distinct, whereas the argonaut eggcase exhibits a consistent microstructure across both layers. Thus, while the overall architecture and bidirectional growth appear similar, the underlying formation mechanisms differ. These similar microstructural traits may reflect developmental or functional constraints and likely represent a case of convergent evolution within the cephalopod lineage.
Tuberculate micro-ornamentation restricted to the exterior surface has been reported in Mesozoic ammonites and some gastropods and bivalves59,60,61,62,63. The formation process of tubercles in these groups remains poorly understood. Hickman (2004) proposed that such structures may result from mineralization under weak biological control, based on observations of tubercles in gastropod larval shell. In contrast to these uncertain primordia, our observations indicate that tubercles in the argonaut eggcase develop progressively and are fully developed in the marginal region under tightly regulated biological control (Figs. 4D–F, 7, S4D–F, S12, S13). Landman (1994) suggested that the ornamental tubercles in embryonic or juvenile ammonites may aid hatching by facilitating egg capsule rupture. In argonauts, we propose that tubercles help protect the eggcase from mechanical impact caused by waves or contact with rocks, probably similar to gastropods61. This interpretation is supported by their widespread distribution on the outer surface of adult eggcase specimens. Although mechanical testing has yet to be performed to confirm their role, the ornamental tubercles may also enhance shell strength before and after hatching.
Unique mode of eggcase repair in argonauts
Mollusks repair damaged shells by secreting new material from mantle tissue at the injury site. When damage occurs beyond the shell edge away from the mantle, the repair mechanism differs from normal shell formation, as evidenced by distinct microstructural and mineralogical features65,66,67,68. In the argonaut eggcase, the surface of the repaired region, which was damaged by perforation in the posterior section of the keel, appears optically opaque and smooth, clearly differing from the non-repaired regions (Figs. 5A–C, 5G1, 5G2, 8A). Our microstructural observations showed that the repaired area comprises two new layers, the outer organic and the inner calcified layers, lacking the outer calcified layer observed in the intact three-layered structure (Figs. 5F–H and 8B). Moreover, the outer organic layer does not extend from the middle organic layer but instead is located beneath the inner calcified layer of the intact area (Figs. 5F and 8B). At the adhesion site of repaired and non-repaired regions, we observed a gradual thinning of the organic layer with increasing distance from the perforation (Figs. 5F2, 5F3, 5F7, 5F8, 8B). Crystal orientation became increasingly consistent with that observed in the original structure (Figs. 5F7–13, 8B). These proximodistal differences in regional growth patterns likely produce mechanical strength at the adhesion site, thus enhancing the repaired structure’s resistance to subsequent damage. Therefore, in contrast to that of a typical molluscan shell, the repair process of the eggcase was apparently identical to eggcase formation, as confirmed by the presence of identical microarchitecture and similar continuous crystal growth patterns.
Fig. 8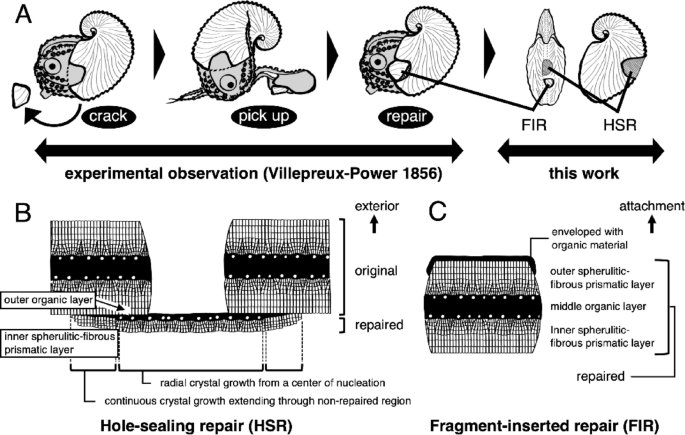 The alternative text for this image may have been generated using AI.
The alternative text for this image may have been generated using AI.
Diagram of the microstructure of repaired argonaut eggcase. (A) Schematic illustration of the repair traits of the eggcase. Two types of repair traits are confirmed: hole-sealing repair (HSR) and fragment-inserted repair (FIR). (B, C) Diagram of the microstructure of (B) the HSR and (C) the FIR. Abbreviations: fragment-inserted repair (FIR), hole-sealing repair (HSR).
Calcified granule deposition during the early stages of exoskeleton repair is a noteworthy characteristic of exoskeleton repair in mollusks69,70, and was also observed in the repaired regions of the argonaut eggcase. These granules were abundant on and between the initially formed organic membranes in the repaired eggcase, resembling previously reported deposits (Figs. 5D, E, G and 6). Vertical sections revealed radiating growth patterns originating from these calcified granules, which resemble the center of nucleation in original eggcase formation (Figs. 5F9, F13). This suggests that the calcified granules in the repaired eggcase can serve as center of nucleation (Figs. 5E, F13, S10). Mount et al. (2004) reported that granules involved in calcification are associated with hemocytes. However, chemical compositions of the calcified particles in the repaired region and the original region in the eggcases were identical, in contrast to previous reports of compositional differences, particularly regarding phosphorus70. These findings suggest that eggcase regeneration does not entirely follow distinct repair pathways but employs a mechanism similar to normal formation.
Our findings allow us to reexamine Villepreux-Power’s widely accepted conclusion that argonauts form their eggcases using the first dorsal arms, based on her repair experiments (Fig. 8A). In her study, she partially damaged eggcases and observed the animals reattaching fragments using their first dorsal arms. Her observation likely corresponds to what we describe as fragment-inserted repair (FIR; Figs. 5, 8B). However, since not all perforating damage can be repaired through FIR, the argonauts apparently possess another mechanism, the hole-sealing repair (HSR), which plugs the perforated area with newly secreted eggcase-forming materials lacking the layered structure of original eggcases (Figs. 5, 8C). Overall, our observations raise doubts about the direct involvement of the first dorsal arms in eggcase formation, as suggested by Villepreux-Power, whose conclusions were based solely on behavioral observation and are thus inconclusive. Our findings imply that the first dorsal arms can secrete organic material, as indicated by the presence of the outermost organic membrane, and by the organic material enveloping fragment-inserted repair (Fig. 7). Microstructural characteristics support this skepticism, as layers mature within a few tens of millimeters from the aperture, where growth primarily occurs (Fig. 7). This growth mode contrasts with the fact that the expansive, web-like first dorsal arms of argonauts can cover the entire outer surface of the eggcase. Based on our observations, we propose that the first dorsal arms primarily serve a supportive role in eggcase formation. Future studies involving detailed histological observations, experimental re-examinations, and spatial gene expression analyses are essential to test this hypothesis.
A sequential model of eggcase formation and biomineralization
The formation of mineralized skeletal structures is a sequential process, with each phase producing distinct morphological features. We identified four stages of prismatic structure formation in the argonaut eggcase (Fig. 7, S12). Stage one begins with crystallization during the formation of organic microfibers, referred to as the nucleation phase. We also observed an organic–inorganic particle within the organic fibers, likely serving as the nucleation site (Figs. 5E, S12, S13). Nucleation occurs both on the organic surface and within the organic microfibers (Figs. 5E, S12). In stage two, we observed a radial growth originating from the center of nucleation (Fig. 2C, 3G, 5F13, 5H3–4, 5H2–4, 7, 8, S3A, B, SS12), followed by growth perpendicular to the eggcase surface, which we designate as stage three (Figs. 2B, 2D, 3C, 3F, 3D, 3H, 5F13, 7, 8, S3A, B, S12, S13). Similar growth stages have been described in stony coral skeletons72 and avian eggshells73. Both crystal formation stages progress through competitive growth, but crystal morphologies differ between stages two and three. In the second stage, the outlines of the crystals and organic matter are clearly defined, while in the third stage, they are less distinct. This transition in crystal shape suggests the influence of different secreted matrix proteins involved in each growth stage. In the final stage, an organic membrane forms over the crystal layers (Figs. 4C, F, 7, S4C, S4F, S12).
Our proposed four-stage formation process, which includes nucleation within organic fibers, radial crystal growth, perpendicular crystal growth, and the final organic membrane covering, successfully describes the microstructure observed in the argonaut eggcase. This resembles rapidly growing calcareous structures like fibrous calcite in avian eggshells and fibrous aragonite in stony corals74,75,76,77. This resemblance suggests a possible convergence in microstructural strategies optimized for rapid growth. Stony corals with fibrous aragonite skeletal structures, such as Acropora, are among the fastest-growing corals, with a growth rate of up to 10 cm yr-174. Similarly, avian eggshells form in the oviduct at an approximate growth rate of 15 cm yr-1 (0.02 mm h-1)77. The vertical growth rate of the microstructure during argonaut eggcase formation remains unknown, but it has been reported that argonaut eggcases undergo rapid growth towards the adoral area, reaching a maximum length of 17.4 cm in 4–5 months78. Such convergence may reflect the functional demands of efficiently forming robust calcareous structures under biological and environmental constraints. Understanding these processes may shed light on the evolution of extended phenotypes like the argonaut eggcase.
The argonaut eggcase as an extended phenotype
Being a structure independent from the argonaut’s body but functionally integrated into the organism’s reproductive and ecological strategies11,23, the argonaut eggcase may be considered an extended phenotype79. Although its formation is behaviorally controlled1,8, the eggcase’s features like morphology, timing of production, and repair mechanisms, are consistent within species15, suggesting a degree of heritable regulation14,16,24. Its design is analogous to structures shaped by evolutionary selection acting on internal physiology or organism-mediated environmental modifications80,81,82,83. Like other examples of extended phenotypes such as beaver dams81 or spider webs84,85, the eggcase contributes directly to the organism’s fitness by facilitating buoyancy, pelagic reproduction, and egg protection1,35. Although not homologous to the molluscan shell14,22, the eggcase plays multiple organism-level roles and therefore could be interpreted as a behaviorally constructed external phenotype shaped by natural selection for the benefit of the replicator79,80,85,86. This may represent an example of the extended evolutionary synthesis (EES87), where adaptation to pelagicity might have induced the evolutionary trajectory to reinvent a shell-like mineralized structure as an extended phenotype16. In these contexts, the argonaut eggcase illustrates how behavioral and ecological contexts can convergently drive the evolution of functional and structural complexity.

