ExperIENCES MAY VARY:
A handful of users in Taiwan and abroad have had adverse reactions to drugs such as Ozempic, causing governments to issue warnings
By Lee I-chia / Staff reporter
Some GLP-1 receptor agonists, including drugs such as Ozempic, are prescription drugs for losing weight that should only be prescribed by a doctor, the Food and Drug Administration (FDA) said yesterday, adding that 21 cases of suspected adverse reactions have been reported in the past three years.
The FDA issued a news release reminding the public to responsibly use the weight loss drugs, medication originally developed to treat diabetes by mimicking the GLP-1 hormone to lower blood sugar and induce a long-lasting feeling of fullness. This comes after thousands of users in the US reported serious side effects and filed lawsuits and the UK’s health authority updated its guidance for using the drug.
A story in USA Today on Jan. 28 said since the first lawsuit was filed in 2023, there are at least 4,400 patients in the consolidated litigation against weight loss GLP-1 drugmakers, who say drugmakers did not warn them about the serious side effects they have encountered.
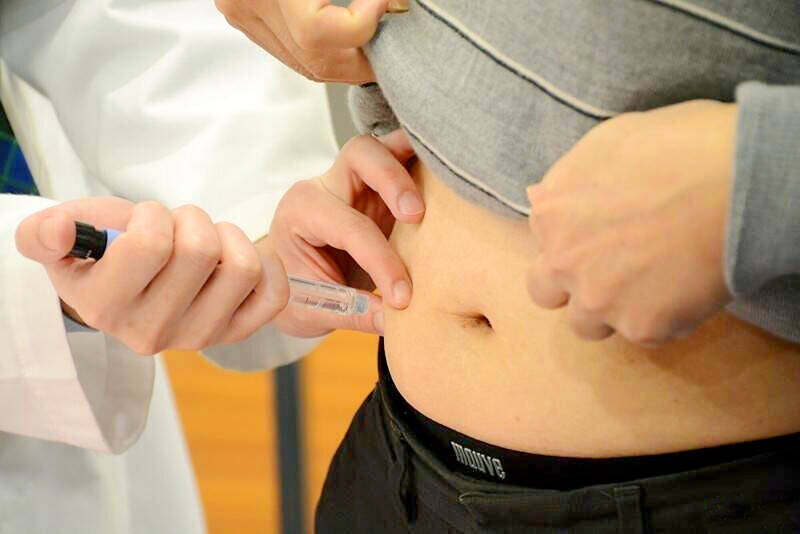
Photo: Taipei Times
The UK Medicines and Healthcare products Regulatory Agency on Jan. 29 also updated its guidance for GLP-1 prescribers and patients, saying “acute pancreatitis is a known, but infrequent side effect of taking GLP-1s,” and “in some extremely rare cases the complications of acute pancreatitis can be particularly severe.”
The FDA said Taiwan has only approved the use of three types of GLP-1 medications — tirzepatide, semaglutide and liraglutide — for weight control.
Between Jan. 1, 2023, and Dec. 31 last year, the FDA’s National Adverse Drug Reaction Reporting System received a total of 21 reports about suspected adverse reactions to these GLP-1 drugs, commonly known as “slimming injection (瘦瘦針)” in Taiwan, it said.
The reported symptoms include gastrointestinal disorders, hypoglycemia (low blood sugar), nausea, vomiting and injection site discomfort, the FDA said.
The agency said it has not observed any abnormalities in overall drug safety or new risks involving the GLP-1 drugs, but users should still remain mindful of side effects.
“The ‘slimming injections’ are prescription drugs that must be evaluated and prescribed by a physician and dispensed by a pharmacist,” it said. “People should not purchase such products online or through unverified channels, as counterfeit or substandard drugs, along with improper use, not only fail to help them lose weight but can also jeopardize their health.”
While some GLP-1 drugs can be prescribed to adolescents aged 12 and older following proper medical assessment, they are mainly prescribed to adults who meet specific criteria, such as having a body mass index above certain thresholds, the FDA said.
“If an adverse reaction occurs, people should seek immediate medical attention,” it said, adding that it “will also continue to monitor local and international safety alerts on GLP-1 drugs, and will re-evaluate their safety, when necessary, to ensure public medication safety.”
