The rise of drug-resistant bacteria is complicating care in hospitals around the world.
Among the most feared are the so-called ESKAPE pathogens, a rogues’ gallery of microbes that “escape” many treatments and drive stubborn hospital-acquired infections. One of them, Acinetobacter baumannii, has become infamous for causing pneumonia and bloodstream infections that can resist even “last-line” drugs.
Now a team in Australia is proposing a different way to hunt it down: by pointing the immune system to a telltale feature on its surface—a sugar.
The sugar is called pseudaminic acid. Humans don’t make it. Many bacteria do, using it as part of their outer coating, helping them move, cling to tissues, and slip past immune defenses.
In a new work published in Nature Chemical Biology, researchers report that antibodies trained to recognize this sugar can clear an otherwise lethal A. baumannii infection in mice, essentially turning the sugar into a molecular beacon.
 Representation of antibody binding the pseudaminic acid to target bacteria with this sugar for destruction. Credit: WEHI
Representation of antibody binding the pseudaminic acid to target bacteria with this sugar for destruction. Credit: WEHI
A Target on Its Back
Pseudaminic acid belongs to a family of unusual sugars found only in bacteria and archaea, and it shows up on proteins and on larger surface structures such as capsules.
That exclusivity is the point. If a therapy targets a molecule our own cells don’t use, it may be less likely to damage healthy tissue. It also offers a way to bypass the antibiotic arms race, in which bacteria develop resistance faster than scientists can create new drugs.
But pseudaminic acid has been hard to study in the messy reality of bacterial surfaces. So the researchers built it themselves.
×
Thank you! One more thing…
Please check your inbox and confirm your subscription.
“This study shows what’s possible when we combine chemical synthesis with biochemistry, immunology, microbiology and infection biology,” said Professor Richard Payne of the University of Sydney.
“By precisely building these bacterial sugars in the lab with synthetic chemistry, we were able to understand their shape at the molecular level and develop antibodies that bind them with high specificity.”
By making the sugar from scratch and pairing it with tiny protein pieces, the scientists created clear targets for the immune system to learn from. The resulting antibodies could recognize the sugar across a range of chemical variations.
The researchers found that their antibodies could spot the sugar on several disease-causing bacteria, including Helicobacter pylori, Campylobacter jejuni, and A. baumannii. Using this approach, they also identified bacterial proteins carrying the sugar that had not been recognized before.
When antibodies do the chasing
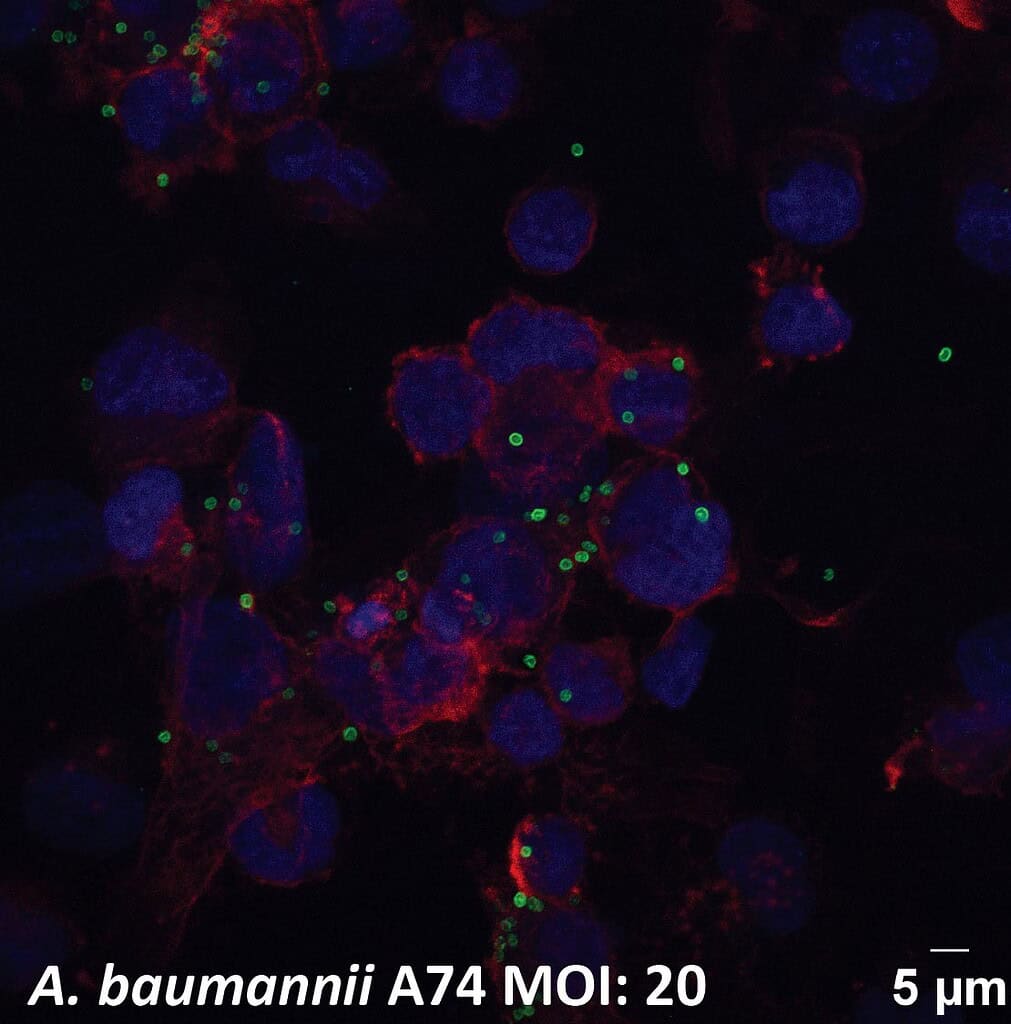 Image showing macrophage immune cells (cell boundaries in red and nuclei in blue) engulfing the multidrug-resistant bacteria Acinetobacter baumannii (green) after treatment with carbohydrate-specific antibodies. Credit: Peter Doherty Institute
Image showing macrophage immune cells (cell boundaries in red and nuclei in blue) engulfing the multidrug-resistant bacteria Acinetobacter baumannii (green) after treatment with carbohydrate-specific antibodies. Credit: Peter Doherty Institute
The strongest test came from an experiment meant to resemble a real medical emergency: treating an infection after it had already started.
Researchers infected mice with A. baumannii. One hour later, they gave some of the animals an antibody designed to recognize the bacterial sugar pseudaminic acid. Every treated mouse survived the following seven days. In contrast, mice that received a control treatment or salt solution became severely ill within about 12 hours.
Blood tests told the same story. Untreated animals carried extremely high levels of bacteria, while scientists could find no living bacteria in the blood of treated mice.
The antibody does not kill bacteria directly. Instead, it acts like a marker that helps immune cells see and swallow the microbes more easily. Laboratory tests showed this immune cleanup happening more efficiently when the antibody was present.
This approach is known as passive immunotherapy—giving patients ready-made antibodies rather than waiting for their bodies to produce their own. The speed of this response could matter most for people at highest risk, such as patients in intensive care units, where drug-resistant infections can become life-threatening in a short time.
The next step is to see whether this success in mice can translate into treatments for people. If it does, the work could point toward a new class of therapies that sidestep antibiotic resistance entirely—by teaching the immune system to recognize bacteria’s molecular fingerprints.