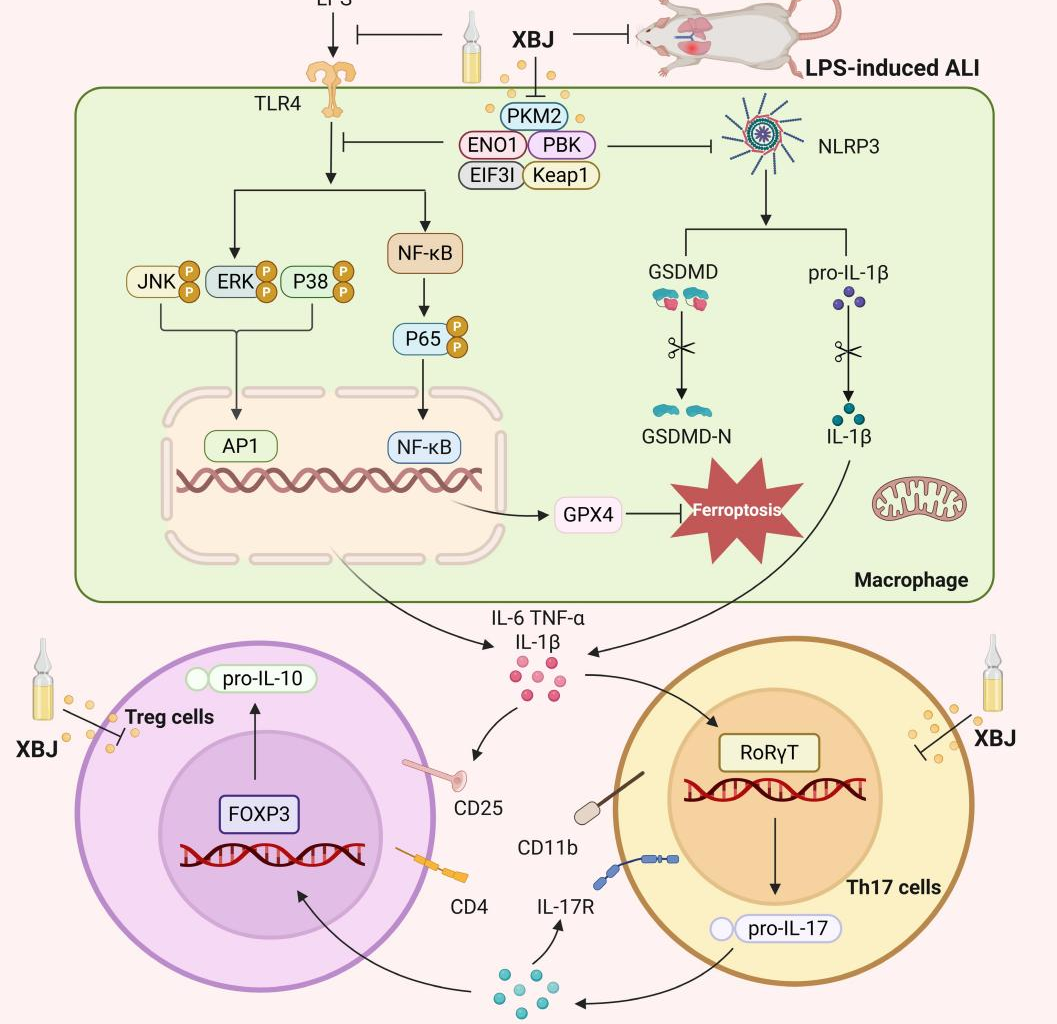
Using an LPS-induced ALI mouse model and LPS-stimulated alveolar macrophages, they found XBJ reduced inflammatory cytokine surges, preserved alveolar barrier integrity, and limited tissue injury, while probing the molecular pathways and intracellular targets underlying these benefits.
ALI can escalate rapidly—from infection-triggered inflammation to leaky air–blood barriers, fluid-filled alveoli, and respiratory failure—yet there are still no specific, widely effective drugs that directly target its core biological drivers. New preclinical research reports that Xuebijing injection (XBJ), a multi-herb traditional Chinese medicine formulation used clinically for respiratory infections, reduced lung damage and inflammation in experimental ALI. The work links XBJ’s protective effects to a coordinated shutdown of major inflammatory signaling pathways, rebalancing of immune responses, and suppression of an iron-driven form of cell death called ferroptosis.
A study (DOI:10.48130/targetome-0026-0005) published in Targetome on 09 February 2026 by Chengpeng Sun’s team, Tianjin University of Traditional Chinese Medicine, provides mechanistic evidence that Xuebijing injection protects against acute lung injury through coordinated multi-target suppression of inflammation, immune imbalance, and ferroptosis, supporting its therapeutic potential.
Using a stepwise in vivo–in vitro–chemical biology workflow, researchers first established an intratracheal LPS-induced acute lung injury (ALI) mouse model and quantified inflammation by cytokine assays (IL-6, TNF-α), enzymatic injury markers (MPO, LDH), lung histopathology (H&E), and immune-cell infiltration profiling via immunohistochemistry and flow cytometry. These measurements showed that XBJ markedly attenuated ALI, lowering pro-inflammatory cytokines and tissue injury indices while reducing alveolar wall thickening and inflammatory infiltration, including fewer CD68+ macrophages and CD11B+LY6G+ neutrophils in lung tissue. Next, to test whether barrier disruption and immune imbalance were corrected, the team assessed tight-junction proteins by immunohistochemistry and western blot and evaluated Treg/Th17-associated transcription factors (Foxp3 and RORγt) alongside inflammatory gene expression by qPCR; XBJ reversed LPS-driven losses of occludin and claudin-1, shifted Foxp3/RORγt signals consistent with improved Treg/Th17 balance, and decreased mRNAs for cytokines such as IL-17a, IL-6, TNF-α, and IL-1α. To connect these outcomes to signaling control, they analyzed MAPK and NF-κB pathway proteins and performed immunofluorescence localization studies; XBJ suppressed pathway activation and modulated cellular patterns of Foxp3, RORγt, COX-2, and IL-6. Because inflammasome activation and ferroptosis can amplify lung injury, the investigators then quantified NLRP3/IL-1β components and ferroptosis/iron-homeostasis markers (e.g., GPX4, TFRC, ACSL4, Slc7a11; DMT1, FPN1) at mRNA and protein levels; XBJ downregulated NLRP3 inflammasome signaling and restored antioxidant/iron-handling defenses, with immunofluorescence confirming reduced ASC/NLRP3 and TFRC plus increased GPX4. Complementary LPS-stimulated MH-S alveolar macrophage experiments used CCK-8, NO assays, ELISA/qPCR, flow cytometry, and pathway immunoblotting, revealing non-cytotoxic XBJ doses that decreased NO, cytokine release, inflammatory transcripts, and MAPK/NLRP3 activation, while also limiting lipid peroxidation and iron accumulation and normalizing GPX4/Slc7a11 and TFRC/DMT1/FPN1. Finally, bead-conjugated XBJ affinity capture with CETSA/DARTS validation identified five direct intracellular targets (ENO1, PBK, EIF3I, PKM2, Keap1), and LC–MS/MS plus docking linked blood/lung-exposed XBJ constituents (e.g., oleic acid, palmitic acid, sugiol, ethyl 4-hydroxy-3-methoxycinnamate, 10,12-octadecadiynoic acid) to target binding, supporting a multi-target anti-inflammatory mechanism.
The findings propose XBJ as a multi-target modulator that can simultaneously dampen inflammatory signaling, stabilize barrier function, rebalance immune responses, and limit ferroptosis—an approach that may better match ALI’s network-like biology than single-target strategies. Beyond XBJ itself, the identified targets (PKM2, ENO1, PBK, EIF3I, Keap1) provide a shortlist of potential intervention points for future drug development aimed at breaking the inflammation–cell death feedback loop in acute lung injury.
###
References
DOI
Original Source URL
https://doi.org/10.48130/targetome-0026-0005
Funding information
This work was supported by the Non-communicable Chronic Diseases-National Science and Technology Major Project (No. 2024ZD0528804), Support Program for Young Faculty’s Research and Innovation Capability Development in Tianjin Higher Education Institutions Initiated by the Ministry of Education, Outstanding Youth Foundation of Tianjin (No. 25JCJQJC00180), Science Foundation of Tianjin (No. 25JCYBJC00620), Young Scientific and Technological Talents (Level Two) in Tianjin (No. QN20230212).
About Targetome
Targetome refers to the complete collection of molecular targets (e.g., proteins, RNA or DNA) that interact with and mediate the effect of a specific biomolecule, such as a drug, toxin, metabolites, transcription factor or microRNA, within a biological system. Targetome is an open access journal publishing rigorously peer-reviewed original research articles, reviews, break-through methods, and perspectives that advance our understanding, identification and validation of molecular targets for new drug development.