Putting carbon to use
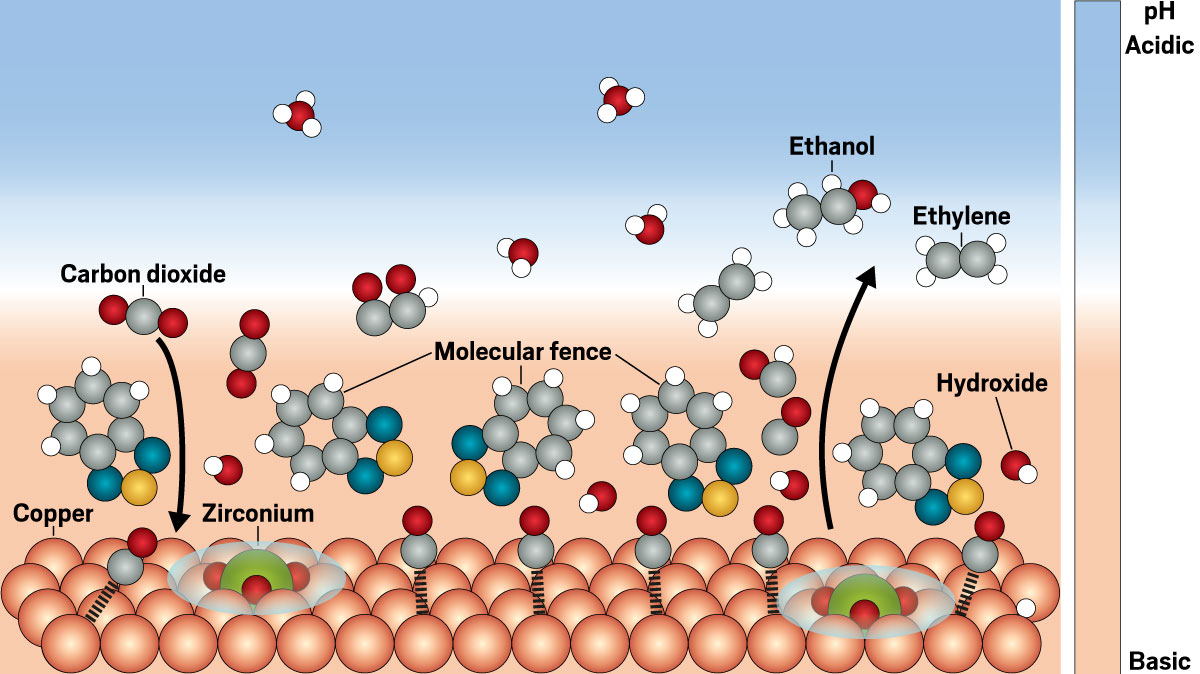
A space-filling model shows a surface of closely packed copper atoms with carbon-oxygen molecules bound to it, that interacts with aromatic thiadiazoles, shown as ball-and-stick models. Carbon dioxide molecules approach the surface, and ethylene and carbon monoxide molecules come out.
Molecules of benzo-2,1,3-thiadiazole (blue and yellow) can stack on top of a copper-zirconium catalyst to create a “molecular fence.” The structure keeps basic hydroxide ions near the copper surface and accelerates the conversion of carbon dioxide into chemical fuels like ethylene and ethanol.
Credit:
Yang H. Ku/C&EN/Adapted from J. Am. Chem. Soc.
Converting carbon dioxide to hydrocarbons could become a cleaner alternative to producing fossil fuels. Now, a team of researchers in China has envisioned a strategy using “molecular fences” to trap reaction intermediates, increasing the conversion rates and energy efficiency of the process (J. Am. Chem. Soc. 2026, DOI: 10.1021/jacs.6c02917). This process is especially selective toward the production of ethylene, the principal pillar of the petrochemical industry, with an annual global production of about 225 million metric tons.
The discovery is innovative and interesting, as well as “highly competitive with state-of-the-art systems under similar conditions,” according to Reginaldo J. Gomes from Argonne National Laboratory, who specializes in the electrocatalytic conversion of CO2 and wasn’t involved in the new study. The molecular fence mimics the mechanism of enzymes, creating an enclosed environment for the reaction intermediates. This increases interactions between the reactants and the catalytic surface to create carbon-carbon bonds, while suppressing simultaneous side processes, such as hydrogen production. “It treats the catalyst interface more like an ecosystem than a single material,” Gomes explains.
Until now, the electrochemical processes for converting CO2 had several downsides, including low selectivity, low stability, and a limited transformation of carbon atoms into fuels, explains corresponding author Guoliang Chai, a materials scientist at Fujian Institute of Research on the Structure of Matter. The molecular fence “reconfigures the catalyst with unprecedented carbon efficiency and 64% selectivity” toward ethylene, he says. The benchmarks surpassed previously published reports and could pave the way to the production of longer hydrocarbons, including propanol and propylene, Chai explains.
The process hinges on getting the pH just right to favor various steps in the overall reaction. The current trend in carbon conversion leans toward running reactions in acidic conditions because they avoid generating counterproductive carbonate salts. On the other hand, basic conditions avoid hydrogen gas formation and promote the creation of carbon-carbon bonds.
With the molecular fence, researchers get the best of both acidic and basic worlds, says Ya-Mi Chuang, who develops fossil fuel alternatives at Imec and wasn’t involved in the study. The molecular fence is built of molecules of benzo-2,1,3-thiadiazole, which both bind to the catalytic surface and stack on top of itself, creating a structure that partially confines the reaction. So the bulk pH of the reaction is still acidic, but the molecular fence keeps basic hydroxide ions—which are generated as oxygen atoms are plucked off CO2—concentrated on the catalytic surface. The molecular fence “decouples the bulk pH from the interfacial pH,” Chuang adds.
She thinks the scalability of the process is promising too. The reaction is compatible with converting high volumes of CO2 into clean chemicals and fuels, and it’s scalable to an industrial electrolyzer. Plus the researchers have reached record selectivity for ethylene and longer hydrocarbon products using “a current density that is industrially relevant,” Chuang says.
Besides synthesizing longer hydrocarbons, molecular fences could find applications in other electrocatalytic reactions, such as oxygen and nitrate reduction reactions, Chai says. Additionally, the team is currently exploring optimizing the reaction in the lab with the hopes of finding an industry partner to launch a pilot-scale plant. So far, the results seem encouraging to Gomes because “the authors already work in flow-cell architectures, which is the correct engineering direction.”
Chemical & Engineering News
ISSN 0009-2347
Copyright ©
2026 American Chemical Society