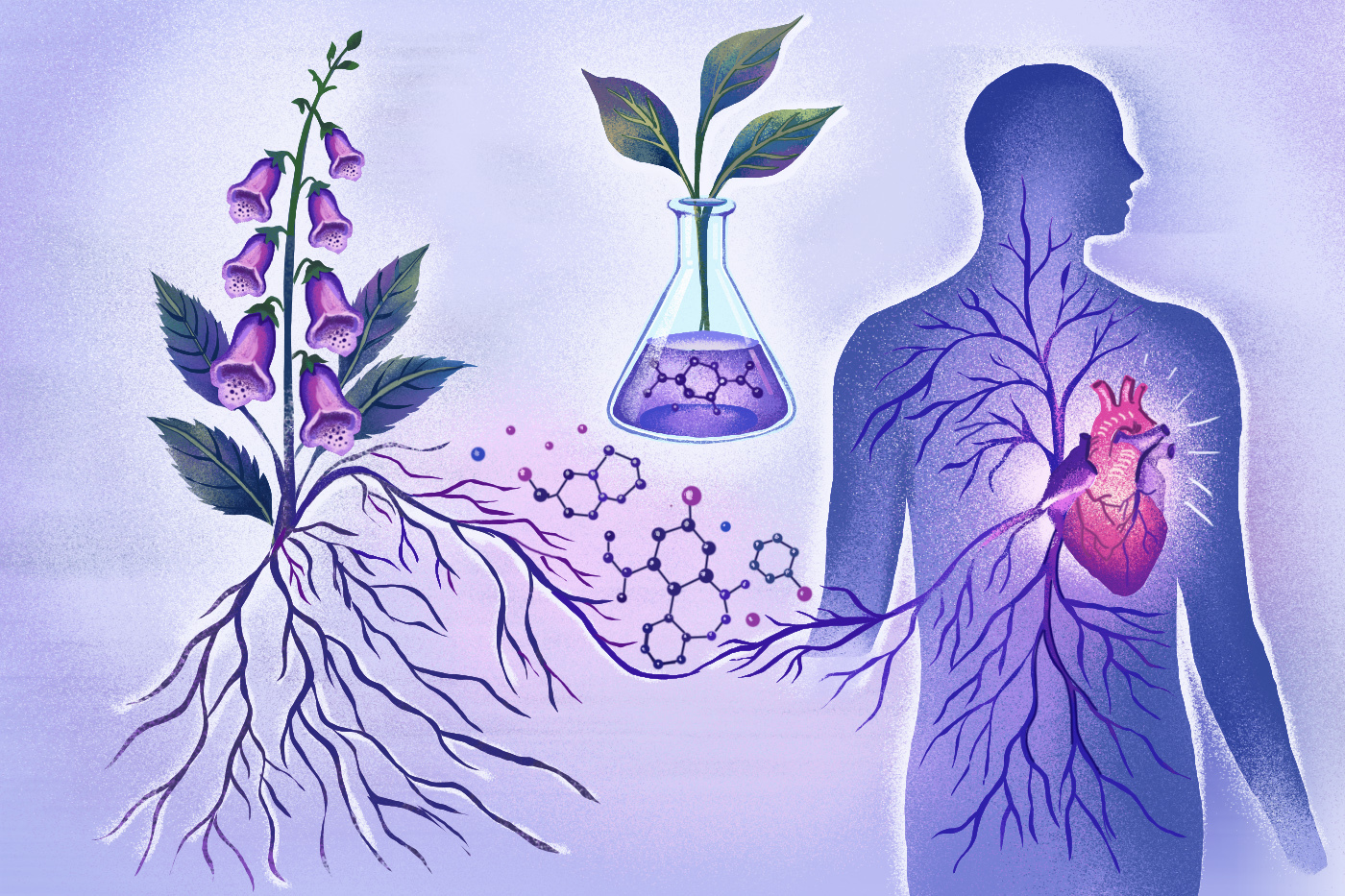
The molecules of a highly toxic plant, known for its bell-shaped purple and pink flowers and found in some home gardens, have long been used to regulate human heart muscles.
But harvesting and purifying the leaves of the foxglove to create the cardiac medication Digoxin has proven to be inefficient, Northeastern University researchers say, because it requires the constant cultivation of new plants. One kilogram of digoxin takes approximately 1,000 kilograms of dried foxglove leaves to produce, for example.
The drug can also be highly toxic if not prescribed in a “narrowly defined therapeutic window,” making its safety and efficacy a point of contention.
But that could soon change.
New research published in Science Advances by Menglong Xu, a post-doctoral researcher at Northeastern University, and Jing-Ke Weng, a Northeastern professor of chemistry and chemical engineering, could make the process of producing the drug safer and more efficient.
Northeastern Global News, in your inbox.
Sign up for NGN’s daily newsletter for news, discovery and analysis from around the world.
Their work, which helps explain how toxic molecules like the one found in Digoxin are formed, might also help in the development of drugs that not only target the heart, but also treat other diseases, the researchers said.
Scientists have long known that different organisms, like foxglove and even some fireflies and toads, evolved to develop similar toxic defense mechanisms.
They are examples of “cross-kingdom endocrine mimicry,” the researchers said, a phenomenon in which an organism from one kingdom of life has hormonal characteristics similar to another and interacts with them. But evolutionarily speaking, the larger scientific community still doesn’t have a clear picture of how plants were able to develop such lines of defense, the researchers said.
 Jing-Ke Weng, Northeastern professor of chemistry and chemical biology, bioengineering and chemical engineering, works in his lab in EXP. Photo by Alyssa Stone/Northeastern University
Jing-Ke Weng, Northeastern professor of chemistry and chemical biology, bioengineering and chemical engineering, works in his lab in EXP. Photo by Alyssa Stone/Northeastern University
For the report, the researchers looked at two species of foxglove, Digitalis purpurea, or common foxglove, and Digitalis lanata, or woolly foxglove.
What Xu and Weng observed was that seed plants like the foxglove follow a steroid-making process similar to one observed in mammals, producing hormones such as pregnenolone and progesterone.
Importantly, this hormonal pathway was developed independently from animals, even though they appear highly similar, the researchers said. These sex hormones “became evolutionary stepping stones” that allowed the plants to grow and adapt. Progesterone, for example, was likely key in seed germination, Weng said, where a plant grows from a seed.
“Basically, these sex hormones were the evolutionary intermediates that eventually gave the leverage for certain plants to be toxic,” he said.
“From a biotechnology perspective, this research provides the long-missing biosynthetic roadmap behind digoxin-like molecules,” said Weng, who is also the inaugural director of Northeastern’s Institute for Plant-Human Interface, which conducts research into plant biology to help to “address humanity’s most pressing challenges and opportunities.”
This discovery could help enable scientists to artificially produce the plants’ toxic molecules in their lab rather than rely on the plants themselves, explained Weng.
“A major knowledge gap is being filled here,” he said.
While the researchers are still working to understand the full biosynthetic pathway at play — more research on that to come, they said — this hormone discovery could bring promise in the development of lifesaving drugs in the future, Weng said.
Take Digoxin as an example. While it’s an FDA-approved drug for congestive heart failure and atrial fibrillation, people who need the drug need to be sure to take it exactly as prescribed. If they exceed the recommended dose, they could be poisoned and die.
Now that researchers have a deeper understanding of Digoxin’s biosynthetic processes, they could theoretically engineer versions of the drug that could be given at higher doses and concentrations, Weng said.
Similarly, Weng posits that the drug can be used to treat cancers not related to the heart by testing the molecules on other organ tissues, such as the liver and pancreas.
“By revealing how foxglove plants precisely make these powerful molecules, our work provides a blueprint for redesigning them, for both safety and potency,” he said.