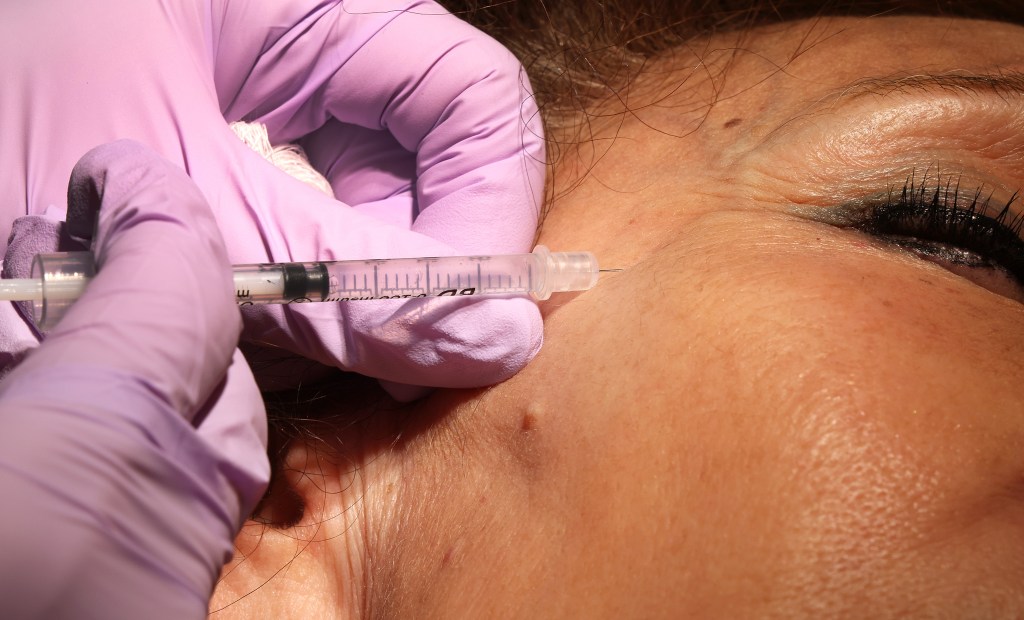
Third in a three-part series.
The promises are alluring: Walk into a Miami med spa and leave with “brighter, more even skin tone” delivered via exosome microneedle injections in your face. Or visit a Winter Haven med spa and get “glowing, youthful skin with as little as one exosome treatment.”
Those promises, though, ignore some dangerous risks.
Federal regulators have issued warnings that unapproved exosomes could leave customers with severe skin infections.
As procedures like exosome therapy become more popular, they draw attention to the potential threat to consumer health posed in some Florida med spas. In their rush to obtain the newest, buzziest treatments, customers may be injected with unapproved, mislabeled or even outright toxic substances.
The U.S. Food and Drug Administration has explicitly warned that some of these products, like exosomes, can cause serious harm.
“People assume that because these treatments are advertised on a website or Instagram, they must be safe,” said Dr. Carlos Wolf, a Miami plastic surgeon. “That’s not always the case. When you inject something directly into your body that hasn’t been tested or approved, you’re taking on risks you may not realize.”
The most common med spa cosmetic injections — Botox and dermal fillers — are FDA-approved, meaning the federal agency has determined, through clinical trials, that they are safe and effective for their intended use.
The offerings at Florida med spas, though, often venture beyond Botox and filler injections.
Customers allow med-spa providers to inject them with everything from salmon sperm to super-concentrated serums of growth factors, despite the risk of serious side effects.
“People increasingly have become obsessed with their looks,” said a Boca Raton med spa worker who asked not to be identified by name. “They only care how big their lips are or how glowy their skin is or how they can use filler to minimize the bump on their nose. They care about results, not necessarily the risks.”
In the last couple of years in Florida, there have been some serious incidents: a woman blinded by a cosmetic injection, another left with facial paralysis, several hospitalizations linked to counterfeit Botox, and the death of a young woman after receiving an IV drip. A Miami woman injected with DNA fragments from salmon sperm sued a local med spa in February 2025, claiming to have been “irreversibly deformed” by the injection.
Florida dermatologist Andrew Miner advises consumers not to assume that all injected products have proven safety studies.
“I’ve seen lots of substances injected into people that should never be injected into a human,” said Miner, with Brevard Skin & Cancer Center and president of the Florida Academy of Dermatology. “What people don’t realize is a lot of those substances will travel widely throughout the body.”
Med spas unmasked: Lack of oversight puts customers at risk
Look no further than Instagram to understand how a stream of glamorous before-and-after photos creates demand for med-spa treatments. Even women in their 20s are going to med spas for preventive care.
“Everyone is doing these cosmetic procedures, and once you start, it becomes insatiable,” said Christine Martinez, a Miami med spa customer. “You start getting injections, and you don’t really stop to think, ‘What are we doing to ourselves?’”
All you need to get a cosmetic injection in Florida is money and a signature on a consent form — regardless of how risky the treatment might be. That signature, along with a verbal discussion of risks, generally protects the provider from legal liability if the patient experiences a disclosed risk and the procedure was performed correctly by a health care provider.
That waiver applies even when a treatment is unproven or off-label.
“Whether a treatment can be legally offered doesn’t turn on whether the patient is told it isn’t FDA-approved,” said Tabitha Taylor, a med-spa attorney with Bryant Taylor Law in Plantation. “The key question is whether the product itself is legally permitted to be marketed or administered under federal and state law.”
In Florida, med spa regulation is complex. No single agency has sole authority or responsibility, although Florida’s Department of Health pursues complaints against med-spa workers, usually after someone is injured or law enforcement gets involved.
“There is no oversight,” said Thomas Graham, a South Florida personal injury attorney who represents people harmed at Florida med spas. “They can buy these products that are unsafe, untested, bring them in and market them to people who don’t have the background to understand why it might be dangerous. If something really bad happens, they are given a slap on the wrist.”
Thus far, attempts to regulate the industry in Florida through legislation have failed.
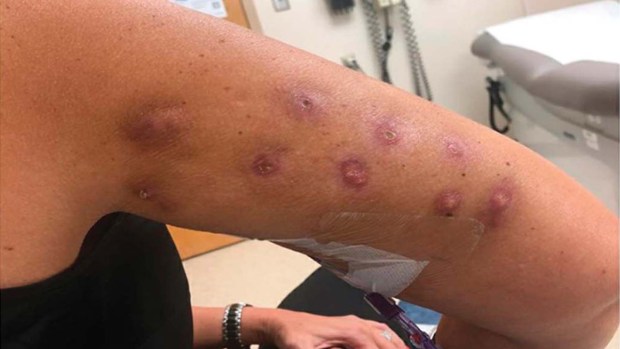 The FDA warns that fat dissolvers are unapproved and may cause significant reactions. A woman who received injections with lipodissolve, a drug that is not FDA approved, reported a severe reaction. (Kaur, H, C Reyes-Barron, WH Sipprell, A Cameron, T Louie, PR Tsai, and G Scott via FDA)
The FDA warns that fat dissolvers are unapproved and may cause significant reactions. A woman who received injections with lipodissolve, a drug that is not FDA approved, reported a severe reaction. (Kaur, H, C Reyes-Barron, WH Sipprell, A Cameron, T Louie, PR Tsai, and G Scott via FDA)
Troubling fat dissolvers
Looking to get rid of love handles or flabby thighs? A quick Google search in Florida for “med spas near you” returns dozens that offer lipodissolve injections to break down fat cells.
The marketing pitch for fat dissolvers is simple: Get a sculpted look without surgery. The risks, though, include uneven lumps or damaged tissue, open sores and permanent scarring.
Med spas in Miami, West Palm Beach, Tampa and Orlando are among dozens across the state that offer the trendy Lemon Bottle fat dissolver shots. They tout the treatment as an “instant” and “painless” fat-dissolving solution, and an alternative to liposuction.
 Lemon Bottle says its product is “completely safe,” has “no side effects,” and delivers “instant results.” The U.S. Food and Drug Administration does not approve or endorse Lemon Bottle shots, classifies them as unapproved drugs, and warns that they carry significant and unknown risks. (AP file via Business Wire)
Lemon Bottle says its product is “completely safe,” has “no side effects,” and delivers “instant results.” The U.S. Food and Drug Administration does not approve or endorse Lemon Bottle shots, classifies them as unapproved drugs, and warns that they carry significant and unknown risks. (AP file via Business Wire)
On its Instagram page, Lemon Bottle, made in South Korea, says its product is “completely safe,” has “no side effects,” and delivers “instant results.” The U.S. Food and Drug Administration says differently. It does not approve or endorse Lemon Bottle shots, classifies them as unapproved drugs, and warns that they carry significant and unknown risks.
A surge in reports of safety issues linked to Lemon Bottle has sparked international concern. Save Face, a UK-based organization that accredits medical aesthetics practitioners and investigates complaints, has received 210 complaints about Lemon Bottle over the last two years. These include claims that customers were left with uneven results, saw no results at all, or suffered prolonged bruising, swelling, infections, abscesses and even necrosis (the death of body tissue).
“There is no data about how safe or effective these treatments are or what potential complications could arise as a result,” Ashton Collins, director of Save Face, told the South Florida Sun Sentinel. “We have had reports of ladies experiencing huge lumps at each injection site. They could be on the stomach, the thighs, the neck, or the chin area … in very visible places.”
South Florida resident Carmen Hernandez says she visited a Miami med spa for Lemon Bottle injections to reduce the loose fat on her upper arm. She said the experience was life-changing — although not in the way she had wanted. “Instead, I got months of pain, and I still have scars,” she told the Sun Sentinel.
Capitalizing on the strong desire for weight loss, many Florida med spas are offering untested or unregulated fat-dissolving injectables to eliminate fat in “problem areas” such as the chin, legs, waist, upper arms and abdomen.
Only Kybella is FDA-approved — and only for use under the chin. Yet dozens of Florida med spas offer various lipodissolve treatments — including Kybella — for many parts of the body, according to their websites and social media accounts.
On its website, a Miami-area med spa offers patients lipodissolver injected into their chin, abdomen, thighs, upper arms, and flanks — a procedure the promotional offer says takes only 20 to 30 minutes.
While convenient, these injections can also be harmful.
In December 2023, the FDA issued a warning about the dangers of fat-dissolving injections after many reports of severe side effects, including permanent scars, serious infections, skin deformities, cysts, and deep, painful knots. The warning included disturbing photos of a woman who received lipodissolve treatments and sustained multiple red, infected knots at the injection sites on her upper arm.
In Central Florida, a med spa promotes fat-dissolving treatments, including Kybella and PCDC (lipodissolve), to reduce bra fat, upper arms, love handles and thighs. Neither is FDA-approved for this use, and the federal agency has said they pose significant risks.
Yet the med spa’s website indicates otherwise. “Both PCDC Liquid Lipo and Kybella are safe, FDA-approved treatments. Phosphatidylcholine and deoxycholic acid are naturally occurring substances in the body, making them well-tolerated by the majority of our patients,” the website reads.
Dr. Joshua Lampert, a South Florida plastic surgeon, said there are concerns about injecting fat dissolvers into various body parts and even using Kybella in ways it wasn’t intended.
“Kybella is approved for injection into the chin, which is a smaller area. But if someone is injecting into a larger area, they are giving a larger dose, and that’s guesswork,” Lampert said.
 The FDA warns that fat dissolvers are unapproved and may cause significant reactions. A woman who received injections with lipodissolve, a drug that is not FDA approved, reported a severe reaction. (Kaur, H, C Reyes-Barron, WH Sipprell, A Cameron, T Louie, PR Tsai, and G Scott via FDA)
The FDA warns that fat dissolvers are unapproved and may cause significant reactions. A woman who received injections with lipodissolve, a drug that is not FDA approved, reported a severe reaction. (Kaur, H, C Reyes-Barron, WH Sipprell, A Cameron, T Louie, PR Tsai, and G Scott via FDA)
Where fat dissolvers are injected can also cause harm.
Kate Dee, author of “MedSpa Mayhem: The Good, the Bad, and the Ugly Secrets of the Aesthetic Industry,” told the Sun Sentinel about the risk of hitting or damaging a nerve when administering a fat-dissolving injection. “Some nerves are coated with fat,” explained Dee, who owns Glow Medispa in Seattle. “If you inject the fat dissolver near a nerve, you can temporarily or permanently damage the nerve.”
Regenerative treatments, anti-aging injections
The newest injection treatments drawing customers to Florida med spas may also be the riskiest. These fall into categories like anti-aging and regenerative treatments.
Exosomes: A Broward County med spa calls its exosome treatment “a cutting-edge cellular skin regeneration treatment that restores skin health from the inside out.”
Other med spas across the state make similar claims. But the FDA has not approved any exosome products for injection, IV treatment or topical use with microneedling.
The agency has issued consumer alerts stating that its true efficacy and safety are unknown. There have been cases of serious infections from contaminated exosome injections.
Safety concerns have also put exosomes on Save Face’s radar in the UK.
“The concern is that there is a black-market trade of human-derived exosomes and obviously, with that, you have the risk of blood-borne viruses and all sorts of contaminations being passed through the skin,” Collins at Save Face, the UK accreditor for aesthetic practitioners, told the Sun Sentinel.
Save Face’s website advises, “Choosing a skilled practitioner who uses high-quality, clinically tested exosome products enhances the safety and effectiveness of treatments.”
Med spas unmasked: Can you trust the hand that holds the needle?
PDGF: Marketed as a next-generation treatment at Florida med spas, Platelet-Derived Growth Factor injectables mimic the growth factor found in humans but are manufactured in a lab. The FDA has approved certain PDGF drugs for clinical “topical use only” so any med spa that injects it is doing so outside of the manufacturer’s labeling. There is no data on safety when it’s injected into the under-eye area or the scalp.
The lead manufacturer of PDGF has cautioned that injecting PDGF too superficially at high concentrations or in delicate areas may potentially cause localized collagen nodules, transient swelling, or discoloration. Yet some med-spa providers are doing so, according to their websites.
Louiza Tarassova, an Orlando attorney who represents people harmed at med spas, said one of her clients had a temporary loss of vision and permanent scars, pain, and bags under her eyes after being injected with PDGF at a Florida med spa.
“It’s an over-the-counter, non-FDA-approved product that a bunch of these injectors are getting directly from the manufacturer,” Tarassova said. “It comes in a syringe and apparently, on the syringe, it says do not inject because it’s not FDA-approved, but they’ve been injecting it into people.”
Tarassova said an additional risk is that when unproven injectables cause a reaction, even licensed doctors often don’t know how to respond.
“The problem with the med spa industry is it’s so lucrative and it’s a cash business and it’s all about what’s the latest product that we can sell to our customers, but they’re forgetting that they’re practicing medicine.”
 Peptide therapies have exploded in popularity at Florida med spas, touted for their benefits in healing and longevity. (Carline Jean/South Florida Sun Sentinel)
Peptide therapies have exploded in popularity at Florida med spas, touted for their benefits in healing and longevity. (Carline Jean/South Florida Sun Sentinel)
Touted as an energy booster, anti-aging solution, and even a brain enhancer, nicotinamide adenine dinucleotide costs as little as $50 to $75 a shot.
However, the FDA has not approved NAD+ injections for anti-aging.
While NAD+ treatments are marketed at med spas as a way to support “longevity,” there is no medical evidence to support this claim, according to federal regulators. The potential serious risks of NAD+ injections include infections at the injection site, nerve damage, and unpredictable side effects such as nausea, rapid heartbeat, chills and vomiting, federal regulators say.
Several med spa customers have detailed their experience with an NAD+ injection on social media. One writes on Reddit.com of a rapid surge in his heart rate that landed him in the Emergency Room and took about a week to normalize. Another writes of a similar reaction, calling it terrifying.
Aware that med spas are giving out NAD+, and even using compounded product, the FDA issued a warning:
“FDA has received adverse event reports following use of NAD+ injectable drugs, including severe chills, shaking, vomiting and fatigue with some requiring medical treatment. These reactions are consistent with excessive levels of endotoxins.”
Peptides: Peptide therapies have exploded in popularity at Florida med spas, touted for their benefits in healing and longevity. A Pompano Beach med spa promotes peptide injections for improved energy, while an Orlando med spa touts peptide therapy for firmer skin.
However, the FDA has expressed safety concerns and notes that there is no way to know the right dosage, purity, potential contaminants and impact of long-term use of some peptides.
The federal agency said unapproved peptides tend to come from pharmacies that mix or compound ingredients and cited risks such as potential immune reactions or manufacturing impurities. Many of the products have never been extensively studied in humans, raising concerns about insufficient human safety data and the potential for serious side effects. The FDA has created a list of more than two dozen peptides that should not be compounded and used by providers due to safety concerns. A few med spas and wellness clinics have pushed back against the FDA’s stance on peptides, calling it an overstep.
However, if the vial says “not for human consumption,” or “for research use only,” it is illegal for a Florida provider to inject it into a customer outside of a study.
But Florida healthcare attorney Jeff Cohen likens enforcement of the law to exceeding the speed limit, with violators rarely being pulled over. “The speed limit may be 65, but everyone is doing 75.”
Counterfeit and diluted product
Every year, thousands of Floridians seek Botox injections to reduce wrinkles, some buying services for below-market prices, unaware that the products are fake, expired or diluted versions of the cosmetic treatment.
As a result, what may seem like a routine beauty treatment — a few Botox injections — has left some patients with slurred speech, droopy eyelids and even breathing difficulties requiring respiratory support.
“Patients don’t really know what part of the world these med spas are bringing products in from and whether they are diluting it before injecting,” said Wolf, the Miami plastic surgeon.
In spring 2024, dozens of people in nine states, including Florida, became sick and even hospitalized from reactions to fake Botox they received at med spas — triggering a Centers for Disease Control & Prevention investigation.
“If the price is incredibly low, that’s a red flag,” said Dr. Sean McNally, a plastic surgeon who consults for medical boards.
Martinez, the Miami med spa customer, said she learned this through experience. She wanted Botox but found the prices charged by dermatologists and plastic surgeons to be outside her budget. So she searched Instagram and found a med spa deal. She now questions the product’s authenticity.
“I feel like I got duped,” she said. “It didn’t last long.”
In the last two years, med spa workers in Orlando, Fort Lauderdale, Miami, Jacksonville and Hallandale Beach have been arrested for practicing medicine without a license and for injecting patients with unknown substances after the products they were injecting led to injuries. None of the criminal cases has gone to trial yet.
Only weeks ago, on Feb. 19, a worker at a Brickell med spa was arrested by the Miami-Dade Sheriff’s Office and accused of administering Botox injections without a license. Investigators determined the woman was using an injectable called Toxa, an unapproved Botox alternative imported from South Korea. She had been advertising her business on Instagram.
Experts recommend that med spa customers look for branded product brochures. They also suggest asking to see the vial and the product’s box before receiving a cosmetic injection — and question any pricing deal that sounds too good to be true.
“If they already have it drawn up in an unmarked syringe on a table, that’s not okay,” McNally said.
IV drip
Beyond injectables, med spas are increasingly jumping on the broader wellness culture, offering intravenous drips.
South Florida’s Nicole Prol says she regularly gets immunity-boosting IV drips at med spas. “You choose all the add-ons you want to put in,” she explained.
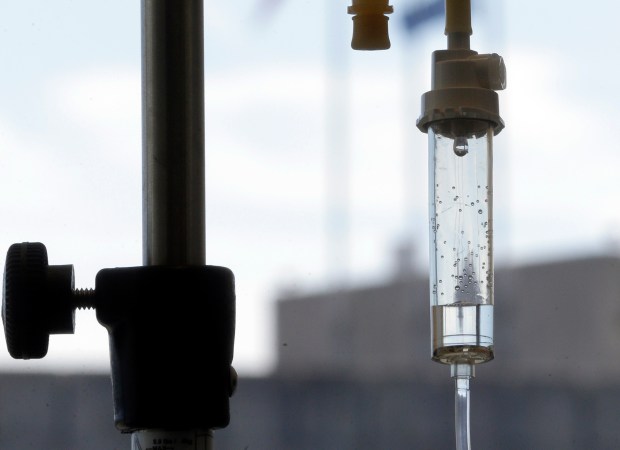 IV drips have become more common in Florida, marketed for a variety of anti-aging and rejuvenation uses. (Gerry Broome/AP file)
IV drips have become more common in Florida, marketed for a variety of anti-aging and rejuvenation uses. (Gerry Broome/AP file)
For $175 to $250 and 20 to 40 minutes of her time, she usually gets energized from a multivitamin drip, she said. However, she has found that not all med spas selling IV drips offer the same quality. “At some places, I have gotten no effect from them.”
Among Florida’s youth-oriented, sun-worshipping residents, IV infusions have surged in popularity. These drips are positioned as a fast-track solution for everything from curing hangovers and boosting energy to improving skin clarity and strengthening the immune system.
Using catchy names like Skinny Drip, Athlete Drip or even Million Dollar Glow IV Drip, high doses of vitamins, minerals and other compounds are infused directly into the customer’s bloodstream.
“I think that patients usually feel good after doing an IV treatment,” said Jennifer Martinez, a physician assistant and co-owner of SkinLocal, which operates four Florida med spa locations. “They feel more hydrated. They can feel some more energy. I do think there’s a benefit to it.”
Doctors and scientists, though, are raising serious flags about the potential danger of IV drips for wellness. The risk includes everything from vein irritation to infection, to toxicity from fluid overload to complications from a lack of medical history screening.
Some infusions can be dangerous for individuals with heart disease and kidney problems, whose bodies are not capable of processing the salts and fluids rapidly added to the body.
Med spas and IV drip bars are supposed to ask customers to fill out a medical questionnaire, according to Florida health officials.
Dr. David Seres, director of Medical Nutrition and professor of Medicine at Columbia University Medical Center, said no one needs an IV infusion unless they have a medically diagnosed deficiency.
“Hopefully, most of the people who are giving these kinds of treatments have a sense of what the toxic extremes are and know enough to avoid them,” Seres said.
Stephanie Balais, an aspiring South Florida nursing student, died in 2018 at age 25 after receiving a lethal dose of the mineral selenium as an IV treatment for immune support.
The nursing student’s death prompted a political push in 2023 to pass a Florida law to create new safety requirements for giving IV wellness treatments. The proposed law, however, did not pass and has not been reintroduced. Lobbyist Chris Nuland says the proposed law was drafted so broadly that it would have affected medically necessary treatments like IV for chemotherapy as well as the med spas and drip bars it was intended to regulate. He said the proposed law “died of its own weight.”
In addition to toxic dosages, concerns with IV drips include infection. The FDA has warned that some med spas and IV hydration clinics improperly sterilize IV equipment. The danger is that bacteria can enter the bloodstream through the IV site.
Dr. Shasa Hu, a dermatologist and director of the cosmetic division at the University of Miami, said she considers this a serious concern. Anytime the skin is broken, there is a risk of introducing bacteria into the bloodstream, which can lead to a severe infection, she explained.
“How do we know what is being given intravenously is not contaminated with bacteria? How do we know the handling procedure is up to the good clinical practice standard?” she said. “Those are questions customers should be asking because you’re paying someone and putting stuff in your body.”
Martinez, co-owner of SkinLocal Med Spa in South Florida, wants to see stricter ownership requirements. In New York, California and Texas, physicians or licensed medical professionals must own all or the majority of a med spa. In Florida, anyone can own a med spa or drip bar. “It would be nice if Florida could eventually pass a law that IV drip bars or even med spas should be owned by a medical provider, not just a business owner,” she said.
She explained: “A business owner can say, ‘Let’s use less of this product because I’m cutting costs,’ and they can be looking at more of the profit part of it and it really should be what we need to do for the patient first.”
Med spas unmasked: The risks of injectables
Dangerous injectors, infusors and laser technicians
In addition to substances being problematic, the person delivering the injections can cause harm.
“A big issue with fillers is if they’re injecting into an area that has blood vessels nearby, and if they’re not using an appropriate technique, if they inject the filler into the blood vessel, it can cause that blood vessel to freeze up completely,” said plastic surgeon McNally. ”It can kill the skin on your nose, on people’s lips, they can even blind someone.
“So, if you’re having these injections done with somebody who doesn’t truly know their anatomy, and doesn’t truly know how they should be done, it can have really serious consequences,” McNally said.
In Port St. Lucie, a couple was arrested twice in the summer of 2024 at two different med spas, just weeks apart, and accused of causing great bodily harm and practicing medicine without a license after several botched surgeries and injuries from harmful injections. Customers complained to the authorities about gaping wounds and permanently damaged skin. The criminal case against the couple is ongoing.
“It seems to be getting worse,” Aventura plastic surgeon Lampert said of Florida’s unlicensed med spa workers. “It’s like fast food places. More and more are opening with people who claim to be experts who have had zero training and still do injections.”
Lampert, who has his own plastic surgery practice but also works in the emergency room at HCA Florida Aventura Hospital, has treated patients for severe abscesses on the face and other complications exacerbated when injectors lack the medical knowledge or credentials to respond to a complication. “They sit on it and wait too long, and then they finally send the patient to the ER, and by that time it is so much worse.”
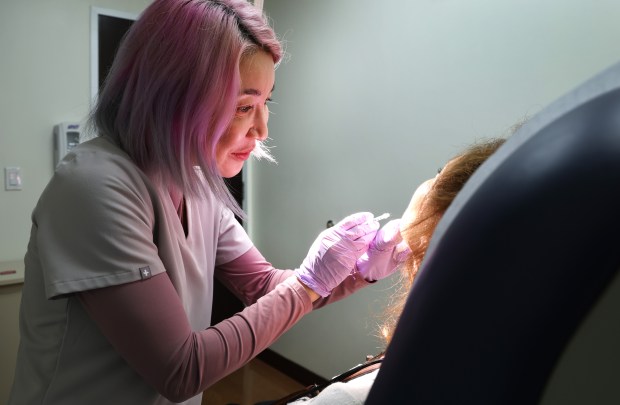 Dr. Shasa Hu, an associate professor at the University of Miami, performs an injection procedure in Miami. She advises patients to make sure that if they are receiving injections, they should be aware that the handling is “up to good clinical practice standards.” She says customers should ask questions “because you’re paying someone and putting stuff in your body.” (Carline Jean/South Florida Sun Sentinel)
Dr. Shasa Hu, an associate professor at the University of Miami, performs an injection procedure in Miami. She advises patients to make sure that if they are receiving injections, they should be aware that the handling is “up to good clinical practice standards.” She says customers should ask questions “because you’re paying someone and putting stuff in your body.” (Carline Jean/South Florida Sun Sentinel)
Injuries, reactions and response
It’s difficult to know how many people have been injured by substances or IV infusions at Florida med spas because the incidents often are not reported to local or state health departments. But as more med spas open, Florida dermatologists and emergency room doctors say they are treating more patients who arrive with complications from med-spa procedures.
In her dermatology practice at the University of Miami, Hu says she has seen everything from oozing infections to skin discoloration to horrible scarring from injectables.
“We end up treating a lot of patients with undesirable or poor results or sometimes even complications from procedures that they receive from a medispa or from an unlicensed provider or people who are not qualified to do these injections because of the complexity,” she said.
Luisa Rodrigues, a patient at the now-closed Tonya Beauty Med Spa in Miami, hired Miami attorney Thomas Graham in February 2025 to sue the med spa after she said an injection harmed her.
Rodrigues’ legal complaint says the aesthetician employed by Tonya Beauty injected a compound extracted from salmon sperm into her face beneath each of her eyes, delivering an excessive amount that left her with irreversible deformities on her face.
“The employee who injected Luisa Rodrigues did so negligently, and directly and the proximity caused Luisa Rodrigues to suffer permanent swelling, inflammatory changes and discoloration beneath her eyes, leaving her with irreversible deformities on her face and corresponding injuries,” the court complaint says.
The case was settled in September for an undisclosed amount. Tonya Silva Pereira, owner of Tonya Beauty, could not be reached for comment. She appears to have relocated to Brazil, according to court documents in a similar case against her med spa in Orlando.
Graham said Rodrigues’ experience is not isolated. Although med-spa providers have become influencers on social media, he emphasizes that a large Instagram following or good Google reviews don’t ensure safe measures are taken or that injectors are licensed or experienced.
“Anyone can create an Instagram profile and advertise injections,” Graham said. “You need to do some checking. You can’t really expect the state to monitor social media.”
Support local journalism like this by considering a tax-free donation to the Sun Sentinel’s Community News Fund. Click here to find out more.
What is a customer to do?
After an injury, it is not easy to sue a med-spa provider for medical malpractice in Florida, according to Graham. Before filing a lawsuit, Florida law requires the person to go through a pre-suit process, which includes hiring a medical expert who must sign a written opinion stating that the provider failed to meet the proper standard of care.
“We turn down nine out of 10 clients that come looking for help for a medical malpractice and almost unanimously, we have to turn down these med spa cases because they’re either so hard to prove or there’s no insurance or there’s a legal challenge with the med malpractice statute,” Graham said.
In Florida, the state regulates the people who work at med spas, overseeing licensing and medical practice rules. Oversight is primarily complaint-driven. Actual physical inspections typically occur only after a patient complaint or a competitor’s tip regarding unlicensed activity.
“You’re not going to have monitoring on a regular basis,” said Taylor, the med spa attorney. Just like with lawyers, “the expectation is there are rules and you follow them. It’s up to the consumer to a certain extent to do their homework.”
When it comes to the products used, the FDA regulates the safety, effectiveness, manufacturing, and marketing of drugs and medical devices. However, the agency’s authority is limited to regulating the product itself, not how the med-spa provider uses it. The agency does not walk into every med spa to monitor what they inject, nor does it monitor whether spas advertise non-FDA-approved treatments.
Instead, the FDA focuses on what can be sold to med-spa providers and has cracked down on several international distributors for selling unapproved versions of Botox to U.S. med spas. It has also sent warning letters to med spas that advertise injections as totally risk-free or use misleading photos, a violation of the Federal Food, Drug and Cosmetic Act.
Lawyers say the industry is evolving faster than the law, leaving each state to figure out how to keep customers safe.
Michael Byrd of BrydAdatto, a business law firm specializing in providing counsel to med spas, explains that the standards and regulations that apply to med spas vary widely and some states are more aggressive about enforcement than others.
“The real problem is that there’s a lack of clarity in each state as to what the rules are,” Byrd said. “We see that with med spas not being compliant because they can’t figure it out, but we also see it with the enforcement arm. They don’t really understand the laws and what they’re trying to enforce.”
Byrd says he would like to see the adoption of model rules for the industry to serve as a guide for all states.
Sun Sentinel investigates: Med spas unmasked
Part 1 (Feb. 19): Florida med spas operate with little government supervision and often without much oversight by physicians.
Part 2 (Feb. 26): Med spa practitioners often tout certifications and other credentials earned after only a brief training session.
Part 3 (today): The youthful glow promised by some Florida med spas often involves unproven substances known to trigger severe adverse reactions.
Ways to protect yourself
Medical experts advise anyone considering a cosmetic treatment to verify that the provider is a licensed medical professional and to ask whether the provider uses only FDA-approved products for the specific treatment area.
“If you were to ask clinicians, physicians, nurse practitioners, and PAs, ‘Hey, do you sometimes recommend or prescribe or inject things that are not FDA-approved?’ In the wellness space, it’s common, and the answer would be yes,” said Cohen with the Florida Healthcare Law Firm. “The patient needs to look carefully at informed consent. They should be reading that paperwork.”
Anyone who wants to try a non-FDA-approved treatment should verify that the provider can handle all potential complications.
“The most differentiating factor between a dermatologist who is an aesthetic provider versus a med-spa provider is the expertise,” says Dr. Marianna Blyumin-Karasik, a Davie dermatologist with Precision Skin Institute. “We have a wealth of training. Patients need to do their due diligence.”
However, Prol, the Broward County med spa customer, thinks legislation on med spa licensure would be a step to keeping her and others safer. But Florida’s attempt to regulate medical spas that offer prescription medications by putting the facilities under direct oversight by the Florida Board of Pharmacy did not advance this Legislative session.
“A lot of people just go wherever it’s cheaper,” she said. “They don’t really know if they are getting fake product, or less product than they should be or something that shouldn’t be injected … so it probably would be a good thing for the state to better regulate med spas.”
Sun Sentinel reporter Cindy Goodman can be reached at cgoodman@sunsentinel.com.