The study is published in ‘Science’. It is led by Eva González-Suárez, researcher at the Spanish National Cancer Research Centre.
The team has unexpectedly discovered that cells from the brain immune system play a role in the sexual maturation process. The link between them is RANK, a protein involved in mammary gland development.
The research has been carried out in animal models, but it has also found genetic mutations associated with a rare syndrome related to infertility in humans.
The kick off signal for puberty begins in the brain. Specifically, in the hypothalamus, where specific neurons release a hormone that activates the hypophysis, at the base of the skull, which then releases other hormones to start maturation of the gonads -ovaries or testes. This mechanism, which ultimately leads to a fertile organism, is known as the hypothalamic-pituitary-gonadal (HPG) axis.
A study by Spain’s National Cancer Research Centre (CNIO) has just discovered in animal models that two previously unsuspected elements are also involved in this hormone regulating system: microglia – defensive cells of the nervous system – and the protein RANK, which contributes to bone remodelling and is essential in the function of the mammary glands.
The article is published in the journal Science. It is led by Eva González-Suárez, head of the CNIO Transformation and Metastasis Group, who discovered in 2010 the key role played by RANK in the development of breast cancer. The first author is Alejandro Collado, a researcher from the same group and co- corresponding author.
Immune cells to modulate fertility
The hypothalamic-pituitary-gonadal axis regulates many processes related to reproduction. Its main players in the hypothalamus are the gonadotropin-releasing hormone neurons (GnRH). Gonadotropins are two pituitary hormones that control the onset of puberty, the development of the gonads, and fertility. It was previously known that GnRH neurons are modulated by other neurons, but not that immune cells could influence their functionality.
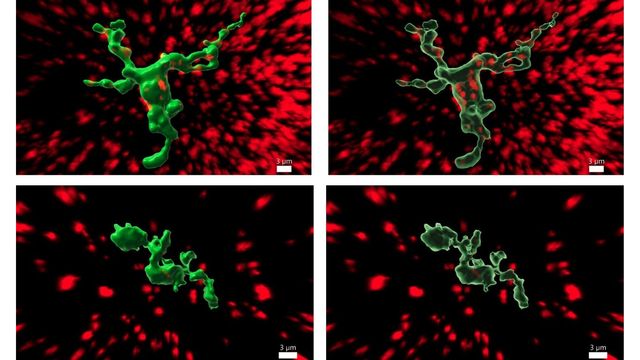
This is the newly discovered function of microglia, cells in the central nervous system that eliminate potential threats and unnecessary molecules. “Finding fertility-regulating cells that are not neurons, but rather immune cells, is important,” highlights González-Suárez.
The study shows that the way microglia regulate the function of GnRH neurons is by expressing the RANK protein.
When the CNIO group suppressed RANK expression in animal models, the reproductive function became distorted, both in males and females. In specimens born without RANK, or when it was removed in prepubescent animals, there was a reduction in sex hormones and a loss of gonad function, known as hypogonadism, and puberty was delayed in these animals. When RANK was eliminated in sexually mature specimens, they became infertile within a month.
New mutations for a human syndrome
To investigate whether RANK might play a role in human fertility, researchers genetically analysed samples from patients with congenital hypogonadotropic hypogonadism, a rare genetic syndrome associated with delayed or absent puberty and infertility. It was known that it is caused by problems in GnRH neurons or in the molecules they produce. The research identified mutations in the gene encoding the RANK protein in some patients.
“These results show that RANK could be a therapeutic target for endocrine disorders and syndromes affecting fertility, as well as a candidate gene for the molecular diagnosis of congenital hypogonadotropic hypogonadism,” according to the authors.
González-Suárez emphasises that “the role of microglia in regulating the function of ‘reproductive’ neurons is new, and this regulation associated with RANK can occur in other axes, for other functions, such as the appetite-satiety axis, the stress axis, etc.”
The importance of collaboration
The authors also want to highlight this research as an example of the importance of interdisciplinary collaboration. “My doctoral thesis started out with the question of whether the RANK protein played any role in the development of mammary tissue, in the breast itself, during puberty,” Collado explains. “When we realised that we needed to explore issues implying fertility, neurons and brain cells, we started consulting colleagues from other fields.”
Reference: Collado-Sole A, Borjini N, Zhai J, et al. Microglia Rank signaling regulates GnRH neuronal function and the hypothalamic-pituitary-gonadal axis. Science. 2026:eaeb6999. doi: 10.1126/science.aeb6999
This article has been republished from the following materials. Note: material may have been edited for length and content. For further information, please contact the cited source. Our press release publishing policy can be accessed here.