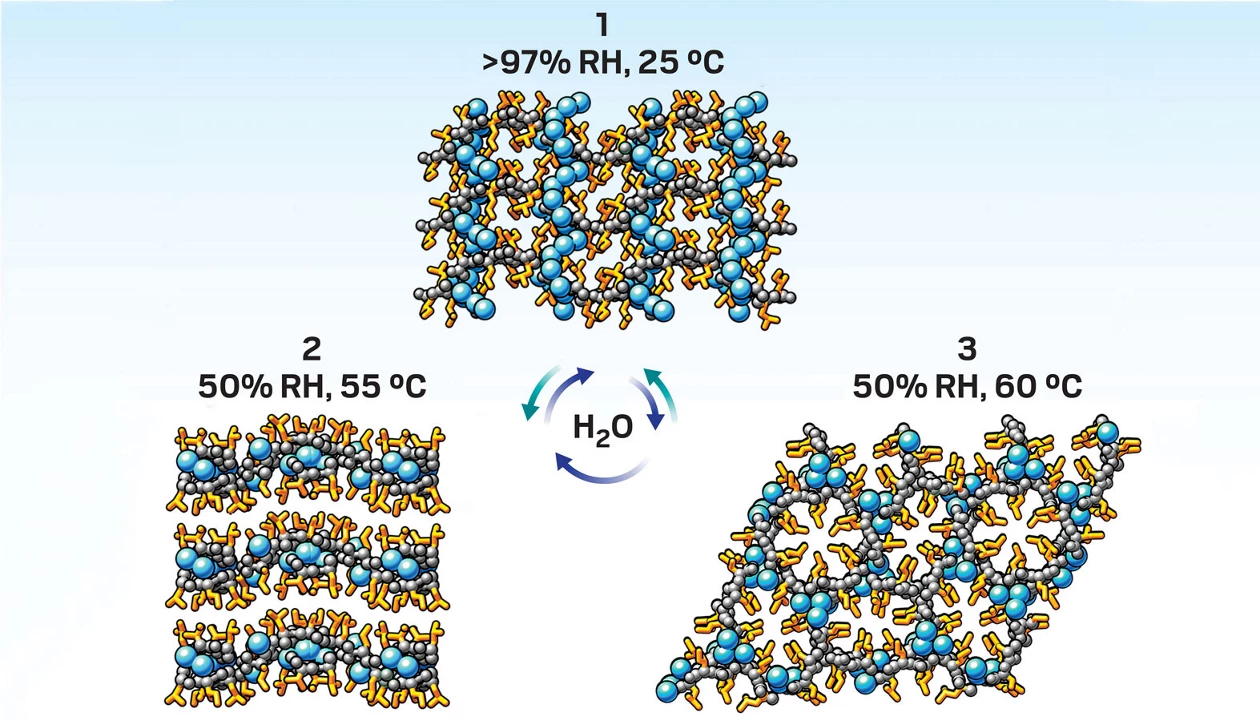
A porous, peptide-based material reconfigures its 3D structure in response to small changes in temperature and humidity (Matter 2026, DOI: 10.1016/j.matt.2026.102669). The different crystal forms exhibit contrasting mechanical properties—most notably shifting from a stiff, hexagonal lattice to a soft, layered structure—potentially opening the door to stimuli-responsive applications in areas such as separation and filtration.
The properties (and therefore function) of any material depend heavily on its structure. Biology has mastered the art of interconverting between closely related forms—such as open and closed ion channels—but human engineering still lags somewhat. “Most human-made materials are static, and there are not very many examples that can reversibly respond in the solid state,” says Rein Ulijn, a bionanoscientist at the City University of New York (CUNY).
Inspired by leucine zippers—structural motifs that zip proteins together—Ulijn and mechanical engineer Xi Chen, also at CUNY, tried to replicate this kind of dynamic behavior. They created four leucine-isoleucine dipeptides, then combined them in solution. By slowly removing the excess water, the pair induced the dipeptides to form a solid, porous crystal hydrate with a stiff, 3D honeycomb structure. “The driver of the assembly is the hydrophobic effect,” Ulijn says. “Leucine and isoleucine are among the two most hydrophobic amino acids. So when you put these in water, they very quickly aggregate together to exclude water, and then they reorganize into a hydrogen-bonded network.”
Crucially, the small amount of water confined within this network dictates the orientation of the conformationally flexible amino acid side chains. In the initial honeycomb structure, the leucine and isoleucine side chains create a hydrophobic column, and a corresponding hydrophilic, water-filled channel at 90° to the hexagonal lattice. But gently removing water (by heating it to 60 °C and reducing humidity to 50%) triggers a rotation in these peptide side chains; that causes the hydrophobic columns to align in parallel to the water-filled channels while retaining the honeycomb structure. Further water loss at 70 °C and 50% humidity triggers a more substantial reorganization to a softer structure in which the side chains form a hydrophobic cavity that separates the layers of dipeptide.
The water not only maintains the structure of the material “but also acts as the fuel to drive the phase transition,” Chen says. “The leucine-isoleucine crystals have multiple, very shallow energy landscapes. Changing the temperature and humidity allows us to change the chemical potential and access those different energy landscapes and their corresponding distinct phases.”
The low energy barrier and mild conditions make each transition completely reversible, and the team successfully interconverted between the stiff honeycomb and soft layer structures over multiple cycles. Real-world applications for this material are still a long way down the line, but Chen and Ulijn think the concept could be useful in barrier and coating materials—particularly in products, such as food packaging, where there’s a need to restrict humidity.
In the meantime, the team remains focused on developing deeper insight into the fundamental principles behind their dynamic material. “We would like to understand better how we can design and optimize these systems further, and ultimately rationally create architectures that can switch in predictable ways,” Ulijn says.
Chemical & Engineering News
ISSN 0009-2347
Copyright ©
2026 American Chemical Society