The Sun washes our planet in a staggering torrent of power. It beams down more energy in a single hour than human civilization consumes in an entire year. But here’s the problem: the Earth stubbornly turns.
Once darkness falls, traditional solar panels snooze. The temperature drops, the power grid strains, and the gap between when we harvest renewable energy and when we actually need it yawns wide open.
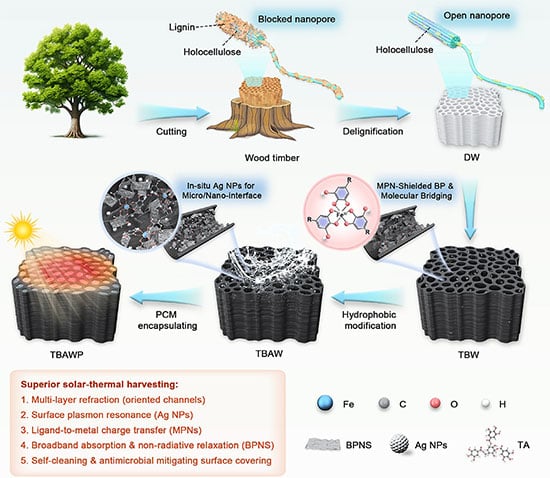
To bridge the gap, scientists in China did something remarkable. They have stripped ordinary balsa wood down to its cellular scaffolding, laced it with chemical armor, and transformed it into a special microscopic sponge. This new material captures sunlight, stores it as heat, and bleeds it out as electricity long after the Sun goes down.
The Daylight Dilemma
We usually solve the nighttime renewable energy problem with brute force. Power companies route excess daytime electricity into massive, expensive battery banks, releasing it when the evening demand spikes.
Alternatively, scientists try to capture heat directly. Standard solar panels convert light to electricity with a hard ceiling of about 25 percent efficiency. But photothermal conversion — trapping light as pure heat — can harvest more than 90 percent of incoming solar radiation.
Engineers harvest this heat using the Seebeck effect. If you place a device called a thermoelectric generator between a hot surface and a cold space, the temperature difference drives an electrical current. But the moment a cloud blocks the Sun, the surface cools, the temperature gap vanishes, and the electricity stops flowing.
Traditionally, engineers tried to solve this nighttime power gap by stacking different materials together. They would embed phase-change materials — like naturally derived waxes — under a dark surface. These waxes act as thermal batteries. They melt in the hot sun to swallow huge amounts of energy, and then slowly solidify in the cold to release that trapped heat, keeping the generator running.
But this stacking method can lead to as many new problems as it solves. Heat struggles to cross the physical borders between separate layers. Furthermore, scientists usually rely on high-temperature carbonization — literally baking materials black — to make them absorb light. This harsh burning process destroys the chemical hooks needed to make the materials waterproof or fire-resistant.
×
Thank you! One more thing…
Please check your inbox and confirm your subscription.
The researchers in China behind the new study realized they needed a completely different approach, abandoning the stacked sandwich entirely.
Stripping the Timber
 Design of interface-engineered wood-based composite phase change materials (CPCMs) for solar-thermal energy conversion. Credit: Advanced Energy Materials, 2026.
Design of interface-engineered wood-based composite phase change materials (CPCMs) for solar-thermal energy conversion. Credit: Advanced Energy Materials, 2026.
The researchers behind the new study, published in Advanced Energy Materials, abandoned carbonization entirely. Instead, they sought a single, unified scaffold that could hold the phase-change material, absorb light, and survive the elements.
They chose balsa wood. Balsa grows exceptionally fast and features microscopic, vertical channels that act like perfectly aligned plumbing.
First, the team gave the wood a chemical bath to strip away its lignin. Lignin is the tough, complex molecule that gives trees their rigid spine. Removing it expanded the wood’s internal channels, boosting its porosity to over 93 percent. It left behind a microscopic structure rich in reactive chemical hooks, ready for modification.
But this created an immediate problem you might already be wondering about. Delignified wood is completely white. White surfaces reflect sunlight, making them useless for solar absorption.
To turn the wood black, the researchers coated the inside of the cellular channels with black phosphorene. This ultrathin nanomaterial absorbs light across the entire spectrum and conducts heat beautifully.
A Molecular Suit of Armor
Here, a new obstacle arose. Black phosphorene breaks down rapidly when exposed to oxygen in the air.
To save the phosphorene, the scientists built a microscopic shield. They wrapped each nanosheet in a metal-polyphenol network made from tannic acid and iron ions. This chemical armor proved remarkably tough. After 150 days of intense solar exposure, the protected nanosheets remained perfectly intact.
Next, they grew tiny silver nanoparticles directly on the coating. These metallic flecks grab onto visible light, boosting the material’s ability to heat up.
Then, the researchers had to address the elements. Raw wood absorbs water like a sponge. In fact, the bare wood framework absorbed almost 200 times its mass in water during testing.
The team grafted long chains of hydrogen and carbon — octadecyl molecules — onto the wood’s surface. This chemical layer forced water to bead up and roll off. The new wood repelled water with an extreme contact angle of 153 degrees, surviving boiling water, mechanical scrubbing, and harsh solvents.
With the scaffold built, blackened, and waterproofed, the scientists filled the microscopic channels with stearic acid.
Stearic acid is a fatty acid derived from nature. It acts as the phase-change material. When the sun hits the black phosphorene, the wood heats up. The stearic acid melts, locking away vast amounts of thermal energy.
When the sun sets, the surrounding air cools. The liquid acid slowly freezes back into a solid. As it changes phase, it exhales the heat it trapped during the day.
Because the team engineered everything chemically rather than physically jamming layers together, the heat flows seamlessly. The modified wood conducts heat nearly four times more efficiently than pure stearic acid alone. Furthermore, the waterproof coating keeps the liquid acid locked firmly inside. Even at 80 degrees Celsius, the liquid never leaked out of the channels.
Trial by Fire and Water
Under simulated sunlight, the black wood captured and stored 91.2 percent of the solar energy that hit it.
When the lights went out, the releasing heat drove a thermoelectric generator, producing a steady 0.65 volts of electricity — enough to run a small fan in total darkness. The material endured 100 heating and cooling cycles with no drop in performance.
But what happens if this heat-storing block of wood catches fire? The researchers anticipated this. Phosphorene naturally retards flames. When scientists held a flame to the wood, it burned briefly but self-extinguished within two minutes, leaving behind a stable charred layer instead of burning to ash.
Finally, the silver nanoparticles served a dual purpose. They killed off common bacteria like E. coli and S. aureus, preventing the microbial rot that usually destroys natural wood left outdoors.
“Our work presents a scalable and environmentally friendly wood-based platform for advanced solar thermal energy harvesting,” the researchers noted in the study.


