New research shows that lipid nanoparticles used in mRNA vaccines may deliver RNA more effectively when their structure is disorganised.

The tiny fatty capsules that carried Covid-19 mRNA vaccines into billions of people may work better when they are a little disorganised, according to new research from scientists at the University of Copenhagen.
Challenging long-held assumptions about drug delivery, the study found that lipid nanoparticles (LNPs), the microscopic bubbles of fat used to ferry fragile strands of RNA into cells, can be more effective when their internal structure is less orderly.
Rethinking a vital vaccine technology
LNPs were central to the success of mRNA Covid-19 vaccines and are now being adapted to deliver treatments for cancer, genetic diseases and other conditions. By encasing delicate RNA molecules in a protective fatty shell, they allow the genetic instructions to enter cells safely.
By encasing delicate RNA molecules in a protective fatty shell, they allow the genetic instructions to enter cells safely.
However, their performance is not perfect, with researchers estimating that only around 1 to 5 percent of the RNA cargo packed inside LNPs is successfully released within cells.
“This low efficiency limits what we can do with LNPs as therapeutics,” said Artu Breuer, a researcher at the University of Copenhagen. “For example, in cancer treatment where cells are dividing rapidly, if you deliver too little RNA, the cells outpace the therapy.”
Looking at one particle at a time
To investigate why delivery efficiency varies so widely, Breuer and colleagues developed a high-throughput method capable of analysing nanoparticles individually rather than averaging measurements across an entire batch. Their technique can assess around a million particles at a time, measuring both the size of each nanoparticle and the amount of RNA it contains.
“Instead of assuming that every nanoparticle in a batch is the same, we found enormous variation,” Breuer said. “And we discovered two distinct subpopulations: organised particles where the cargo is neatly structured, and amorphous particles where it’s more disorganised. The surprise was that the messy ones actually work better inside cells.”
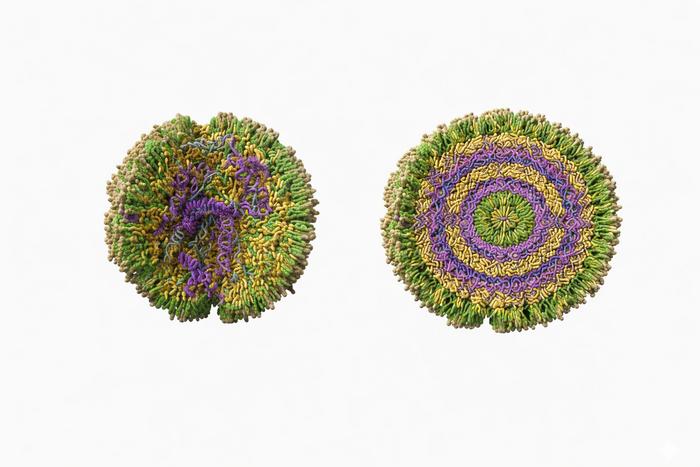
Translational science Organised lipid nanoparticles in which the cargo is neatly structured are shown on the right, and amorphous lipid nanoparticles where cargo is more disorganised are shown on the left. Surprisingly, researchers discovered that the more disorganised lipid nanoparticles delivered drugs more effectively. Credit: Artu Breuer
Why disorder helps
The discovery runs counter to the prevailing strategy in drug development, which has prioritised loading as much medicine as possible into each nanoparticle and arranging it in a highly ordered way.
Breuer explained that some nanoparticles resemble the layers of an onion, with tightly organised structures formed by positively charged lipids bound closely to negatively charged RNA. While this tight packing may appear efficient, it can hinder the release of the therapeutic payload once inside the cell.
“Think of it this way: in an organised nanoparticle, the positively charged lipids are tightly bound to the negatively charged RNA,” Breuer said. “When the particle enters a cell, even though conditions change, those attractions hold everything together. But in a disorganised particle, there’s some separation between the charges. When conditions change inside the cell, the positive charges repel each other, and the particle falls apart – releasing the medicine.”
A potential shift
The findings suggest that future nanoparticle design may need to focus less on maximising cargo and more on maintaining a loosely arranged internal structure that allows RNA to escape at the right moment.
By enabling researchers to screen formulations at the level of single nanoparticles, the new measurement tool could help accelerate the development of more effective RNA-based medicines.
“We’re aiming in the opposite direction of what the field has been pursuing,” Breuer said. “I’m not saying we should have empty nanoparticles, but we need to find ways to load enough RNA while still keeping that disorganised structure that’s more effective inside cells.”
By enabling researchers to screen formulations at the level of single nanoparticles, the new measurement tool could help accelerate the development of more effective RNA-based medicines, potentially changing how treatments for cancer and genetic disorders are engineered.

