Study findings offer potential to accelerate development of RNA-based medicines.
New research suggests that reducing the amount of cargo inside lipid nanoparticles (LNPs) could improve the efficiency of drugs.
This is notable considering LNPs have “low efficiency limits”, with one to five percent of their cargo typically released inside cells, according to Artu Breuer, a researcher at the University of Copenhagen.
“For example, in cancer treatment where cells are dividing rapidly, if you deliver too little RNA, the cells outpace the therapy.”
To overcome this challenge, Breuer et al. developed a high-throughput, single-nanoparticle measurement tool that could provide a way to develop more effective RNA-based medicines.
Instead of observing the average properties of a batch, the team measured both the size of each particle and how much cargo it contained.
[the researchers found] two distinct subpopulations: organised particles where the cargo is neatly structured, and amorphous particles where it’s more disorganised. The surprise was that the messy ones actually work better inside cells”
Breuer explained that “instead of assuming that every nanoparticle in a batch is the same, we found enormous variation”.
Importantly, the researchers found “two distinct subpopulations: organised particles where the cargo is neatly structured, and amorphous particles where it’s more disorganised. The surprise was that the messy ones actually work better inside cells”.
Breuer added: “In an organised nanoparticle, the positively charged lipids are tightly bound to the negatively charged RNA.
“When the particle enters a cell, even though conditions change, those attractions hold everything together. But in a disorganised particle, there’s some separation between the charges. When conditions change inside the cell, the positive charges repel each other, and the particle falls apart – releasing the medicine.”
This is counter to traditional drug development processes whereby each nanoparticle is loaded with as much medicine as possible as efficiently as possible.
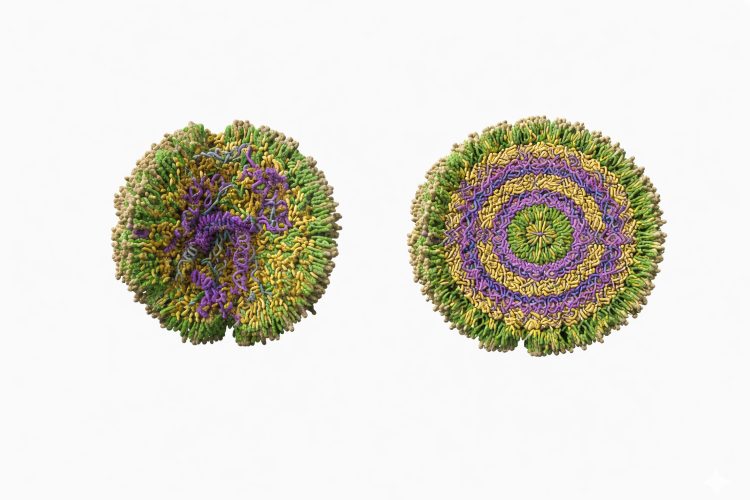
Organised lipid nanoparticles in which the cargo is neatly structured (right), and amorphous lipid nanoparticles where cargo is more disorganised (left). Credit: Artu Breuer, University of Copenhagen.
This finding suggests it is better to establish a disorganised internal structure to enable LNP cargo to be released once at its destination, instead of maximising how much cargo each particle carries.
However, Breuer cautioned this does not imply keeping nanoparticles empty, but should encourage research avenues that focus on loading “enough RNA while still keeping that disorganised structure that’s more effective inside cells”.
Screening LNP formulations with this method could help researchers understand which structural features are critical for delivery.
The research was presented at the 2026 Biophysical Society Annual Meeting.
