Scientists have discovered how an imbalance in gut bacteria can accelerate chronic kidney disease, with early findings pointing to a potential drug that could interrupt the damaging cycle and improve outcomes.
Researchers have identified a harmful cycle in the gut that may accelerate chronic kidney disease. The new study could potentially bring about new treatments that interrupt the process and slow the illness.
The researchers from the University of California, Davis School of Medicine found that an imbalance in gut bacteria can drive the production of toxic compounds that worsen kidney damage and highlighted a potential drug that could break this cycle.
How gut imbalance worsens kidney disease
The research team discovered that impaired kidney function increases nitrate levels in the colon. These nitrates stimulate Escherichia coli (E. coli), to produce higher levels of indole. This compound is then converted into indoxyl sulphate, a toxic waste product that further damages the kidneys.
The research team discovered that impaired kidney function increases nitrate levels in the colon.
This creates a self-perpetuating feedback loop, where kidney damage fuels gut changes that then accelerate further decline.
“Previous research has shown that chronic kidney disease is linked to an elevated faecal abundance of Enterobacteriaceae,” said Jee-Yon Lee, first author of the study and a project scientist in the Department of Medical Microbiology and Immunology. “This study identifies nitrate from the host as a switch that turns common gut bacteria like E. coli into indole producers capable of accelerating chronic kidney disease.”
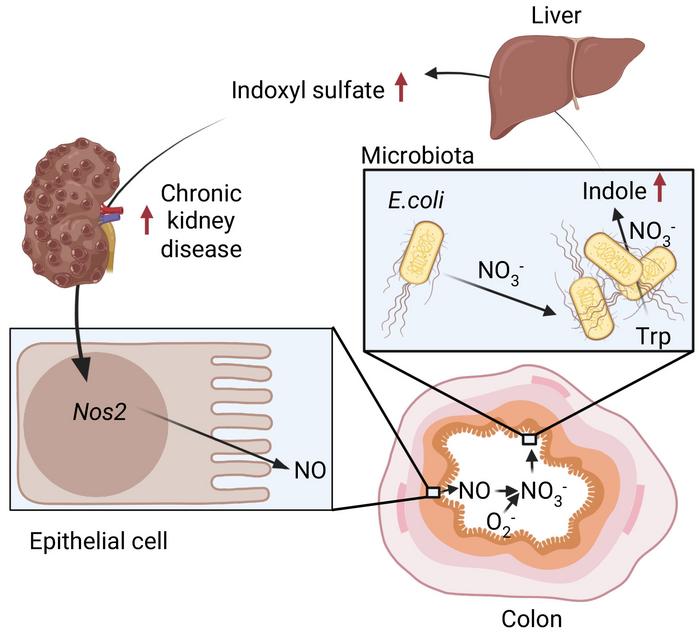
A biological diagram illustrating how chronic kidney disease turbocharges E. coli in the gut to produce indole, which is converted to indoxyl sulphate (a kidney toxin) in the liver, further worsening kidney disease. Credit: UC Regents.
A widespread health challenge
Chronic kidney disease is a progressive condition affecting around one in seven adults in the United States, or an estimated 35.5 million people. It is particularly common among those with diabetes and high blood pressure. Globally, around 788 million people were estimated to be living with the condition in 2023.
Although treatments such as haemodialysis can remove many toxins from the blood, indoxyl sulphate is difficult to eliminate because it binds tightly to proteins. Higher levels of this compound are associated with more severe disease.
Targeting a key enzyme
The study identified a potential way to interrupt this damaging cycle by blocking a single enzyme in the gut known as inducible nitric oxide synthase (iNOS).
Experiments in mice showed that kidney dysfunction increased activity of the gene responsible for producing iNOS.
“By identifying the driver responsible for an increase of Enterobacteriaceae during chronic kidney disease and by demonstrating the importance of these bacteria for indole production and disease progression, our research points to iNOS as a potential target for intervention strategies,” said Andreas Bäumler, senior author of the study.
Experiments in mice showed that kidney dysfunction increased activity of the gene responsible for producing iNOS. This led to higher nitric oxide levels which then formed nitrate and fuelled bacterial growth and toxin production.
Promising early results
To test a possible treatment, researchers used aminoguanidine, an investigational drug that inhibits iNOS. Mice given the drug showed lower nitrate levels in the gut, reduced indoxyl sulphate and improved kidney health.
Human stool samples showed similar patterns to those seen in mice. However, increased indole production only occurred when nitrate was present.
Caution and next steps
Despite the promising findings, researchers say further work is needed. Clinical trials will be required to determine whether targeting iNOS is safe and effective in people with chronic kidney disease.
Clinical trials will be required to determine whether targeting iNOS is safe and effective in people with chronic kidney disease.
They also caution that the gut microbiome is highly complex, and E. coli is not the only bacterium capable of producing indole. Long-term suppression of nitrate pathways may have unintended consequences.
“This study shows that altering the gut environment – not just the microbes themselves – can have profound effects on disease progression,” Bäumler said. “Targeting host pathways that shape microbial metabolism may represent a new way to intervene in chronic kidney disease.”