Colombia announced the approval of the of Benznidazole 100 mg tablets for timely treatment of Chagas disease and the prevention of serious complications, especially in vulnerable populations and territories at risk.
The decision marks a step forward in public health, facilitating early treatment of affected individuals, reducing the progression of the disease, and helping to prevent chronic complications, mainly cardiac and digestive, associated with this parasitic infection.
“ Timely access to this medication allows us to act promptly, prevent serious complications, and protect the lives of thousands of people, especially in the most vulnerable regions of the country. This is an important step toward closing health gaps and advancing the control of Chagas disease in Colombia ,” stated the Minister of Health and Social Protection, Guillermo Alfonso Jaramillo.
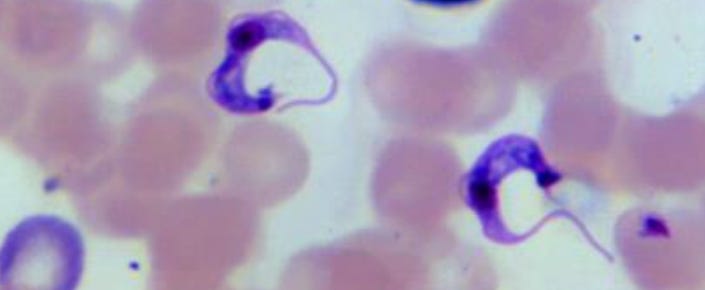
Benznidazole is the primary treatment for Chagas disease, caused by the parasite Trypanosoma cruzi. Its availability in the country strengthens the health system’s capacity to respond more effectively to this highly relevant public health event, which continues to affect communities in different regions of the country.
This achievement is the result of collaboration between the Ministry of Health and Social Protection and the Ministry of Science, Technology and Innovation, through funding from the Health Research Fund (FIS), managed by the University of Antioquia. This partnership reinforces the role of research and technological development in addressing priority health problems.
In addition to expanding access to treatment, the approval of Benznidazole boosts domestic production of strategic medicines, strengthens the country’s autonomy in health, and helps ensure greater availability for those who need it.