Scientists have identified a previously unknown route by which Huntington’s disease spreads from one brain cell to the next.
By exposing the machinery behind that transfer, the finding points to a way of slowing the disease before wider damage takes hold.
Identifying the SLC4A7 protein link

Huntington’s disease is an inherited brain disorder that gradually damages nerve cells and disrupts movement, thinking, and behavior.
In the striatum, the brain region most affected, a toxic form of the huntingtin protein, an altered version that harms cells, moved between neurons through tiny physical bridges.
Following that path, Srinivasa Subramaniam, Ph.D., at Florida Atlantic University (FAU) connected those bridges to a specific partnership at the cell surface that made them possible.
Rhes, a protein active in that vulnerable region, paired with another protein called SLC4A7 to build the connections that carried the toxic huntingtin protein onward.
That link narrowed the discovery to a defined route and a specific protein pair, raising a deeper question about what else these hidden connections might be doing.
What the tubes do
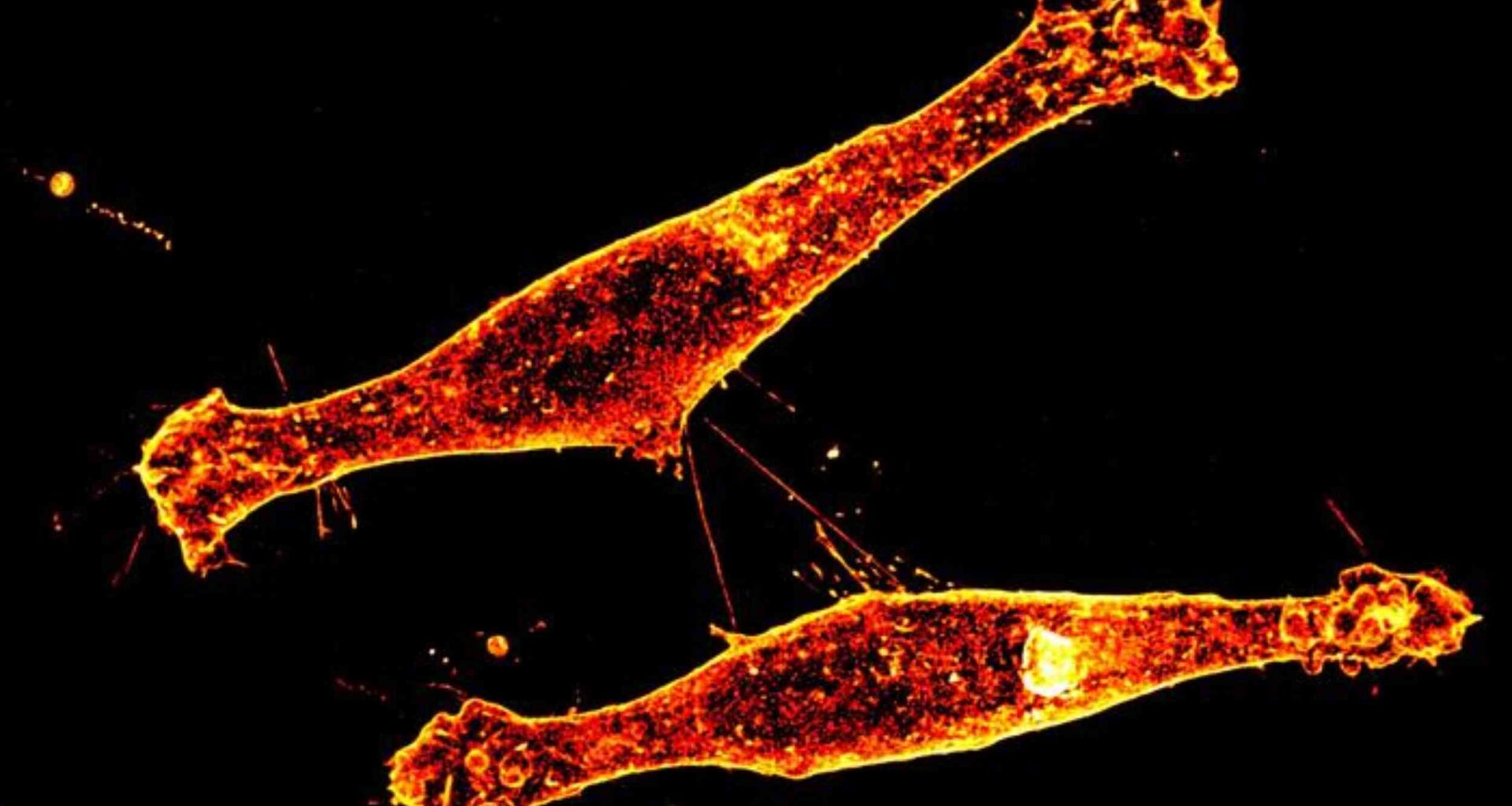
Researchers call the structures tunneling nanotubes, thin membrane bridges that let cells pass material directly to one another.
Instead of releasing cargo into open space, one neuron can send protein packages through the bridge and into a neighboring cell.
FAU researchers found that this route carried mutant huntingtin, the altered protein that damages nerve cells in Huntington’s disease.
That direct transfer helps explain why damage does not stay isolated, even when only some cells first make the toxic protein.
Blocking SLC4A7 prevents bridges
When researchers blocked SLC4A7, either by altering the gene or using a drug, far fewer of these tiny bridges formed between brain cells.
With those connections reduced, much less of the toxic huntingtin protein moved into neighboring cells, showing the bridges were active pathways for damage.
“We’ve known that neurons somehow pass toxic proteins to one another, but now we can see the machinery that makes that possible,” said Subramaniam.
That shift turns a once-hidden process into something scientists can now target more directly with potential treatments.
Evidence in mice
The clearest proof came from mice that lacked SLC4A7, where the toxic protein spread far less through the brain.
In the striatum, a deep region that helps control movement, the harmful protein stayed closer to where it first appeared.
Other signals in the brain remained unchanged, which showed the effect was not due to weaker delivery or fewer surviving cells.
Keeping the damage contained in this region matters, because early symptoms of Huntington’s disease begin there and worsen over time.
SLC4A7 and chemical balance
SLC4A7 stood out because it helps control the chemical balance inside cells, especially how acidic or stable they are.
At the cell surface, Rhes changed that balance in a way that made it easier for the tiny bridges to form.
When SLC4A7 was blocked, that chemical change faded, even though the two proteins could still connect.
That difference suggests scientists may be able to slow the spread of damage without fully breaking apart the proteins themselves.
Anchoring the culprit
Another clue came from how Rhes attaches to the cell surface, using a small chemical tag that holds it in place.
When researchers blocked that attachment, Rhes could no longer link up with SLC4A7, and the bridges mostly disappeared.
SLC4A7 itself stayed in position, showing the effect was specific to Rhes rather than disrupting the entire cell surface.
That finding sharpened the case against Rhes itself, placing its membrane attachment near the start of the harmful chain.
Why families care
For families living with Huntington’s disease, the appeal is obvious because no treatment stops the disease from worsening.
If one parent carries the gene, each child faces a 50 percent chance of inheriting it.
Symptoms usually start in adulthood, and many people die within 15 to 20 years after signs first appear.
A therapy that slows spread between cells would not erase the cause, but it could buy precious time.
Beyond one disorder
Similar cell-to-cell bridges have also appeared in studies of tau disorders and several forms of cancer.
Other experiments have linked these structures to the movement of misfolded proteins, survival signals, and drug resistance.
That broader pattern suggests this Huntington’s pathway may belong to a larger biology of disease spread, not an isolated quirk.
Any future drug would still need careful tuning, because healthy cells sometimes use these contacts during stress or repair.
Therapy and caution
The work points to a drug target, but it does not yet prove that blocking this pathway will help people.
“By identifying SLC4A7 as a key partner of Rhes, we’ve uncovered a new and potentially druggable target to stop that spread at its source,” said Subramaniam.
Even so, SLC4A7 works in many tissues, which means any treatment must avoid disrupting normal chemistry elsewhere.
Researchers now need compounds that reach the brain safely and show the same effect in longer, harder tests.
What changes now
FAU researchers have pinned Huntington’s spread to a physical route between neurons and a specific protein pair.
Closing that route will take years of work, yet the study finally shows where intervention can start.The study is published in Science Advances.
—–
Like what you read? Subscribe to our newsletter for engaging articles, exclusive content, and the latest updates.
Check us out on EarthSnap, a free app brought to you by Eric Ralls and Earth.com.
—–