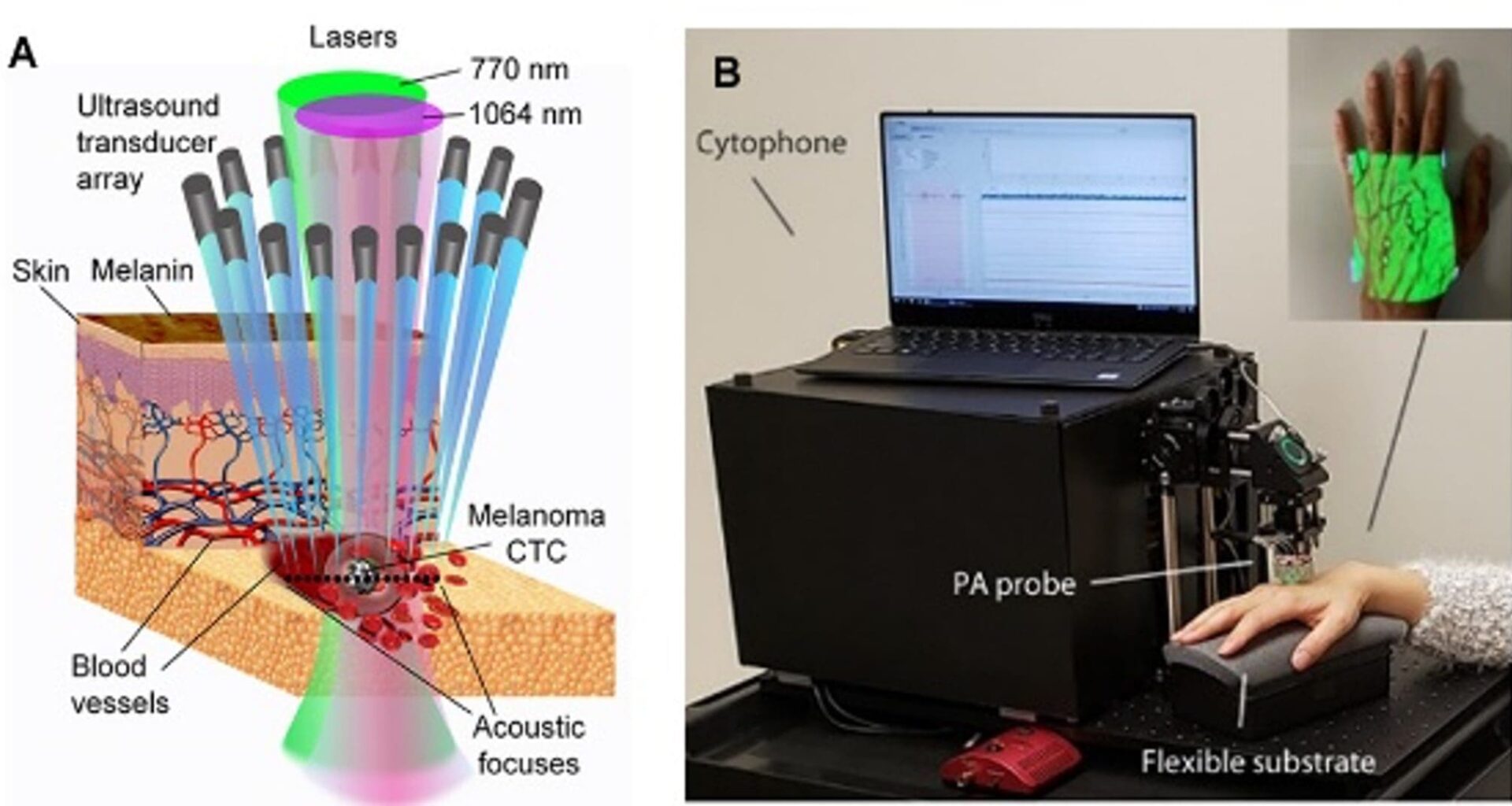
Professor Vladimir Zharov, PhD, DSc, CytoAstra CSO, introduces a clinically proven revolutionary early non-invasive diagnosis of cancer, infections, stroke and infarction by laser assessing the entire blood volume in vivo directly into vessels through intact skin, without the need for blood draw and needle
To date, little progress has been made in diagnosing deadly diseases at an early stage, when treatment is most effective. For example, most deaths (up to 90%) in cancer patients are related to metastatic disease as a result of circulating tumor cells (CTCs) spreading. Another example is malaria, which for centuries has been one of the deadliest diseases on the planet. Nearly half of the world remains at risk of malaria, with more than half a million deaths each year, most of them in children. A final example is thromboembolic complications (e.g., stroke, heart attack, and pulmonary embolism), which are one of the top causes of death in the world.
Diagnosis of these and many other diseases begins with a common medical procedure: the examination of a patient’s blood sample to identify abnormal healthy cell counts or abnormal cells such as CTCs, infected red blood cells (iRBCs), and clots. However, existing blood tests cannot provide early detection of indicated cells and other biomarkers because their sensitivity is limited by the small blood volume extracted (from 10 µL to 10 ml), missing up to half million (for 10 µL sample) of abnormal cells or biomarkers (e.g., CTCs, iRBCs, clots, bacteria, virus and DNA) in the entire blood volume (4-6 litres [L] in an adult); this number of missed cells is sufficient for disease to progress to a stage that is barely treatable or is already incurable (e.g., metastasis, sepsis, or stroke).
Concept of a new non-invasive blood test in vivo
The rationale for a new diagnosis is straightforward: most organs and tissues, and potentially many cells in the body, can signal their pathological status by shedding cells and their fragments into the bloodstream. However, their detection at an early stage, given their typically low abundance, poses many technical challenges for existing methods, such as low sensitivity, low resolution, slow response, and limited efficiency in capturing fast-moving cells with static sensors. Zharov’s team overcame these challenges by introducing a revolutionary concept for the examination of the entire blood volume in the human body using the photoacoustic (PA) flow cytometry (PAFC) platform, called Cytophone. (1-4)
In PAFC, transcutaneous delivery of laser light through intact skin to deep vessels generates the PA effect in circulating biomarkers transforming light into sound (acoustic) waves via the optical absorption and thermal expansion. Acoustic waves are detected with an ultrasound transducer array attached to the skin above the vessels (Fig. 1A, B). When the biomarker passes through the detection volume, local absorption rapidly increases, resulting in a sharp CTC-related peak above the blood background. Many (up to 16) tiny, focused transducers are oriented in the line across the vessel; as a result, each biomarker can simultaneously produce many temporally coincident signals from different transducers (Fig.2). A rainbow Cytophone with multicolour tiny laser diodes provides spectral identification of biomarkers with different spectral fingerprints using innovative time-color coding. (5) Cytophone software provides high-speed time-resolved signal processing with a real-time identification of biomarker origin using specific AI-assisted criteria. What’s more, Cytophone provides a label-free detection of biomarkers with intrinsic absorption (e.g., melanin in melanoma CTCs, hemozoin in malaria-infected RBCs, and abnormal haemoglobin in sickle cells). Low- absorbing biomarkers can be targeted with conjugated labels that provide high PA contrast (e.g., gold and magnetic nanoparticles) directly in the bloodstream. (6) The biomarker identification can be significantly enhanced by using ultrasharp spectral resonances with ‘Zharov splitting’ phenomena, providing doubling colors. (7-8)
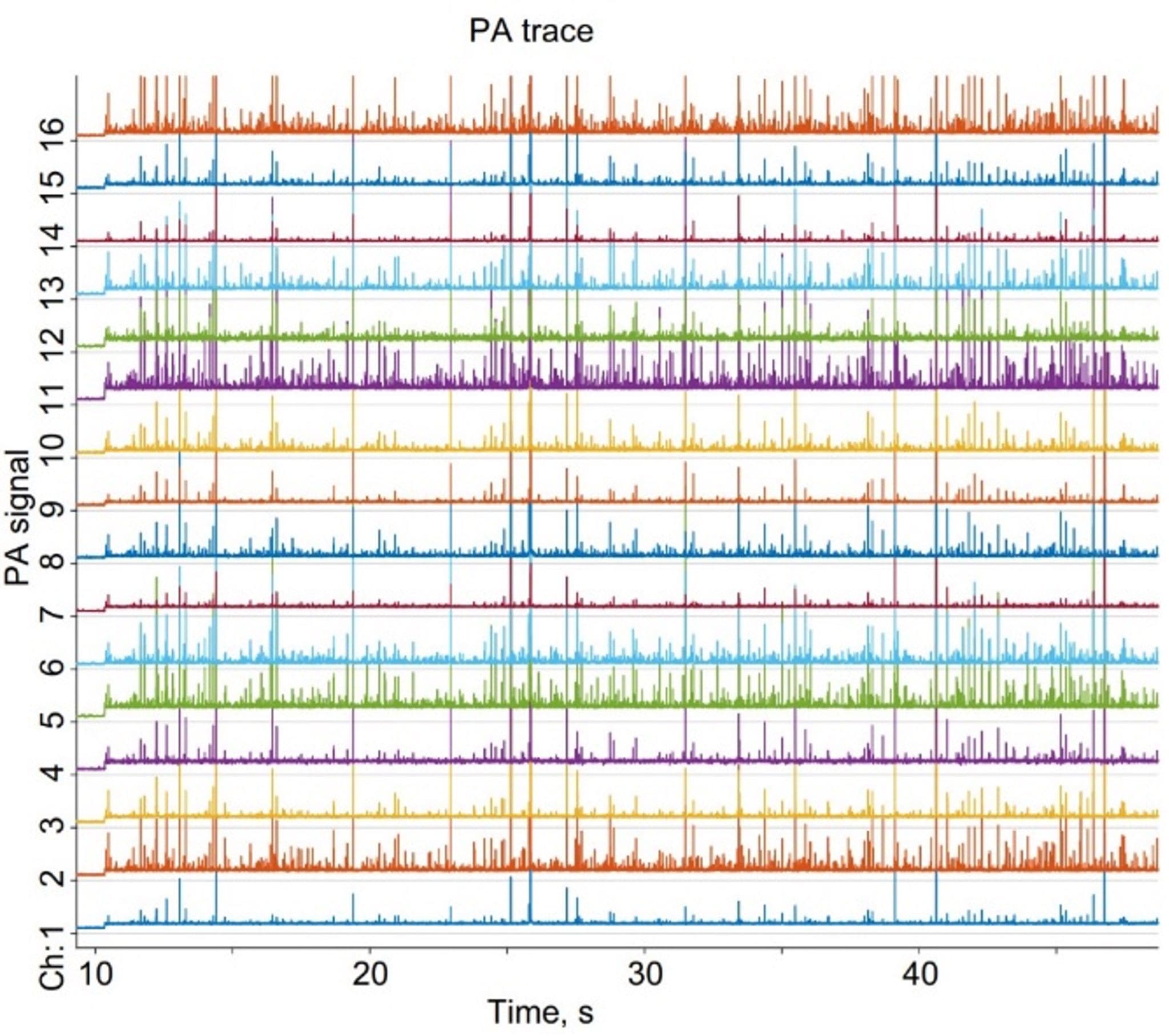 Figure 2. Multiple PA traces from 16 ultrasound focused transducers.
Figure 2. Multiple PA traces from 16 ultrasound focused transducers.
Pre-clinical and clinical validation of new diagnostic platform
The concept of a new diagnostic platform was comprehensively validated in vitro using multiple animal and human cell lines and in vivo on different animal models (e.g., mouse, rats, and rabbits), resulting in approximately 100 publications, including 17 papers in Nature and Science family journals. (9) A total of 60 national and international patents were issued in different countries, including the US and European Union.
In preclinical studies, versatile Cytophone demonstrated the applicability for non-invasive diagnosis of in vivo melanoma, malaria, stroke, bacteremia, sickle anemia, thrombosis, and abnormal hemoglobin forms, (10) and guidance of in vivo physical destruction of melanoma CTCs, and malaria-infected iRBCs directly in the bloodstream by laser-induced nanobubbles around overheated melanin and hemozoin clusters, respectively. (3,11)
Six clinical PAFC/Cytophone devices with different schematics and designs were developed, and the FDA determined PAFC to be a non-significant risk (NSR) medical device under the Investigational Device Exemption (IDE) regulation. Seven clinical trials were organized in the US and Africa (four complete, and three ongoing). To date, almost five hundred subjects have been clinically tested, including healthy controls and patients, with three to four visits per subject.
In vivo detection of CTCs in melanoma patients
Up to 50% of melanoma deaths are linked to late diagnosis of already incurable metastasis, even with new therapies (e.g., immune-checkpoint inhibitors) caused by CTCs. Some melanomas in very early stages can metastasize significantly over several months. There are no FDA-approved methods for detecting melanoma CTCs, and existing research assays fail to detect CTCs at early stages due to poor detection limits (~1 CTC/mL) and limited blood sample volume (e.g., 7.5 mL for CellSearch, Menarini Silicon Biosystems). As an alternative, the use of circulating tumor DNA (ctDNA) or other molecular biomarkers is not well validated for early stages because these biomarkers derive from apoptotic and necrotic cancer cells, which are extremely rare at the earliest stages of tumor development.
By examining a large blood volume in a short time (up to 1 L in 15-30 minutes, depending on vessel parameters), Cytophone demonstrated a ~1,000-fold threshold improvement (down to 1 CTC/L) in melanoma patients. (3) No free melanin was found in normal blood. The ongoing study revealed CTC detection at all melanoma stages, especially at stages 0 (in situ) and I, suggesting parallel progression of the primary tumor and CTCs. These clinical discoveries open a door for early non-invasive, label-free, rapid, tolerant to skin pigmentation and motion diagnosis of melanoma and potential screening through detection, identification, and automatic counting of CTCs, even from small (≤ a few mm) tumors, in many cases not diagnosed yet with conventional biopsy and radiology methods.
In vivo detection of infected RBCs (iRBCs) in malaria patients
Malaria, caused by parasites transmitted to the blood through mosquito bites, has been one of the deadliest diseases in the world. Since 2000, 2.3 billion malaria cases and 14 million deaths have been averted worldwide. While some progress has been made in controlling malaria and developing a vaccine, this has stalled recently, with a growing number of deaths since 2019. At the heart of the challenge is the lack of a sensitive, rapid diagnostic test for malaria. Zharov’s team provided the Cytophone to detect hemozoin, a pigment produced by malaria parasites during hemoglobin digestion in iRBCs in Africa. This work was performed through collaboration with Prof. Sunil Parikh at Yale School of Public Health, Prof. Yap Boom from Doctors Without Borders/Médecins Sans Frontières (MSF) and Prof. Matthew Laurens from the University of Maryland School of Medicine. Like melanin, hemozoin strongly absorbs laser light and generates acoustic waves that can be detected with the ultrasound transducers. Four clinical trials (2020, Cameroon; 2021, US; 2024, Cameroon; and 2025, Burkina-Faso, including children) successfully demonstrated the advantages of label-free Cytophone in terms of sensitivity and time-to-response without blood drops and in monitoring drug efficacy over time, all without needles or laboratory work. (4)
In vivo detection of circulating blood clots (CBCs) in stroke patients
CBCs are common complications of surgery, trauma, catheterization, implants, extracorporeal systems, and the second-leading cause of death among cancer patients. CBCs, also called emboli, block vessels at different locations, leading to venous thromboembolism, pulmonary embolism, stroke, and ischemic infarction.
In preclinical trials, Cytophone demonstrated the ability to identify white (platelet-rich), red (hemoglobin- rich), and mixed CBCs in arteries and veins, with negative and positive PA peaks for white and red CBCs in the blood background, respectively, (12) and to detect CBCs in stroke patients in vivo using handheld PA probes (ongoing trial). This approach can provide real-time monitoring of asymptomatic individuals as the size and number of CBCs increase, until large CBCs become dangerous in circulation. This serves as a sign for well-timed, optimized anticoagulant therapy to prevent thromboembolic complications (e.g., stroke) while minimizing the risk of bleeding. Cytophone can detect CBCs ranging in size from 30-50 µm to a few mm in hand, leg, and neck vessels at a depth of 1–1.5 cm. The Cytophone could be used for personalized monitoring of drug efficacy and surgery-induced CBCs in the operating room and, potentially, at home.
Integration of laser detection and treatment of CTCs, iRBCs, and CBCs
The Cytophone is a powerful, versatile platform applicable for the diagnosis and simultaneous treatment of various diseases. Early detection raises the question of how to treat individuals diagnosed at a very early disease stage, while most treatments apply preferentially to advanced stages. Based on a previous study of the impacts of acoustic and cavitation bubble effects on cellular structures induced by nanosecond and picosecond lasers through absorption and optical breakdown effects, (13) the Zharov team introduced Cytophone-guided, resistance-free, destruction of CTCs, iRBCs, and potentially CBCs directly in the bloodstream. (2,4,9) The goal of new treatment is to prevent disease progression by laser-induced therapeutic nanobubbles around intracellular melanin, hemozoin and clustered hemoglobin, respectively. They pioneered also using conjugated with different ligands (e.g., antibodies) strongly light absorbing plasmonic nanoparticles for targeting of disease biomarkers in abnormal cells in circulation. These nanoparticles alone, and especially during clustering on cell membrane or after endocytosis on intracellular organelles significantly amplify acoustic waves and nanobubble formation improving of diagnostic and killing efficiency. In a preclinical malaria-related study, a 671-nm pulse laser significantly reduced the concertation of iRBCs in the bloodstream. (11) In melanoma patients with strong CTC pigmentation, laser provided highly localized (≤0.1-0.3 µm) nanobubble-based mechanical destruction up to 96% of CTCs without impact on surrounding healthy cells during one hour non-invasive exposure of the cubital vein (3,14) that can potentially reduce the risk of metastasis by 25 times.
In conclusion, the revolutionary development of the Cytophone platform for combined early diagnostics and therapeutics, in which a disease is diagnosed and treated almost simultaneously, has the potential to eradicate melanoma, malaria, stroke, and other serious illnesses. The discovery, with Cytophone, of early release of CTCs in melanoma patients at stage 0 (in situ) and I offers the potential for melanoma screening by detecting CTCs in asymptomatic individuals by gently attaching a PA sensor to the hand skin for ten minutes. This technology is at the stage of developing commercial, portable, and wearable battery-based Cytophones (5) and seeking FDA approval for different applications in rural villages, urban hospitals, diagnostic centers, ambulances, medical helicopters, and potentially at home.
References
Zharov V, Galanzha E, Tuchin V. Photothermal imaging of moving cells in lymph and blood flow in vivo animal model. Proc SPIE 2004; 5320: :256–63.
Galanzha EI, Shashkov EV, Spring PM, Suen JY, Zharov VP. In vivo, noninvasive, label-free detection and eradication of circulating metastatic melanoma cells using two-color photoacoustic flow cytometry with a diode laser. Cancer Res. 2009; 69:7926–34.
Galanzha EI, Menyaev YA, Yadem AC, Sarimollaoglu M, Juratli MA, Nedosekin DA, Foster SR, Jamshidi-Parsian A, Siegel ER, Makhoul I, Hutchins LF, Suen JY, Zharov VP. In vivo liquid biopsy using Cytophone platform for photoacoustic detection of circulating tumor cells in patients with melanoma. Sci Transl Med. 2019; 11 (496).
Yadem AC, Armstrong JN, Sarimollaoglu M, Ndifo J-M, Menyaev YA, Mbe A, Richards K, Wade M, Chen R, Zhou Q, Meten E, Ntone R, Tchuedji YGN, Ullah S, Galanzha EI, Eteki L, Gonsu HK, Biris A, Suen JY, Boum Y, Zharov VP, Parikh S. Noninvasive in vivo photoacoustic detection of malaria with Cytophone in Cameroon. Nat Commun 2024; 15 (9228).
Jawad HJ, Yadem AC, Menyaev YA, Sarimollaoglu M, Armstrong JN, Watanabe F, Biris AS, Parikh SP, Stumhofer JS, Nedosekin DA, Suen JY, Zharov VP. Towards rainbow portable Cytophone with laser diodes for global disease diagnosis. Sci Rep 2022; 12 (8671).
Galanzha EI, Shashkov EV, Kelly T, Kim JW, Yang L, Zharov VP. In vivo magnetic enrichment and multiplex photoacoustic detection of circulating tumour cells. Nat Nanotechnol. 2009; 12:855-60.
Zharov VP. Ultrasharp nonlinear photothermal and photoacoustic resonances and holes beyond the spectral limit. Nat Photonics 2011; 5:110-16.
Mertiri A, Altug H, Mi K Hong MiK, Pankaj Mehta P, Mertz J, Ziegler LD. Nonlinear midinfrared photothermal spectroscopy using Zharov splitting and quantum cascade lasers. ACS Photonics. 2014; 1:696-702.
Galanzha EI, Zharov VP. Circulating tumor cell detection and capture by photoacoustic flow cytometry in vivo and ex vivo. Cancers 2013; 5:1691-1738.
A.V. Brusnikin, Nedosekin DA, Ryndina ES, Proskurnin MA, Gleb EYu, and Zharov VP, Determination of various hemoglobin species with thermal-lens spectrometry. Appl. Spectrosc., 2007; 64:145-54.
Cai C, Carey KA, Nedosekin DA, Menyaev YA, Sarimollaoglu M, Galanzha EI, Stumhofer JS, Zharov VP. In vivo photoacoustic flow cytometry for early malaria diagnosis. Cytometry A 2016; 89:531-42.
Juratli MA, Menyaev YA, Sarimollaoglu M, Melerzanov AV, Nedosekin DA, Culp WC, Suen JY, Galanzha EI, Zharov VP. Noninvasive label-free detection of circulating white and red blood clots in deep vessels with a focused photoacoustic probe. Biomed Opt Express 2018; 9:5667-77.
Beilin E, Buyanov-Uzdalsky AY, Lotchilov VI, Zharov VP. Investigation of laser-induced acoustic effects in water and their influence on cell structure. Akustichesky Zhurnal (J Acoustics in Russian) 1987; 23: 194–8.
Kim JW, Galanzha EI, Zaharoff DA, Griffin RJ, Zharov VP. Nanotheranostics of circulating tumor cells, infections and other pathological features in vivo. Mol Pharm. 2013; 10:813-30.