Researchers at Purdue University have harnessed a protein in blood to assemble conducting polymers, potentially opening the door for better, more compatible polymers to integrate bioelectronics with existing physiology (Science 2026, DOI: 10.1126/science.adu5500).
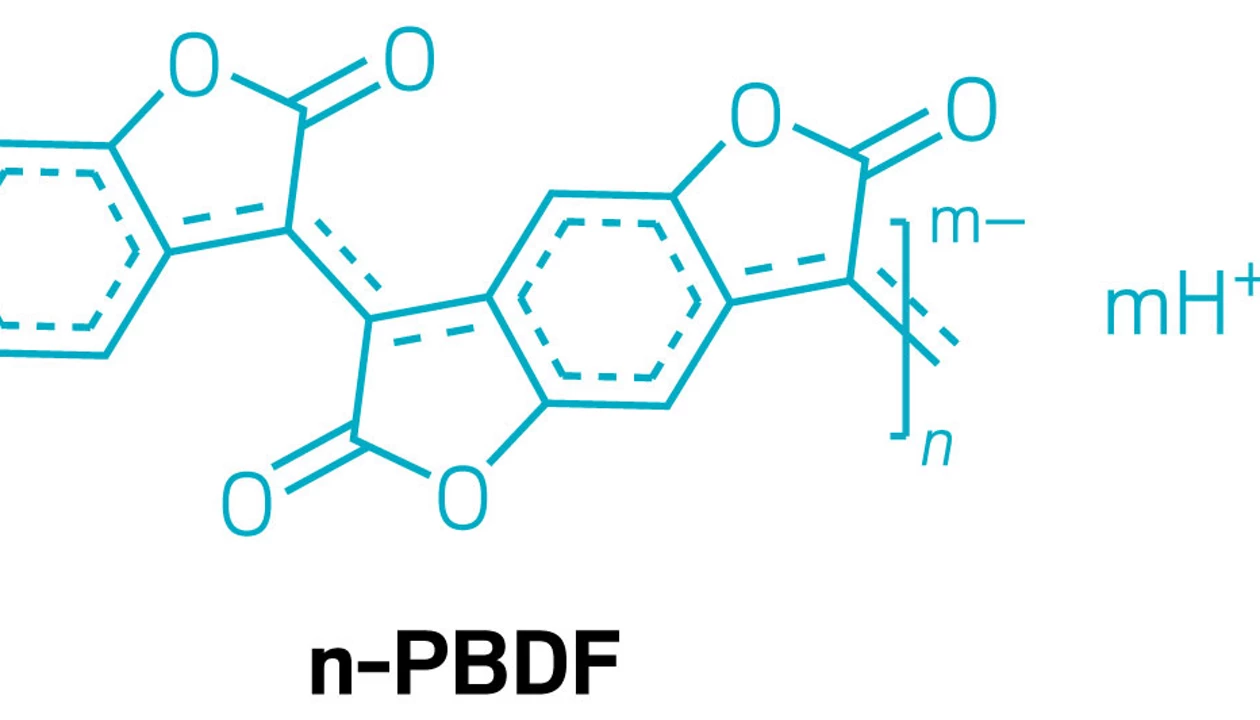
The polymer is made from monomers of n-doped poly(benzodifurandione), or n-PBDF. When n-PBDF is mixed with blood, the iron in heme proteins like hemoglobin and myoglobin catalyzes the reaction to connect the monomers into a polymer.
Jianguo Mei, one of the lead researchers, says that previous work with n-PBDF involved copper salts, but “biologic systems don’t like copper.” However, the body has iron readily available in blood proteins like hemoglobin, which led to this new paper.
Once the team had established that the reaction worked in vivo, it moved to integrate the reaction into existing neuronal structures in mice. This was done by injecting a slightly prepolymerized molecule, along with some whole blood, into the precise area of the brain where the researchers wanted the polymer to form.
The finished polymerization formed a mesh-like structure around the neurons, and the mice with the polymers behaved normally. The researchers saw no adverse effects like neuronal degradation or the polymers drifting in the brain. “This innovation is the first step toward what we would like to term as living electronics, which is that you can actually have electronics synthesized inside the body, inside the brain,” says Krishna Jayant, another member of the research team.
The process is somewhat akin to assembling an IKEA sofa in your apartment versus hauling in a preassembled sofa. Assembling the sofa in the apartment allows the builder to use tools they already have and to integrate it more seamlessly with their existing furniture.
“The elegance of this approach lies in its simplicity: with just a well formulated monomer BDF, Jayant, Mei, and coworkers show hemoproteins can catalyze in vivo polymerization into an optoelectronically active polymer,” says Helen Tran, a polymer chemist at the University of Toronto, in an email.
She also adds that it will be important to monitor the components for any toxicity. “It takes time for the polymers to form and over time the polymer may degrade; we need to make sure these periods of time do not elicit unpredictable and adverse biological responses,” she says.
Chemical & Engineering News
ISSN 0009-2347
Copyright ©
2026 American Chemical Society