Make better investment decisions with Simply Wall St’s easy, visual tools that give you a competitive edge.
AtaiBeckley (NasdaqGM: ATAI) reported peer reviewed Phase 2a results for its BPL-003 program in treatment resistant depression.
The study showed a rapid and sustained antidepressant response from a single intranasal dose, with no serious adverse events.
The data were published in a major scientific journal, and the company has received FDA End of Phase 2 alignment indicating Phase 3 studies are on track.
AtaiBeckley, trading at around $4.0 per share, has seen a 181.7% return over the past year and a 95.1% return over three years. This puts recent trial news in a meaningful context for existing shareholders. In the shorter term, the stock has a 6.4% return over the past week and 9.6% over the past month, with a 3.4% return year to date. This suggests the market has been actively reacting to company specific developments.
For readers tracking psychedelic based mental health treatments, these Phase 2a results and FDA alignment on Phase 3 design indicate a clearer clinical path for BPL-003. Future updates are likely to focus on Phase 3 trial initiation, patient enrollment progress, and how AtaiBeckley funds and prioritizes this program alongside the rest of its pipeline.
Stay updated on the most important news stories for AtaiBeckley by adding it to your watchlist or portfolio. Alternatively, explore our Community to discover new perspectives on AtaiBeckley.
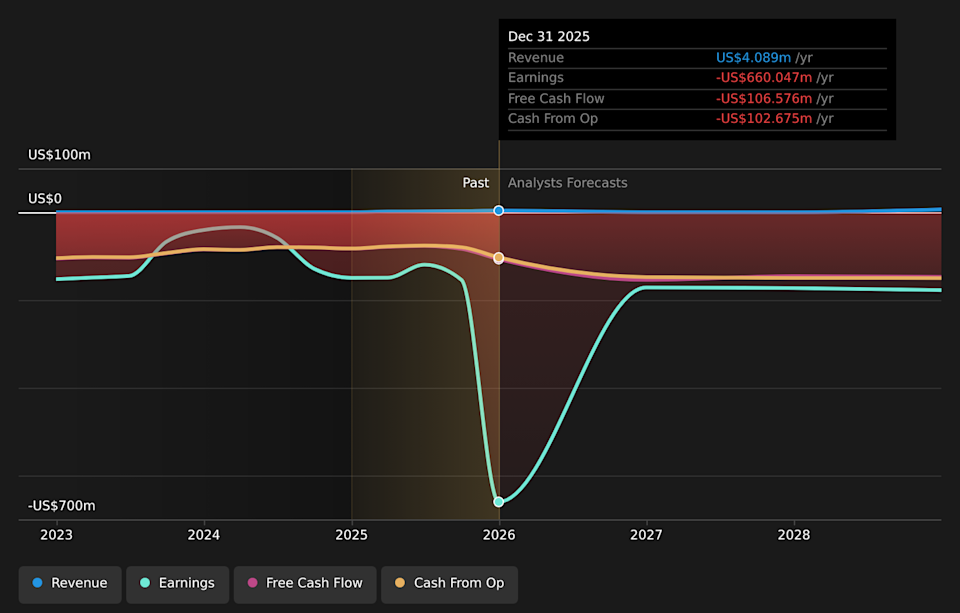 NasdaqGM:ATAI Earnings & Revenue Growth as at Apr 2026
NasdaqGM:ATAI Earnings & Revenue Growth as at Apr 2026
2 things going right for AtaiBeckley that this headline doesn’t cover.
BPL-003 sits in a crowded but early-stage field of psychedelic-based therapies for treatment-resistant depression, where Johnson & Johnson’s Spravato and various psilocybin programs from COMPASS Pathways and Cybin often shape investor expectations. The Phase 2a data matters because it shows rapid and sustained changes on a standard depression scale from a single intranasal dose, with patients kept on their existing SSRI treatment and no serious safety events reported. For AtaiBeckley, that combination of convenience, a short treatment time of about two hours, and the ability to keep background antidepressants in place could be commercially relevant if later stage trials reach similar outcomes. FDA Breakthrough Therapy Designation and End of Phase 2 alignment mean regulators see enough early signal to support a Phase 3 plan, which often helps companies communicate a clearer development path to partners and payers. At the same time, the trial is open label with only 12 participants, so the real test will come in larger, controlled Phase 3 studies that need to support pricing, reimbursement, and adoption against existing options.
⚠️ AtaiBeckley does not currently generate meaningful revenue of around US$4m, so the business still depends heavily on external funding and future product progress.
⚠️ Shareholders have been substantially diluted in the past year, and further capital raises are a possibility as the company funds Phase 3 trials and broader development.
🎁 BPL-003 has FDA Breakthrough Therapy Designation with rapid, sustained responses reported and Phase 3 planning aligned with the FDA, which may support a clearer route toward potential commercialization.
🎁 Analysts indicate the company is trading at a large discount to their estimate of fair value and expect strong revenue growth, which some investors may view as upside if execution on the pipeline continues.
Investors will likely focus on the timing and design of the Phase 3 BPL-003 trials in the second quarter of 2026, including whether they are randomized, controlled, and sized to support regulatory filings. Funding plans for those studies, any partnership activity, and updates from the remaining parts of the Phase 2a program, especially the two-dose regimens, will also be important. In the broader psychedelics space, sales trends for approved treatments like Spravato can shape expectations for how new therapies might be adopted in real-world clinics, so it is worth tracking how payers and treatment centers respond to this class of drugs over time.
To ensure you’re always in the loop on how the latest news impacts the investment narrative for AtaiBeckley, head to the community page for AtaiBeckley to never miss an update on the top community narratives.
This article by Simply Wall St is general in nature. We provide commentary based on historical data and analyst forecasts only using an unbiased methodology and our articles are not intended to be financial advice. It does not constitute a recommendation to buy or sell any stock, and does not take account of your objectives, or your financial situation. We aim to bring you long-term focused analysis driven by fundamental data. Note that our analysis may not factor in the latest price-sensitive company announcements or qualitative material. Simply Wall St has no position in any stocks mentioned.
Companies discussed in this article include ATAI.
Have feedback on this article? Concerned about the content? Get in touch with us directly. Alternatively, email editorial-team@simplywallst.com