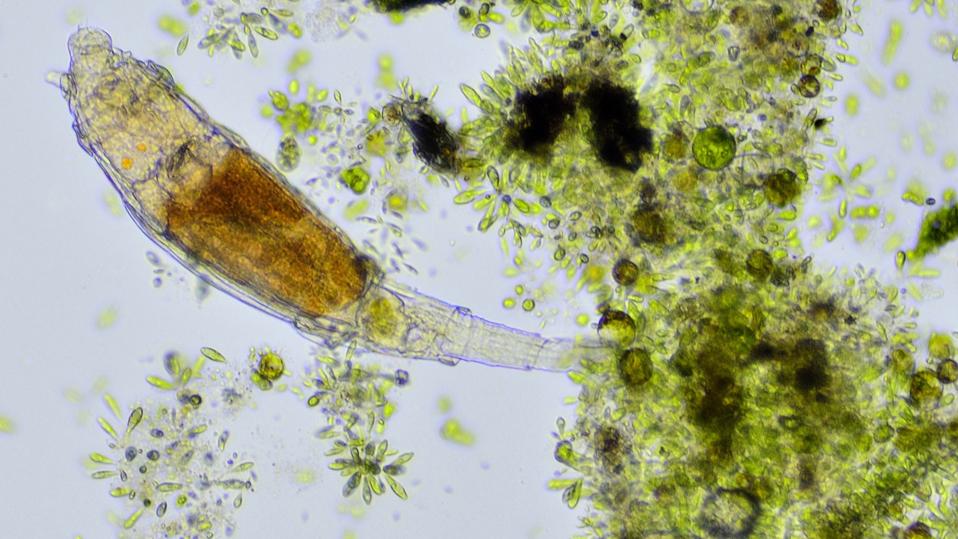
Radiation is one of the most lethal stressors that a living creature can face. In humans, acute exposure to even just a few grays (units of absorbed radiation) can cause nausea, organ failure, and death. And yet, in some microscopic animals, the limits of this radiation resistance are incomprehensibly higher than what we humans can tolerate. Among these super-survivors are minuscule aquatic animals, so small that they can’t even be seen with the naked eye: bdelloid rotifers.
For evolutionary biologists, molecular geneticists or anyone intrigued by life at its extremes, bdelloid rotifers are a fascinating case study in the adaptive strategies of life on Earth that push the boundaries of what we thought possible.
What Are Bdelloid Rotifers?
Bdelloid rotifers aren’t household names. They are tiny, mostly freshwater animals, usually less than a millimeter long, that can be found worldwide in freshwater habitats, on wet mosses, in soil films and even in ephemeral puddles that dry out periodically.
But under a microscope, they are incredibly complex for their size: they have muscular pharynges, ciliated structures used for movement and feeding, as well as entire organ systems. Notably, one of their most unique defining features is that they’re ancient asexuals, meaning that they appear to reproduce almost entirely without sex.
However, what makes them especially extraordinary is that they can survive exposure to ionizing radiation doses up to 1,000 times greater than the lethal dose for humans — and recover completely. Somehow, an organism smaller than what we can even see can repair DNA damage that would be catastrophic in most other animals.
To quantify just how extreme this is, humans exposed to anything around 5 to 10 grays (Gy) of radiation, even in a short time, are very likely to succumb to acute radiation syndrome without medical intervention. By contrast, bdelloid rotifers can endure doses in excess of 5,000 Gy, and the majority of their reproductive capacity will be preserved after recovery.
It’s important to note that this ability is not entirely unique among extremophiles; however, among multicellular animals, bdelloid rotifers’ tolerance is virtually unmatched.
Bacteria such as Deinococcus radiodurans can also survive enormous doses, and certain tardigrades (water bears) also endure high radiation levels. What distinguishes bdelloids is the specific combination of their multicellularity, reproductive resilience and ability to resume active life after extreme damage.
Tardigrades, for instance, can endure high radiation while in cryptobiotic states, but they would perish if exposed to the same levels while metabolically active. Bdelloids, by contrast, can repair radiation damage while active.
Many wonder how such a feat is possible for such a small organism. Intriguingly, researchers have discovered that their radiation resistance appears to be tied to their ability to survive desiccation (drying out completely). That is, the same mechanisms that enable them to repair the DNA damage caused by extreme dehydration are also what help them withstand ionizing radiation.
In a 2014 study published in the Journal of Evolutionary Biology, researchers uncovered that bdelloid rotifers can survive prolonged desiccation by entering a state of suspended animation. In this state, their metabolism drops to near zero as their DNA undergoes fragmentation. But as soon as they’re reexposed to water, they rehydrate and swiftly repair the damage with no issue.
And as the researchers discovered, this robust DNA repair machinery is also incredibly effective against radiation-induced DNA breaks.
Bdelloid Rotifers’ DNA Repair Secret
Ionizing radiation kills cells primarily by breaking the strands that make up organisms’ DNA. When DNA is broken in many places at once — as it does with high radiation doses — biologists previously assumed that there was no way for most multicellular organisms to reassemble their genomes correctly.
Researchers assumed that, due to the accumulation of errors in repairs, radiation exposure would almost always lead to cell death or malfunction. Bdelloid rotifers, however, have evolved exceptionally efficient DNA repair systems, as 2008 research from PNAS explains.
Specifically, when radiation shatters their genomes into dozens or hundreds of fragments, their cellular machinery stitches the DNA back together with remarkable fidelity. Indeed, the study confirmed that rotifers exposed to extreme radiation exhibited massive DNA fragmentation shortly after exposure. However, within hours to days, most of these fragments were rejoined, and normal cellular functions subsequently resumed.
This ability most likely didn’t evolve due to rotifers being regularly exposed to radiation in the wild, at least not at lethal doses. More likely, it’s because of the frequent desiccation and rehydration cycles that occur so commonly in their natural habitats.
Bdelloid rotifers often inhabit environments that dry out completely. When water disappears, they enter a state called anhydrobiosis, in which their metabolism halts, and their cells lose almost all water. In this state, cellular components are tightly packed, and the rotifer appears almost completely lifeless.
The molecular challenge of anhydrobiosis is enormous. This is because dehydration can cause DNA to fragment while also leading proteins to aggregate. To survive, bdelloids need to produce protective sugars and proteins that stabilize their cellular components. They also express a suite of DNA repair enzymes that quickly fix the damage incurred during the dry period.
It’s this very same machinery that explains their resistance to radiation: the sheer oxidative stress induced by radiation produces DNA breaks that are highly similar to those caused by desiccation. Rotifers evolved to cope with one form of extreme damage, a mechanism that coincidentally allows them to tolerate another.
Why Bdelloid Rotifers Matter To Biology
Radiation resistance is a side effect of adaptation to a different environmental stress. This is a prototypical example of something commonly seen throughout evolutionary biology: organisms evolving solutions to one problem that incidentally confer resilience to another. And this extraordinary resilience has multiple implications beyond their extreme habitats:
Understanding DNA repair. The mechanisms these organisms use to reassemble highly fragmented genomes reveal how cells can reliably fix even catastrophic damage. This knowledge may inform research into cancer therapy and aging, which are processes where DNA damage and repair play central roles.
Evolution without sex. Bdelloids have managed to persist for millions of years without sexual reproduction. This fact raises several important questions about how they maintain genetic diversity and avoid the accumulation of life-threatening DNA mutations. It’s likely that their repair systems may contribute to genome stability in the absence of recombination.
Life in extreme environments. Studying organisms that survive extremes expands our understanding of the limits of life. This has relevance in astrobiology, where scientists consider what forms life might take on other planets, or even on moons with intense radiation or desiccating conditions.
Biotechnology. Understanding the molecular basis of desiccation and radiation resilience could inspire new strategies for preserving biological materials (e.g., vaccines, cells, etc.) without the need for refrigeration. This has been a long-sought goal in global health.
The bdelloid rotifer’s ability to survive radiation levels is one of many examples of how evolution enables organisms to adapt to unimaginable environmental stresses — in this case, drying and rehydration. But more interestingly, it also reveals how those adaptations can have surprising secondary benefits.
Bdelloid rotifers push the limits of life itself. Curious how connected you feel to nature’s incredible diversity? Take the science-backed Connectedness to Nature Scale to find out.