Scientists have discovered a previously unknown pathway that allows toxic proteins linked to Huntington’s disease to spread between brain cells.
Huntington’s disease, a debilitating inherited brain disorder, may be driven by a newly discovered cellular mechanism that allows toxic proteins to spread directly between brain cells, according to new research.
Huntington’s disease gradually robs people of movement, memory and personality. It is caused by a toxic form of the huntingtin protein, which accumulates in brain cells and eventually destroys them. Although researchers have long known that this protein can spread from cell to cell, the exact mechanism behind this process wasn’t clear.
Now, scientists from Florida Atlantic University and international collaborators have identified a previously unknown pathway that enables neurons to pass harmful material to neighbouring cells through tiny tube-like connections. The findings show that blocking this pathway can significantly reduce the spread of the disease-causing protein.
Tiny cellular ‘bridges’ enable toxic spread
The study focuses on microscopic structures known as tunnelling nanotubes, which act as direct bridges between cells. Unlike chemical signals that travel through surrounding space, these nanotubes allow cells to transfer proteins and other materials directly.
The study focuses on microscopic structures known as tunnelling nanotubes, which act as direct bridges between cells.
While such connections can help healthy cells respond to stress or injury, they can also be hijacked to spread harmful proteins.
Researchers discovered that a protein called Rhes works alongside a bicarbonate transporter typically involved in regulating cellular acidity, called SLC4A7. Together, these proteins promote the formation of nanotubes, effectively creating pathways for the toxic huntingtin protein to move between neurons.
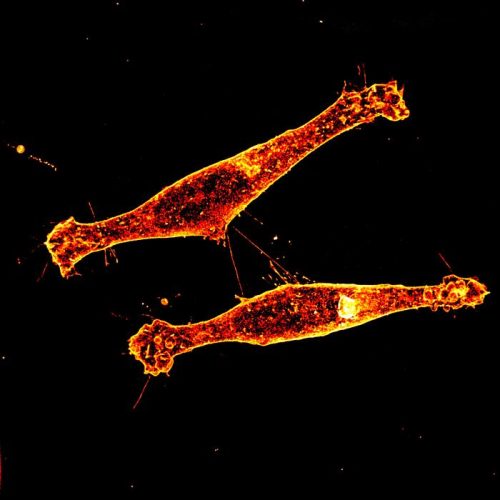
Tunnelling nanotubes connect Rhes expressing striatal neuronal cells. Credit: Emaad Mirza, Florida Atlantic University.
Key protein partnership identified
“This work fundamentally changes how we think about disease progression in Huntington’s,” said Dr Srinivasa Subramaniam, Associate Professor in the Department of Chemistry and Biochemistry at Florida Atlantic University and a senior author of the study. “We’ve known that neurons somehow pass toxic proteins to one another but now we can see the machinery that makes that possible. By identifying SLC4A7 as a key partner of Rhes, we’ve uncovered a new and potentially druggable target to stop that spread at its source.”
This work fundamentally changes how we think about disease progression in Huntington’s.
Using advanced protein-mapping techniques, the team found that Rhes binds directly to SLC4A7 at the cell membrane. This interaction triggers changes that encourage nanotube growth. When SLC4A7 was blocked, either genetically or with drugs, nanotube formation was prevented and the spread of the toxic protein was largely halted.
Promising results in animal studies
The findings were not limited to laboratory cell models. In mice engineered to develop Huntington’s disease, those lacking SLC4A7 showed a marked reduction in the transfer of toxic protein between neurons in the striatum, the brain region most affected by the condition.
This suggests that targeting the newly identified pathway could slow disease progression by containing damage before it spreads further.
Wider implications for other diseases
The discovery could have implications beyond Huntington’s disease. Tunnelling nanotubes have also been linked to other neurodegenerative conditions, including those involving tau protein, as well as cancer, where similar structures help tumour cells share resources and resist treatment.
Tunnelling nanotubes have also been linked to other neurodegenerative conditions, including those involving tau protein, as well as cancer.
“This research shines a spotlight on an entirely new way cells communicate in health and disease,” said Dr Randy Blakely, Executive Director of the Florida Atlantic University Stiles-Nicholson Brain Institute. “By learning how harmful proteins physically move from cell to cell, we gain powerful new leverage points for therapy. The idea that we could slow or even halt disease progression by blocking these microscopic tunnels opens an exciting frontier for treating not only Huntington’s disease but a wide range of neurological disorders and cancers in the future.”
Hope for future treatments
Huntington’s disease affects an estimated three to seven people per 100,000 worldwide. Symptoms usually appear between the ages of 30 and 50 and progressively worsen, leading to severe physical, cognitive and psychiatric difficulties. There is currently no cure and treatments are limited to managing symptoms.
As scientists continue to explore how cells communicate and how these processes can go wrong, the discovery offers fresh hope that stopping diseases like Huntington’s could one day be achieved by interrupting the pathways that allow them to spread.
Related topics
Central Nervous System (CNS), Disease Research, Drug Discovery Processes, Drug Targets, In Vitro, In Vivo, Molecular Biology, Molecular Targets, Neurons, Neurosciences, Protein, Target Validation, Translational Science