Video-assisted thoracoscopic surgery (VATS) has already become the dominant surgical approach for lobectomy in early-stage non-small-cell lung cancer (NSCLC), largely because of its well-established perioperative advantages. Patients undergoing VATS generally report less postoperative pain, fewer complications, shorter hospital stays, faster recovery, and better quality of life compared with open thoracotomy, as shown in earlier randomized trials by Bendixen et al. (2016), Lim et al. (2022), and Long et al. (2018). What remained uncertain, however, was the oncologic question that matters most over the long term: does VATS offer survival outcomes comparable to, or even better than, open surgery?
A new individual patient data meta-analysis provides the strongest answer to date. By pooling patient-level data from all eligible randomized trials, the investigators found that VATS lobectomy was associated with a 21% reduction in the risk of death compared with open lobectomy, with no compromise in disease-free survival (Harris et al., 2026). These results strengthen the scientific rationale for prioritizing VATS when technically feasible in patients with early-stage NSCLC.
Why This Study Matters
The adoption of VATS in thoracic surgery has been driven mainly by non-oncologic benefits. Although observational studies and registry analyses have long suggested that minimally invasive surgery may also improve survival, those findings have always been vulnerable to selection bias and residual confounding. Patients chosen for VATS in routine practice are often healthier, have smaller tumors, or are treated at more specialized centers, making it difficult to conclude that the surgical approach itself causes better survival.
That is why this new meta-analysis is important. Instead of relying on retrospective comparisons, the authors performed an individual patient data meta-analysis of randomized controlled trials, which is among the highest levels of evidence available for comparative effectiveness research. This design allowed them to move beyond assumptions and examine survival directly at the patient level (Harris et al., 2026).
Study Design and Methods
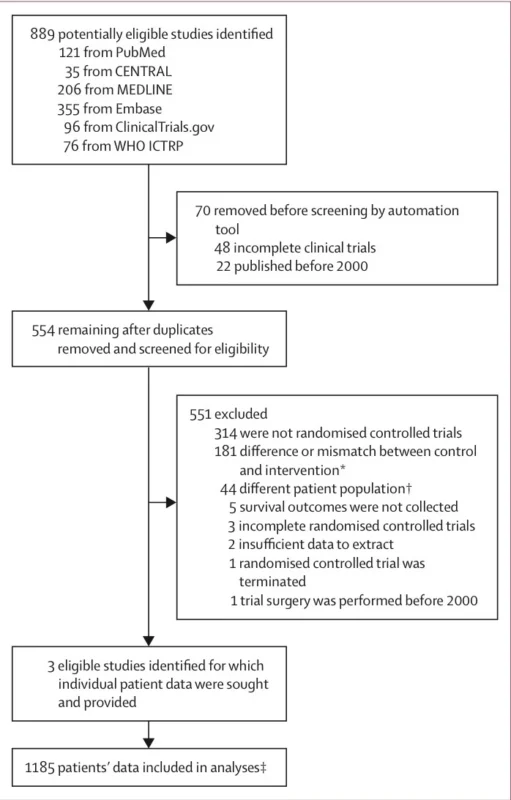
The investigators systematically searched PubMed, MEDLINE, Embase, CENTRAL, ClinicalTrials.gov, and the WHO International Clinical Trials Registry Platform for randomized trials published between January 1, 2000 and June 13, 2025. Eligible studies had to compare VATS versus open lobectomy in adults with clinical stage I–II NSCLC, include contemporary surgery performed after 2000, and collect mortality and recurrence data.
Out of 554 screened records, only three randomized controlled trials met eligibility criteria. These were the PLEACE trial from Denmark, the trial by Long and colleagues from China, and the VIOLET trial from the United Kingdom. Individual patient data were obtained for 1,185 patients, including 586 randomized to VATS and 599 randomized to open lobectomy (Harris et al., 2026).
The primary endpoint was overall survival, defined as time from randomization to death from any cause. The secondary endpoint was disease-free survival, defined as time from randomization to recurrence or death. The main statistical analysis used a one-stage random-effects Cox proportional hazards model, with a two-stage approach performed to confirm consistency. Importantly, the analyses were conducted on an intention-to-treat basis, meaning patients were analyzed according to the group they were originally assigned to, even if crossover occurred.
Patient Population and Follow-Up
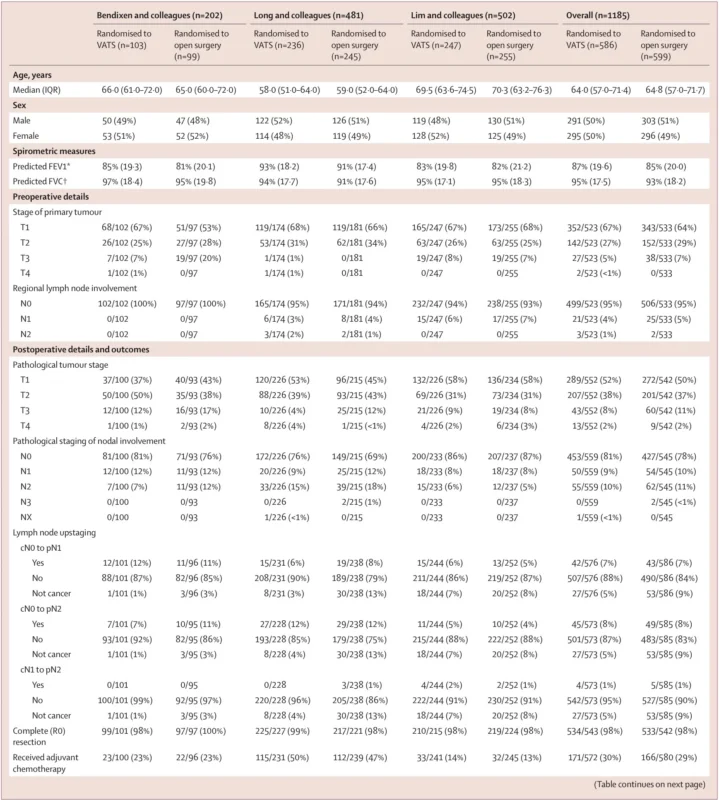
The pooled dataset reflects a robust multinational randomized experience. Median follow-up was long, reaching 9.5 years in the Danish trial, 5.2 years in the Chinese trial, and 3.8 years in the UK trial. More than 91% of randomized patients had at least 3 years of survival follow-up, which gives the study meaningful long-term relevance.
Baseline characteristics were broadly balanced between groups. Median age was around 64–65 years overall, and sex distribution was nearly equal. R0 resection rates were almost identical, at 98% in both arms, and use of adjuvant chemotherapy was also similar, 30% after VATS versus 29% after open surgery. These details are important because they reduce the likelihood that survival differences were driven by imbalances in resection quality or adjuvant treatment rather than surgical access itself (Harris et al., 2026).
Overall Survival: A Clear Advantage for VATS
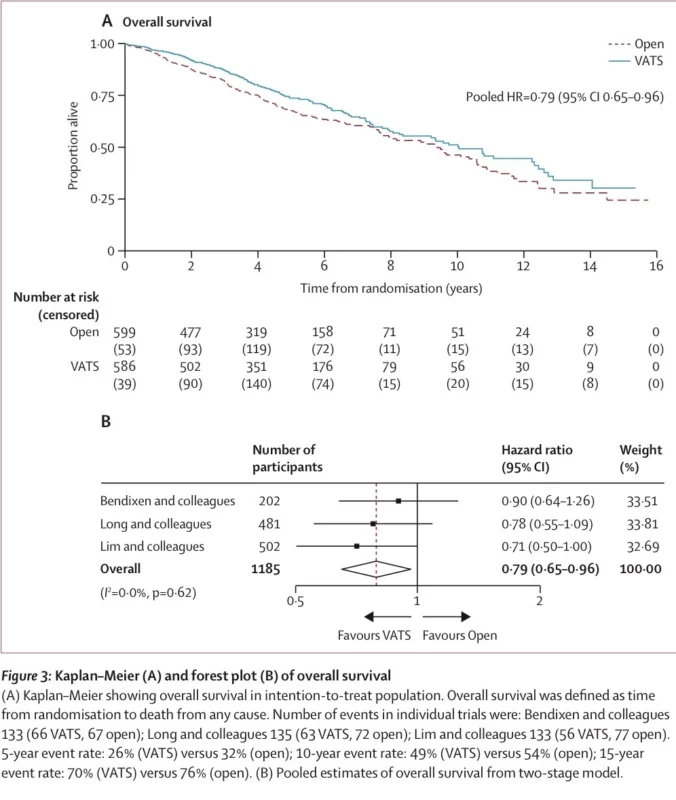
The most practice-changing result of the study is the overall survival analysis. Across the three randomized trials, VATS significantly improved overall survival compared with open lobectomy, with a pooled hazard ratio of 0.79 (95% CI, 0.65–0.96). In practical terms, that corresponds to a 21% relative reduction in mortality risk with the minimally invasive approach (Harris et al., 2026).
The benefit was consistent across trials, with no evidence of heterogeneity. Trial-specific hazard ratios all numerically favored VATS: 0.90 in the Bendixen trial, 0.78 in the Long trial, and 0.71 in the Lim trial. The absence of statistical heterogeneity strengthens confidence that the signal is real rather than driven by a single study.
Kaplan–Meier estimates also support the clinical relevance of this difference. At 5 years, the event rate for death was 26% after VATS versus 32% after open surgery. At 10 years, it was 49% versus 54%, and at 15 years, 70% versus 76%. These are not trivial differences. In resected early-stage lung cancer, where long-term survival remains a central goal, even modest improvements in mortality are meaningful.
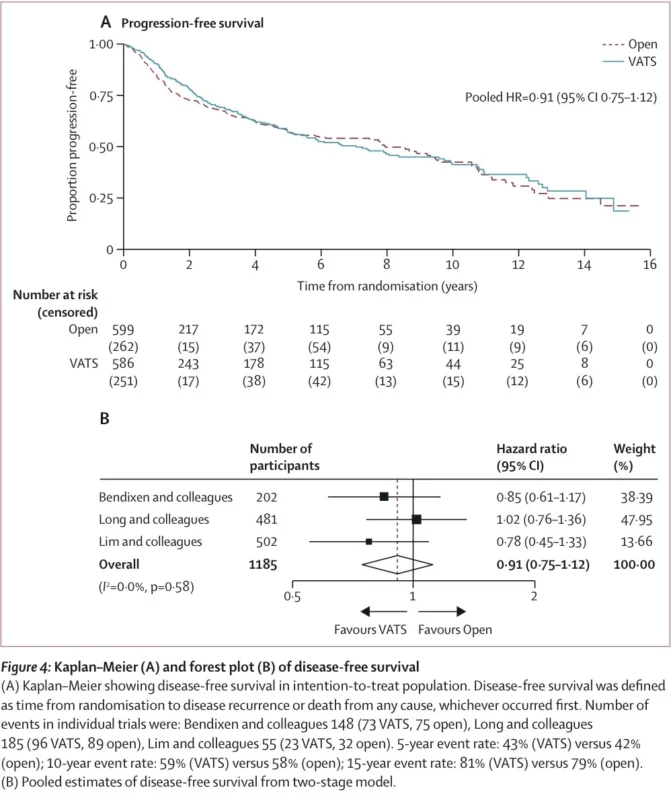
Disease-Free Survival: No Evidence of Oncologic Compromise
Disease-free survival did not significantly differ between approaches in the primary analysis. The pooled hazard ratio was 0.91 (95% CI, 0.75–1.12), indicating no statistically significant difference between VATS and open lobectomy (Harris et al., 2026).
This is an important finding. One historical concern about VATS has been whether a minimally invasive approach might compromise oncologic thoroughness, especially regarding lymph node assessment or local control. This meta-analysis does not support that concern. If anything, disease-free survival numerically favored VATS, though the difference did not reach statistical significance.
A post-hoc sensitivity analysis limited to the first 2 years after randomization did show a statistically significant disease-free survival advantage for VATS, with a hazard ratio of 0.72 (95% CI, 0.55–0.95). However, the primary conclusion remains that VATS did not worsen disease-free survival, which is clinically reassuring.
Why Might Overall Survival Be Better With VATS?
The most interesting scientific question raised by this study is why overall survival improved while disease-free survival remained similar.
The authors suggest that the explanation likely lies in reduced perioperative morbidity rather than a major difference in tumor recurrence. This interpretation fits with prior randomized evidence. In the VIOLET trial, VATS was associated with fewer adverse events and lower readmission rates than open surgery (Lim et al., 2022). Other population-based studies have also shown that unplanned readmission after lung cancer surgery is strongly associated with higher short-term and long-term mortality, including a six-fold increase in 90-day mortality in one analysis (Hu et al., 2014; Puri et al., 2015).
In other words, even if both operations achieve similar cancer control, the less invasive procedure may improve long-term survival by reducing the physiologic burden of surgery, postoperative complications, and downstream non-cancer deaths. This is highly plausible in an older lung cancer population, where comorbidity burden is substantial.
What About Lymph Node Staging?
One argument sometimes used in favor of open surgery is that it might allow better lymph node dissection and more accurate staging. This meta-analysis did not support that theory in a clinically meaningful way.
The pooled differences in nodal upstaging were minimal. The rate of cN0 to pN1 upstaging was 7% in both groups, and cN0 to pN2 upstaging was 8% in both groups. These findings suggest that, in experienced hands and within trial settings, VATS can deliver lymph node staging that is essentially equivalent to open thoracotomy (Harris et al., 2026).
That point matters because it removes one of the main oncologic objections to minimally invasive lobectomy.
Strengths and Limitations
This study has several major strengths. It is based on randomized trials, uses individual patient data rather than only published aggregates, includes long follow-up, and applies rigorous intention-to-treat analyses. More than 99% of randomized patients were included, and the trials were judged to be at low risk of bias.
At the same time, the authors acknowledge important limitations. Only three trials were available, and they were conducted in Europe and China, which may limit generalizability. Cause-specific mortality was not available, so the study could not formally separate lung cancer-specific from non-cancer mortality. Disease recurrence follow-up was also less complete in one trial. In addition, all three trials predated widespread use of perioperative immunotherapy and targeted therapies such as osimertinib, atezolizumab, pembrolizumab, and nivolumab in resectable NSCLC. That means the results apply most directly to surgery itself, rather than to modern multimodality treatment pathways.
What This Means for Practice
This meta-analysis shifts the discussion around VATS. The procedure is no longer supported only by better pain control and faster recovery. It now has randomized, patient-level evidence showing an overall survival advantage over open lobectomy in early-stage NSCLC, without evidence of inferior disease-free survival (Harris et al., 2026).
That does not mean every patient is a VATS candidate. Surgical decision-making still depends on tumor location, anatomy, technical feasibility, and surgeon expertise. But when both approaches are possible, the weight of evidence now favors VATS not just for recovery, but also for long-term survival.
Conclusion
The 2026 individual patient data meta-analysis by Harris and colleagues provides the strongest evidence to date that VATS lobectomy improves overall survival compared with open lobectomy in early-stage NSCLC, with a pooled 21% reduction in mortality risk and no compromise in disease-free survival (Harris et al., 2026). These findings reinforce VATS as the preferred surgical access for lobectomy when technically feasible and support its role as the modern standard of care in early-stage lung cancer surgery.
Full article here