Scientists have identified a small cluster of cells in the mosquito gut that shuts down the urge to hunt humans after blood feeding.
This discovery is surprising because it places a key appetite control mechanism inside a mosquitoes’ gut, rather than the brain, tying feeding, egg-making, and biting together.
The female mosquito gut is key

Inside six tiny pads at the end of the female gut, the cells sit where fullness and nutrients converge.
Tracing that tissue, Laura Duvall at Columbia University found the receptor in those cells and not in the brain.
For decades, female mosquitoes were known to lose interest in biting for several days after a blood meal, but the switch stayed hidden.
Pinning it to the rectum clarified where that switch sits, while leaving its signals and consequences to explain.
Why biting stops
In female Aedes aegypti – the yellow-fever mosquito – a blood meal quiets the urge to seek humans for several days.
In 2019, Duvall linked that pause to NPYLR7, a receptor protein that receives appetite signals, and disabling it erased fullness.
The new work adds location and purpose, showing that the receptor helps turn blood nutrients into yolk for developing eggs.
Once that link snapped, the research stopped being only about biting and became a story about reproduction.
Poor egg outcomes
When researchers knocked out NPYLR7, the mosquitoes still drank normal blood meals and laid clutches of ordinary size.
Yet their eggs hatched far less reliably, and mature ovaries carried less protein, meaning the meal was not invested well.
Wild-type females laid about 82 eggs after full blood meals, while diluted meals cut that average to about 28.
Because mutants still failed after extra or diluted meals, the weak point looked like allocation, not appetite or digestion.
Not just waste
Plumbing was the obvious explanation, because the rectum normally helps insects recover water, salts, and small nutrients.
Across blood, saline, and sugar meals, the mutants mostly cleared food on schedule and survived the load.
Some mid-course delays appeared, but the meals still passed through within the expected hours or days.
Instead, the result narrowed the problem to signaling, pushing the story away from waste handling and toward decision-making.
Signals after feeding
Nearby nerve endings became the next suspect, because they sit close enough to talk directly with the rectal cells.
After a meal, those nerves release RYamide, a peptide signal from nerve cells, and the target cells answered with rising calcium.
Amino acids also stirred the cells, which fits the idea that they monitor both fullness and nutrient quality.
These responses make the rectum look less like a drain and more like a checkpoint.
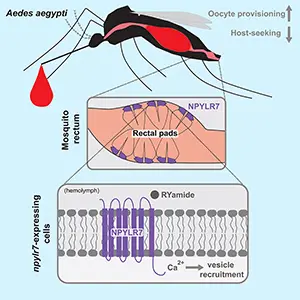 Neuropeptide Y-related pathways regulate feeding in many animals, and loss of neuropeptide Y-like receptor 7 (NPYLR7) in the mosquito gut disrupts host-seeking regulation in Aedes aegypti. Credit: Current Biology. Click image to enlarge.Mosquito gut sends messages
Neuropeptide Y-related pathways regulate feeding in many animals, and loss of neuropeptide Y-like receptor 7 (NPYLR7) in the mosquito gut disrupts host-seeking regulation in Aedes aegypti. Credit: Current Biology. Click image to enlarge.Mosquito gut sends messages
The strange part was what these rectal cells seemed ready to send back to nearby nerves.
Genes active in that gut tissue pointed to vesicles, tiny packets cells use to release signals, plus machinery for common nerve chemicals.
Electron microscope images then showed pools of those packets gathering after blood feeding in normal females, but not mutants.
Missing packets suggest the receptor helps prepare a return message, although the exact chemical signals remain unknown.
A cross-species clue
Beyond mosquitoes, animal guts often help decide when eating should slow or stop in many species.
In people, some weight-loss drugs mimic a gut hormone that helps signal fullness through nerve and brain pathways.
Elsewhere in mammals, gut sensory cells can synapse with nerves linked to the brain and pass nutrient information in milliseconds.
Even so, the parallels do not make mosquito biology the same as ours, but they strengthen the case that gut-to-brain control is ancient.
A target to feed
Control researchers care because a receptor in the gut is easier to reach than one buried in the brain.
Newer compounds that activate NPYLR7 suppressed blood feeding at doses 100 times lower than earlier molecules.
“It’s a much more accessible target than a receptor in the brain,” said Duvall.
Still, the strategy remains early and untested outside controlled experiments, but it points toward bait-based tools that change biting behavior.
Controling mosquitoes through the gut
Important pieces are still missing, especially what signal these cells send after they sense blood-derived nutrients.
The team suspects messages travel back to the nervous system, but other organs could also receive the call.
No one yet knows whether the same pathway operates in other blood-feeding insects or stays mostly mosquito-specific.
For any control strategy, those gaps remain important, since gut-based tools must work reliably outside one laboratory species.
A mosquito meal now starts a signaling chain, with gut cells judging nutrients and helping decide when biting ends.
If scientists can tap that chain from the outside, they may gain a practical way to curb bites before they happen.
The study is published in Current Biology.
—–
Like what you read? Subscribe to our newsletter for engaging articles, exclusive content, and the latest updates.
Check us out on EarthSnap, a free app brought to you by Eric Ralls and Earth.com.
—–

