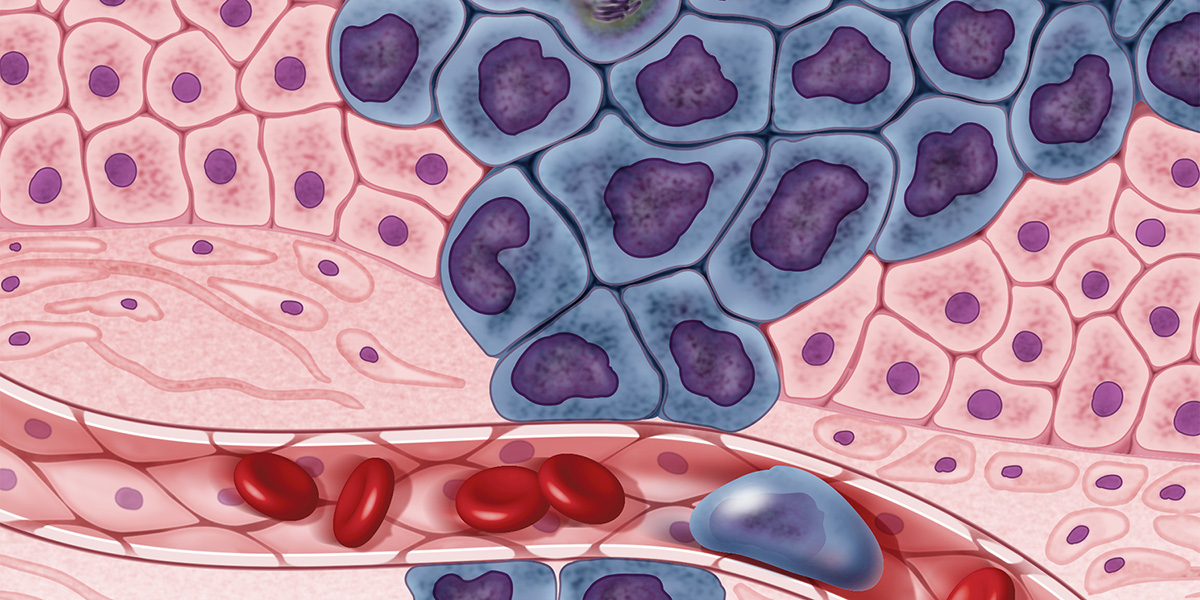
Cholangiocarcinoma, or CCA, is a rare form of cancer that originates in the bile duct. One commonly observed feature of CCA is disruption of glycosylation, a type of protein posttranslational modification. If proteins are modified with too many, too few or incorrect sugars, this can encourage cancer progression by disrupting the interactions between the glycosylated proteins and other cells, helping cancer cells avoid immune responses. Studies have revealed the glycoproteomic profile of the intrahepatic CCA subtype, or iCCA, which arises inside the liver. But, until recently, the extrahepatic subtype, or eCCA, counterpart, which arises outside of the liver, remained unknown.
In a study published in Molecular & Cellular Proteomics, Zhili Xia, Li Gao, Meng Hu, Yingjie Li, Kexin Yu, Wenbo Meng and a team of researchers at Lanzhou University and Sichuan University investigated the eCCA glycoproteome and compared it to that of iCCA. They performed tandem mass tag glycoproteomics on eCCA tumor samples to identify glycoproteins and modification sites and then compared their data to a publicly available iCCA single-cell RNA sequencing dataset. They found that the two subtypes had different proportions of certain types of sugars; for example, eCCA featured more sialylated glycans capped with sialic acid. Further comparisons revealed different immune cell contents, suggesting that the subtypes undergo different changes to their tumor microenvironments.
These insights may help identify biomarkers for diagnosing the CCA subtypes and guide the development of immunotherapies. More research is needed to validate these findings with larger sample sizes and to understand the roles of specific enzymes for glycosylation-targeting therapies.