Ziziphus spina-christi is a tree that produces edible fruits known as Nabq. It belongs to Rhamnaceae family that is adapted to grow in warm or hot climates. It is one of the oldest medicinal plants used in ancient Egypt medicine for its anti-inflammatory properties7. Its known to be rich in chemical compounds, and nowadays, leaf, fruit, and seed extracts have been reported to possess several pharmacological benefits, including management of diabetes and treatment of malaria, digestive issues, typhoid, liver problems, weakness, skin infections, urinary disorders, obesity, diarrhea, and antimicrobial activity (antifungal, antiviral, and anti-bacterial activity)9. In this study, we tested the antiviral potential of different fruit and leaf extracts of Ziziphus spina-christi and used the docking study to predict the main potent compounds that may inhibit the replication of three viruses, Human influenza H1N1, MERS-CoV-2 spike, and SARS-CoV-2. Five types of extracts from leaves and fruits were tested, (crude original, Hexane, Ethyle acetate, Dichloromethane, and the residual extracts). Each extract was subjected to mass spectrophotometry to identify its chemical compounds.
Flavonoids, phenolic acids, saponins, sterols, and triterpenes and their glucosides were the major detected compounds in both leaf and fruit fractions and extracts. Of these phenolics, caffeic acid (m/z 179.00 [M-H]−) was the most predominant in all six samples in negative ion mode, while its glucoside, caffeic acid 4-O-glucoside (m/z 341.01 [M-H]−) was identified in methanol extract of both leaves and fruits beside the remaining aqueous fraction of the fruits. These findings are in agreement with previous studies on leaf extract of Z. spina-christi41 and Z. lotus fruits42. Additionally, caffeoylquinic acid (m/z 353 [M-H]−) was detected in all tested plant sample extracts and fractions except the ethyl acetate fraction of the fruits. This compound and its analogues have been previously isolated from Z. spina-christi leaf extracts43. Among the flavonoid, quercetin, luteolin, taxifolin, isoquercetin, quercetin 3-O-(2-O-rhamnosyl-arabinoside), 5,7-Dihydroxy- 8,3’,5’ trimethoxy-flavone, and quercetin-3-O-α-L-arabinopyranosyl-(1→2)-α-L-rhamnopyranoside were predominantly detected in the ethyl acetate fraction of the fruits. This is in accordance with the previous studies44,45,46 where quercetin-3-O-α-L-arabinopyranosyl-(1→2)-α-L-rhamnopyranoside was isolated from Z. spin-cristi leaves, and isoquercitrin and quercetin were detected in the methanolic fruit extract. Moreover, the isoflavonoid compound hydroxygenistein methyl ether malonylhexoside (m/z 547 [M-H]−) was identified in all leaf and fruit samples except the remaining aqueous fraction of the fruits. The dammarane saponin, lotoside II (m/z 957 [M-H]−), the triterpenoid saponin Zizyphus saponin I (m/z 911 [M-H]−), and the triterpenoid compound betulinic acid (m/z 455 [M-H]−) were the common saponin compounds present in both leaf and fruit extracts and their fractions. Previous studies reported the isolation of lotoside II from root bark of Z. lotus, while ziziphus saponin I was isolated from the methanol extract of Z. mauritiana47. Additionally, chloroform extracts of Z. jujuba fruits were rich in betulinic acid and other triterpenic acids48. Ceanothic acid (m/z 485 [M-H]−), a ring-A homologue of betulinic acid, was identified in the methanol extract and ethyl acetate fraction of both leaves and fruits, but it wasn’t detected in the aqueous fractions. This compound has previously been reported in fruits of Z. jujuba and Z. jujuba var. spinosa49, as well as in methanolic leaf extracts of Z. mauritiana Lam., Z. spina-christi (L.), and Z. jujuba50. The sterol compound, stephanol, (m/z 397 [M-H]−) was identified in the fruit methanol extract and its fractions, but not in the methanol leaf extracts or fractions.
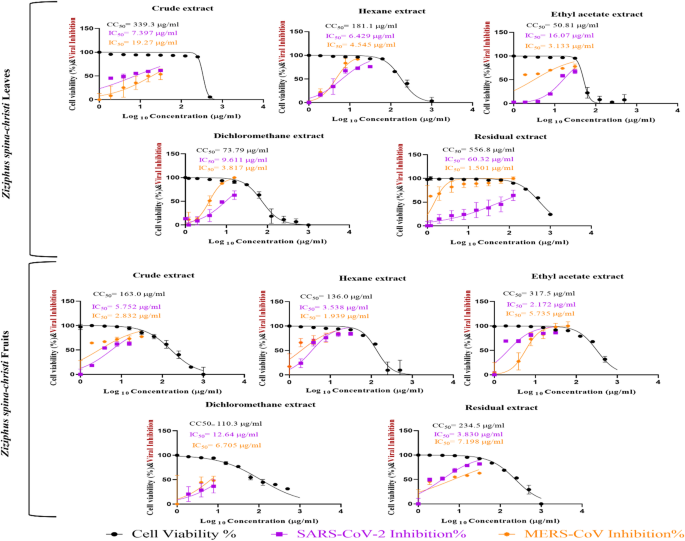
The extracts were then titrated in a dose-dependent manner in VERO and MDCK cell lines, to determine the safe concentration that could be used safely in subsequent assays. In the anti-coronavirus assays, the crude leaf extract (SI = 45.9) and the ethyl acetate fruit extract (SI = 146.3) were the most potent in vitro against SARS-CoV-2, while the residual leaf extract (SI = 371.0) and the hexane fruit extract (SI = 70.1) were the most potent in vitro against MERS-CoV. On the hand, the antiviral activity against the human influenza virus H1N1 indicated that the most potent extracts were the crude leaf extract (SI = 304.0) and the hexane fruits extract (SI = 358.0).
These most active extracts were then tested to determine the stage of the viral cycle at which the observed inhibitions was most pronounced. Interestingly, all tested extracts were shown to interfere with the virus infection step, when the virus was incubated directly with the extract before infecting the corresponding cell line. This pattern may suggest that the active extracts act at an early stage of infection, possibly by affecting viral particles or interfering with virus-cell interaction during adsorption or entry51. However, the present data do not allow confirmation of the exact molecular mechanism, and this possibility should therefore be interpreted with caution. Additional mechanistic studies would be required to determine whether these extracts interact directly with viral surface proteins, host cell receptors, or other factors involved in early infection. This more cautious interpretation is consistent with the plaque reduction findings and avoids overattributing the observed effect to a specific structural change in the virus. In silico docking studies were performed to determine which of the identified compounds in each extract are the most potent in binding to viral surface proteins, the HA and NA of influenza H1N1, and the spike proteins in SARS-CoV-2 and MERS-CoV. Interestingly, two chemical compounds, the lotoside II and hydroxygenistein methyl ether malonylhexoside, showed the most favorable docking profiles across all tested targets.
Lotoside II formed three hydrogen bonds with His38, Asn53, and Ser291 of the HA of H1N1. However, hydroxygenistein methyl ether malonylhexoside formed one hydrogen bond and one hydrogen-pi bond with His38. These residues are located in regions relevant to HA structure and function, site 38 in HA is located adjacent to the conserved stem epitope of the protein that directly controls virus replication rate52 and Asn53 is a conserved site that directly affects HA-mediated cell entry53, suggesting that these compounds may have the capacity to interact with biologically important sites. However, docking results alone cannot establish functional inhibition and should be interpreted only as supportive evidence for possible binding. Similarly, in the H1N1 NA model, lotoside II formed five hydrogen bonds with Asp151 (2), Trp178, Glu276, Glu277, and Arg371. However, hydroxygenistein methyl ether malonylhexoside formed two hydrogen bonds with Glu277 and Arg371. Asp151, Glu276, and Arg371 are the main conserved residues located in the catalytic active site of the NA protein, which are conserved in all influenza A NA subtypes54,55, while Trp178 and Glu277 are conserved structural residues in the active site of the NA protein54. Because several of these residues are located within or near the known active site of NA, these interactions may be relevant to protein binding. Nevertheless, it would be inappropriate to conclude from docking alone that binding to these amino acids directly inhibits protein function, viral replication, or viral release. Rather, these findings indicate that the identified compounds have predicted affinity for regions of functional interest, which supports their prioritization for future biochemical and target-based validation studies.
For the MERS-CoV spike protein, Lotoside II formed one hydrogen bond with Asp902, while hydroxygenistein methyl ether malonylhexoside bound to Asp902 and Glu944 with two hydrogen bonds. These two amino acids are located in the S2 region, which probably affects cell fusion and RNP release. The docking results of the SARS-CoV-2 spike protein showed that lotoside II formed three hydrogen bonds with Glu145, Glu402, and Arg518. Hydroxygenistein methyl ether malonylhexoside bound to Glu145, His345, and Ala348 with three hydrogen bonds, and formed two pi-pi interactions with His345 and Phe504. The 402 position is located in the active site of the cell receptor binding, which impairs proper binding to the hACE2 cell receptor. Position 145 in the N-terminal domain NTD directly affects viral replication rate56 and contributes to spike trimer assembly57. 345 and 348 residues are located in the heparin binding site in the receptor binding domain RBD, affecting the binding of the ACE2 receptor by controlling the opened or closed RBD position and exposure of RBD to the receptor58. However, as with the influenza targets, these docking results should be viewed as hypothesis-generating rather than confirmatory. They provide a computational basis for selecting candidate compounds for further study, but they do not demonstrate direct inhibition of receptor binding, fusion, or viral entry in the absence of orthogonal functional assays.
Taken together, the present findings show that Ziziphus spina-christi leaf and fruit extracts contain diverse bioactive constituents and exhibit measurable in vitro antiviral activity against H1N1, MERS-CoV, and SARS-CoV-2. Among the identified constituents, lotoside II and hydroxygenistein methyl ether malonylhexoside emerged as promising candidate compounds based on docking analysis. However, the contribution of these individual compounds to the observed extract-level antiviral activity remains to be experimentally confirmed. Therefore, while the current findings support the potential of Ziziphus spina-christi as a source of antiviral candidates, further studies are needed to isolate the active constituents, validate their molecular targets, and clarify their mechanisms of action in relevant biological systems.