It is easy to forget how improbable you are. Four billion years ago, Earth was a barren planet. Its oceans contained simple carbon-based chemicals, but nothing that could grow or adapt.
Then, somehow, chemistry performed a remarkable trick: a molecule began to make copies of itself. Those copies were occasionally imperfect. And in those imperfections lay the possibility of change.
From that modest beginning came evolution — and from that, everything alive today.
For decades, scientists have suspected that this ancestral molecule was a strand of ribonucleic acid (RNA). Now, in a Cambridge laboratory, researchers have provided perhaps the clearest glimpse yet of what this first spark of life looked like.

Philipp Holliger oversees the team at the MRC Laboratory of Molecular Biology
TIMES MEDIA LTD
Unlike DNA, which mainly stores genetic information, or proteins, which mostly carry out the work of cells, RNA can do the work of both DNA and proteins. It can store information in its sequence, and it can fold into shapes that carry out chemical reactions. In theory, life may have begun when a single RNA molecule acquired the ability to replicate.
However, that idea has proved difficult to demonstrate. The RNA molecules known to copy other RNA strands have generally been large and structurally intricate — consisting of 150 to 300 basic building blocks, known as nucleotides. That complexity poses a problem. Molecules that large are hard to copy, even under carefully controlled laboratory conditions. Expecting one to arise spontaneously on the early Earth is a bit like expecting a Boeing 747 to assemble itself as a whirlwind tears through a junkyard.
Now a team overseen by Philipp Holliger at the MRC Laboratory of Molecular Biology in Cambridge has identified a much smaller candidate: a tiny RNA enzyme made of just 45 nucleotides. In keeping with its size, they have called it “Quite Tiny 45”, or QT45.
A molecule that small begins to look chemically plausible — something that might have assembled by chance when Earth was young.
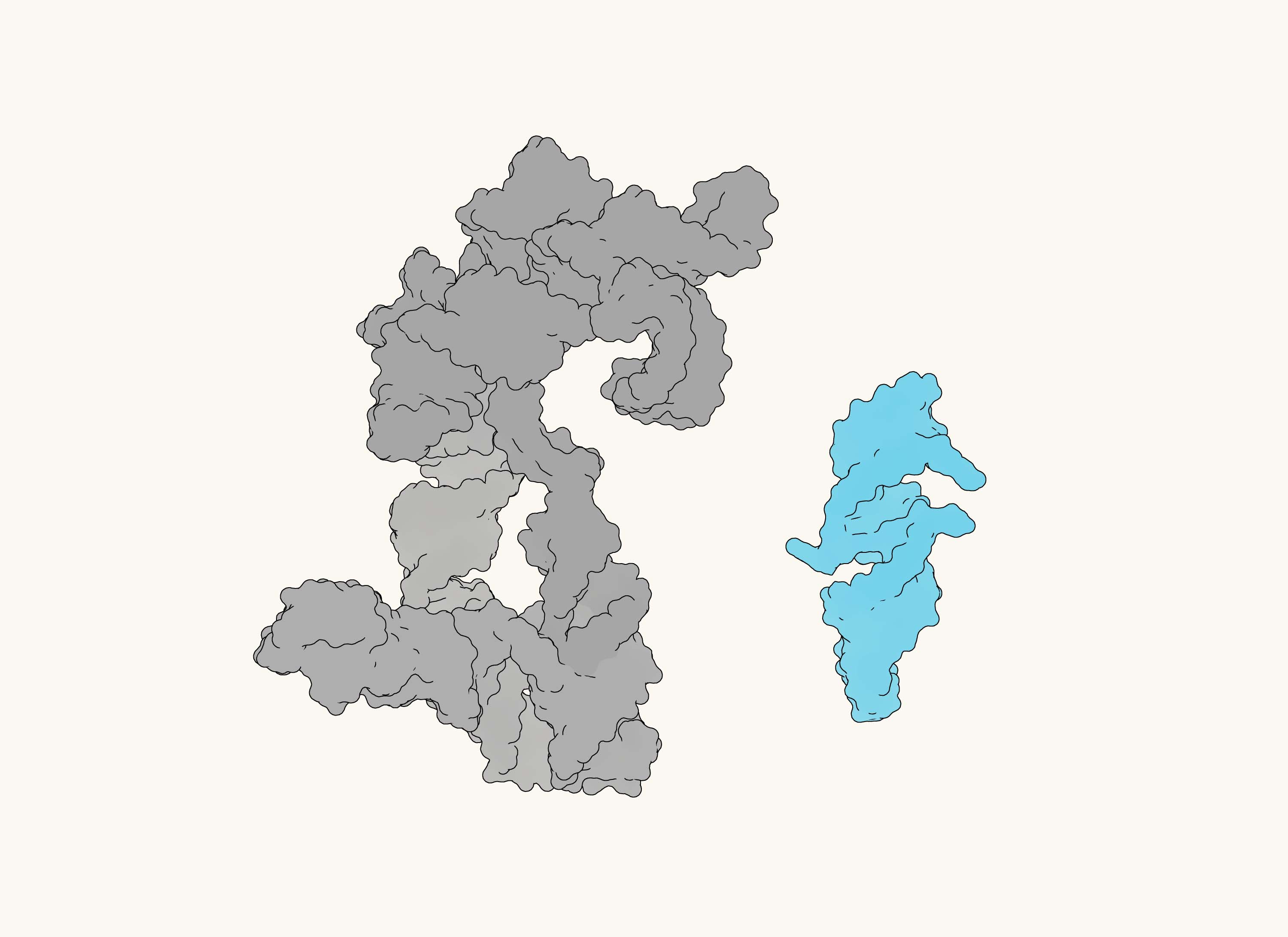
A size comparison between a previously discovered RNA structure and QT45
EDOARDO GIANNI
It narrows the gap between chemistry and biology in a way that origin-of-life researchers have been chasing for decades. “It is the first RNA molecule that can, in a true sense, make copies of itself,” said Holliger.
“It was critical to demonstrate this particular step because this is really the cornerstone of what had become known as the RNA world hypothesis, the idea that life really starts when RNA begins to make copies of itself — to self-replicate, to mutate and then to evolve.”
When he first saw the results, he did not believe them. “The word he used was ‘flabbergasted’,” said Edoardo Gianni, who led the experiment.
• Controversial project sets out to create synthetic human DNA
To find QT45, they created an enormous collection of short, random RNA sequences — trillions of different molecules — and repeatedly selected the rare ones that could join together small RNA fragments. After many rounds of refinement, one candidate stood out. Even when trimmed down to just 45 units, it retained strong copying ability.
Intriguingly, QT45 works best in mildly alkaline, icy conditions — a setting likely to have been seen on the early Earth. As water freezes, narrow channels of chemical-rich brine form between ice crystals. QT45 thrives in these tiny, slushy crevices, acting like a molecular welder. It scavenges short scraps of RNA floating nearby and pieces them together to build a new RNA strand.
A rendition of QT45 in a frozen environment and, below, ice crystals
ELFY CHIANG, MICROSCOPY IMAGE BY JAMES ATTWATER

JAMES ATTWATER/FEDERICO SCAGLIA
Crucially, it can carry out both of the steps needed for self-copying. First, it can make a kind of mirror image of its own nucleotide sequence. Separately, using that complementary strand as a template, it can assemble a new copy of itself.
The reactions are slow and inefficient. After weeks in the freezer, only a tiny fraction of molecules complete the full process. And the two steps must still be run separately in the lab; QT45 does not yet sustain a continuous, self-driven cycle.
But the essential loop is there: a molecule this small can, in principle, produce both its mirror strand and a fresh version of itself.

Edoardo Gianni and Philipp Holliger
JASON BYE FOR THE TIMES
Moreover, while it works most efficiently with building blocks that are three nucleotides long (called triplets), it is not picky. The researchers found it is chemically flexible — or “promiscuous” — meaning it can also build new RNA using single units, pairs or even longer chunks.
This trait is important because the early Earth was home to a chaotic primordial soup, filled with a messy mixture of different chemical fragments. For life to start, a molecule needed to be able to grab whatever was available.
As Holliger explained, the discovery suggests that RNA molecules capable of basic self-copying may be simpler and perhaps more common than previously thought. “We do not yet know exactly how QT45 works — but we are sure there must be other similar molecules,” he said.
The implications are far-reaching. Was life on Earth a fluke, or an outcome of chemistry likely to be seen elsewhere? If the step from simple molecules to self-copying systems can be taken by something as small as QT45, the transition may not be quite as rare as once imagined.

