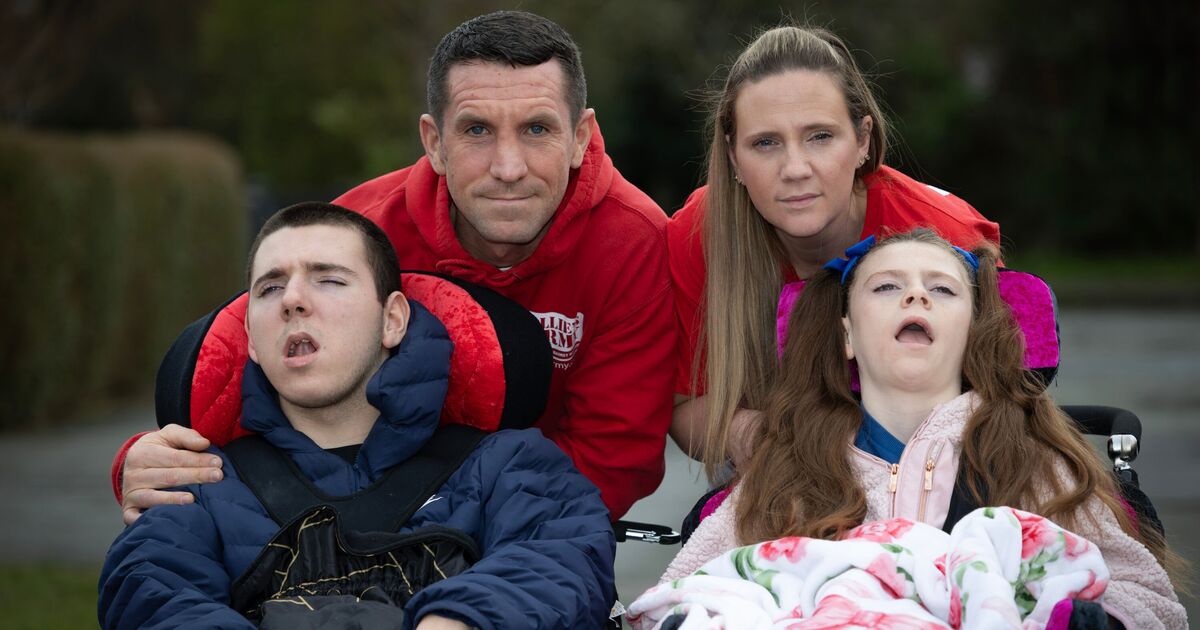
Lucy and Mike Carroll with their children, Ollie and Amelia, who have Batten Disease (Image: Andrew Stenning / Daily Express)
Children diagnosed with a heartbreaking neurodegenerative disease will be left to lose their mobility, speech and young lives after health chiefs ruled against providing life-changing treatment. NHS England currently funds treatment for children like Ollie Carroll, 15, and his sister Amelia, 13, who live with Late Infantile Batten Disease CLN2.
The cruel condition causes seizures and dementia, robbing sufferers of their ability to walk, talk, see, swallow – and causing death on average between the ages of six and 12-years-old. The only reason Ollie and Amelia are still alive is because their parents Lucy and Mike fought tirelessly to maintain access to the treatment which slows down the debilitating nature of the disease.
But committee members of NICE – the National Institute for Healthcare and Excellence – have ruled that children diagnosed from now on will not receive the enzyme therapy, called Brineura (cerliponase alfa). NICE twice refused funding for the drug in the past, but Ollie and Amelia’s parents Lucy and Mike Carroll were instrumental in challenging the decision and won a Supreme Court battle.
Read more: Parents in desperate battle to secure drugs to save their children’s lives

Prince Harry greets little Ollie Carroll at the WellChild Awards in 2016 (Image: PA)
Lucy, 40, said: “My heart hurts for the families of the future knowing the pain they will feel. This news is absolutely devastating. As a family we are in a unique position of knowing how it feels to be told at diagnosis that your child will die, that there is no treatment for your child, as when Ollie was diagnosed in the trial had closed and we were told there was no treatment available.
“We had to fight hard for compassionate use, and we’ve been fighting ever since to have the treatment continued. It will be heartbreaking for parents to know that other children are having treatment, which is slowing the progress of the disease, but that your child won’t receive it because of the cost. To know there is something out there, but you can’t get it for your child is devastating.
“The NHS is giving future families no chance. Data has proven diagnosis has got earlier, that’s been achieved, but treatment started as early as possible could make the difference of their child being able to walk and talk. NICE has said their committee recognises it’s a transformative treatment but won’t fund it, and saying the long-term effects are not known. Define long-term – it’s been ten years.”
Ollie shot to fame in 2016 when he mustered all his strength to stand from his wheelchair to hug Prince Harry at the Wellchild Awards, captured in a touching photograph. The royal later visited him and Amelia in hospital. Brineura, administered via a fortnightly brain infusion, is manufactured by US pharmaceutical company BioMarin, at a cost of £520,000 per child each year.
Commercial negotiations between NICE failed to result in an agreement.
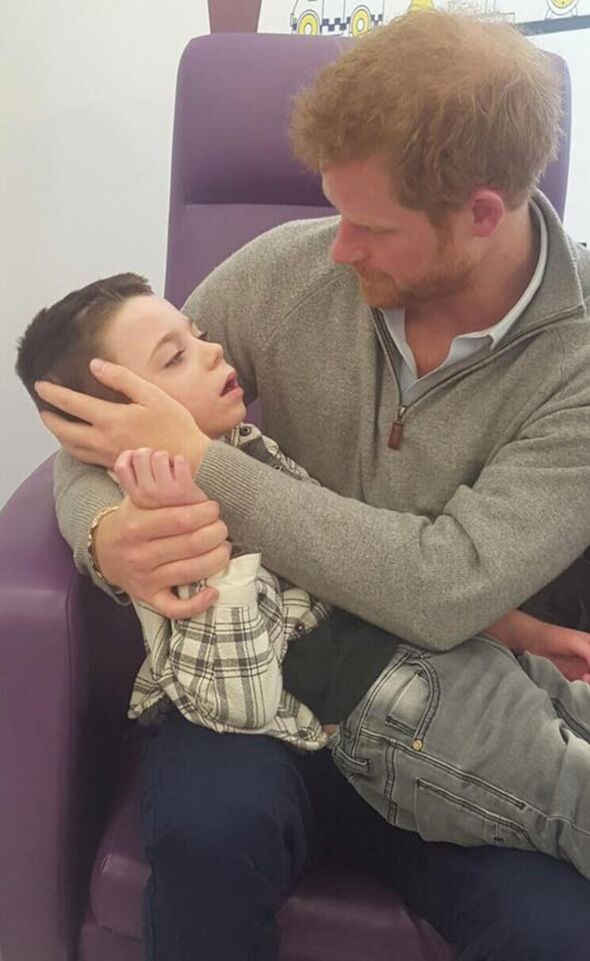
Ollie Carroll with Prince Harry at Great Ormond Street Hospital in 2017, their second meeting (Image: Ollie’s Army)
Now Lucy, from Poynton in Cheshire, fears the decision will also impact the funding of other medicines. “The whole rare disease community should be really worried about this decision,” she said. “Pharmaceutical companies might decide not to trial their treatments because England might not fund the drugs in the end anyway.”
Lucy and Mike, 41, are also parents to Danny, 18, Micky, 17, and six-year-old Renesmee.
Despite fighting hard to give their children access to treatment as ‘patient experts’ sitting alongside campaigners including the Batten Disease Family Association, six months ago they made the heart-wrenching decision to stop Ollie and Amelia’s treatment.
Sharing news with followers of Facebook page Ollie’s Army Battling Against Battens, Lucy stressed that their belief in the treatment had not changed.
She wrote: “Since Ollie and Amelia’s diagnosis they have fought the most incredible battle against this disease, a disease which has robbed them both of their mobility, their ability to play, speak, swallow and has turned their world into complete darkness.
“The strength, resilience and bravery that they have both shown over the years has been nothing short of remarkable… There is only so much you can put your child through…there comes to a point when you realise that you are now doing things to keep them alive for yourself and no longer for them.”
Liz Brownnutt, CEO of the Batten Disease Family Association, said: “This is a heartbreaking and unacceptable decision. Newly diagnosed children with CLN2 Batten disease are being denied the only treatment that can slow this cruel, life-limiting condition. For families, this feels like hope being taken away at the very moment they need it most.
“We are calling on NICE and BioMarin to urgently resolve their differences and agree a fair deal that reflects the real value of Brineura for children and families. The BDFA will not stop fighting until every eligible child can access this treatment. These children cannot wait, time lost can never be recovered.”

The Carroll family at home in Poynton, Cheshire (Image: Andrew Stenning / Daily Express)
Within its published recommendation statement NICE wrote: “The new evidence includes data from clinical trials and from children having treatment in the NHS in England. The clinical trial evidence shows that cerliponase alfa slows progression of CLN2. Patient experts and clinical experts have also explained that cerliponase alfa is a transformative treatment. But there is not much evidence about how well it works long term.
“The committee took into account the condition’s rarity, severity and the effect of cerliponase alfa on quality and length of life. But using the proposed price of the medicine, the most likely cost-effectiveness estimate is not within what NICE considers an acceptable use of NHS resources for highly specialised technologies. So, cerliponase alfa is not recommended.”