After donating their mother’s brain to UCSF, Deborah Kan and Susan Whitaker learned her dementia involved three diseases. With their mom’s UCSF neurologists, they explain what the autopsy revealed and why it matters.
After Deborah Kan’s mother, Alvera Kan, died in December of last year, the family donated her brain to the University of California San Francisco (UCSF) to better understand what type of dementia she had. The answer came back with not just one diagnosis, but three different types of dementia: Alzheimer’s disease, vascular dementia, and LATE.
Kan and her sister, Susan Kan Whitaker, are joined by Dr. Bruce Miller, director of the UCSF Edward and Pearl Fein Memory and Aging Center, and Dr. David Soleimani-Meigooni, a neurologist at UCSF’s Memory and Aging Center and assistant professor focused on precision diagnosis in Alzheimer’s and related neurodegenerative diseases. Miller’s work has helped shape how clinicians identify and distinguish different forms of dementia, including frontotemporal dementia, while Soleimani-Meigooni’s clinical and research work includes using imaging and biomarkers to better understand amyloid, tau and other drivers of cognitive decline.
In this conversation with Kan and Whitaker, Miller and Soleimani-Meigooni discuss how dementia diagnosis can remain uncertain during life and how an autopsy can reveal multiple diseases. They underscores how common mixed dementias are, why symptoms such as getting lost or struggling with numbers can point to specific brain changes, and why better diagnostic tools are needed.
They also highlight the lasting scientific value of brain donation, showing how one family’s decision can contribute to more precise diagnoses, better biomarkers, and, ultimately, more individualized treatment approaches for future patients.
Deborah Kan: Dr. Miller can start by just talking about the diagnostic factors in getting an autopsy. Can you confirm all dementias through autopsy?
Dr. Bruce Miller: Yeah, all the dementias that we know today — I put that equivocal answer. The Alzheimer’s Disease Research Centers (ADRC), which we are part of, tries to autopsy the cause for every patient that is seeking the cause for their dementia. I think that it is increasing our precision, our knowledge in terms of understanding why people develop cognitive impairment as they age.
When I started in this field, it was one thing — everything was Alzheimer’s disease, and it was simple and easy, but it was wrong. Increasingly over time, we have been able to fill in a number of disease categories.
We now know that a lot of people have an entity called LATE, or TDP-43 aggregates. This is really important. It’s new to our field. And we know that this protein, TDP-43, is a very important component of cognitive impairment associated with aging. So the more experience we get with neuropathology, the more we start to understand the many different things that go on in the brain that lead to cognitive impairment over time. But without an autopsy, we miss the cause for many of these etiologies.
Kan: From your experience, Dr. Soleimani, how many of the patients you see end up having mixed dementia?
Dr. David Soleimani-Meigooni: A couple of really outstanding researchers in our group, Dr. Salvatore Spina, who’s a neuropathologist as well as a neurologist, as well as Dr. Renaud Lajoie and a broad number of other researchers at our center — Dr. Lajoie is an imaging specialist who also thinks a lot about neuropathology — they went and looked back at our brain bank, at everyone who had a diagnosis of Alzheimer’s disease. And in our whole brain bank, I think it was 95 percent of people or more with that pathological diagnosis of Alzheimer’s disease also had other pathologies.
For older adults with a diagnosis of Alzheimer’s disease, the median number of pathologies was three. So it’s more the rule than the exception that when a person comes to autopsy and we’re looking at their brain tissue, we see multiple different pathologies that are there and that might be contributing to their symptoms.
Kan: Dr. Miller, do we know yet whether these diseases start separately, or is it that they all converge as the brain deteriorates?
Miller: It’s a very astute question, and it’s really one of the key questions that researchers are trying to answer today. This very strong association between TDP-43 and Alzheimer’s disease makes many of us think that they’re linked.
The same is also true, which was not true in your mother, is that we often see Lewy body pathology, Parkinson’s pathology, associated with Alzheimer’s disease. In the case of TDP-43 and Parkinson’s pathology, I think the co-occurrence is too common to believe that they aren’t linked in some way.
I believe that this is a Gordian knot for our field. We’re not going to be able to treat Alzheimer’s until we really are able to understand and treat TDP-43. I don’t think we’re going to be able to treat Parkinson’s until we can treat the Alzheimer pathology that co-occurs. So this is one of the fundamental questions of the 21st century.
“For older adults with a diagnosis of Alzheimer’s disease, the median number of pathologies was three. So it’s more the rule than the exception…”
Kan: Dr. Soleimani, can you take us through my mom’s diagnosis and try to explain it to people in language that we can all understand.
Soleimani-Meigooni: So what our pathologists do is they basically take a person’s brain, and they take one hemisphere, or one half of the brain, and they have a sampling scheme. So they’ll take little slices of tissue out of very important regions of the brain where these pathologies tend to concentrate. And so they’ll stain these different pieces of tissue, see what pathology is there, and then they have some diagnostic schemes that they can use to assign these different diagnoses.
Another thing that they do is after they look and examine what pathologies are there, they usually contact the neurologist and get a sense of what the history of that person’s cognitive symptoms were during life. And based on that history and progression, in combination with the pathologies that are present and the severity of those pathologies, they then rank them in order based on what they think were the likely contributors to the symptoms.
Kan: Are we looking at right now — is this the order that they’ve presented us in the autopsy?
Soleimani-Meigooni: This is the order. And so, based on the symptoms during life and the severity of what was seen in the brain tissue, they thought that Alzheimer’s disease was the main pathology and the main explanation for the cognitive symptoms during life. Though, of course, we see other changes in the brain that are likely contributing to some degree.
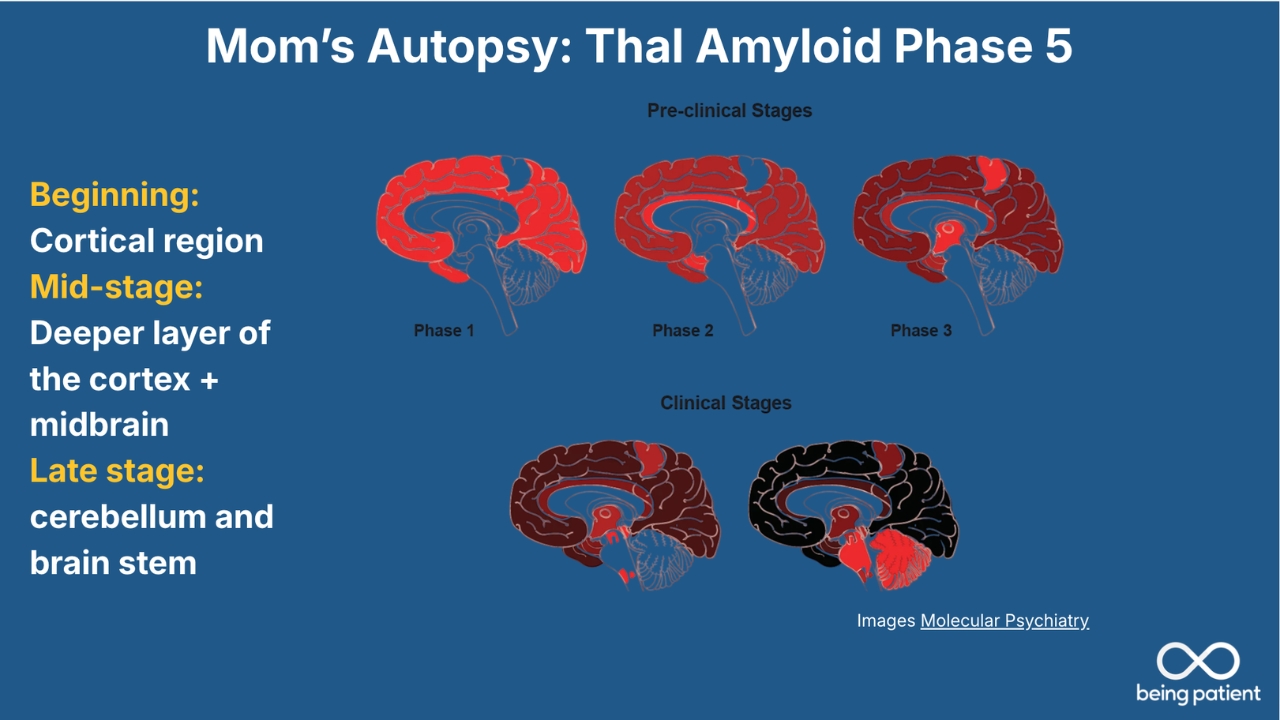
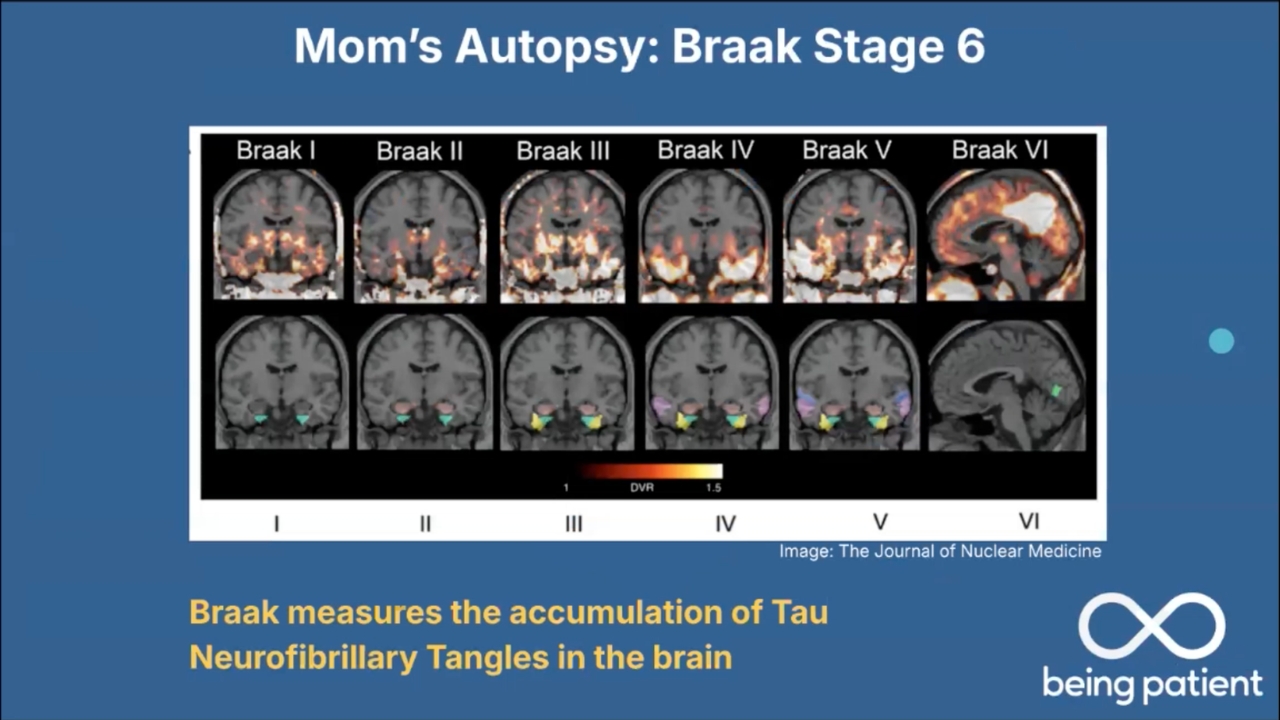
Alzheimer’s disease was number one, and they said high Alzheimer’s disease neuropathological change with A3, B3, C3. There’s a specific criteria that the pathologists use, looking at the location of amyloid plaques, which is this Thal phase, the density of these plaques, which is the CERAD score, and then also the distribution of tau neurofibrillary tangles in the brain. And that’s what the A, B and C stand for.
So, based on these factors, they plug it into an algorithm, and that algorithm says that there is a high degree of Alzheimer’s changes. And that’s essentially what the first point is highlighting.
This [slide] is reflective of those Thal phases that I talked about earlier. So the pathologists look at the distribution of the amyloid plaques throughout the brain. This is based on earlier neuropathological studies where they’ve collected a bunch of brains in people with different severities of cognitive symptoms who have Alzheimer’s changes in the brain and tried to figure out how the amyloid plaques progress.
These begin scattered in outer parts of the brain, what we call the cortex, and then they spread to deeper parts of the brain, like limbic regions around the memory centers, and then eventually to even deeper parts, the brainstem, which helps with autonomic function, and the cerebellum, which is more of a coordination area of the brain.
Kan: This is the amyloid presentation, so plaque, which is usually in the earlier pathology, right? Does plaque come first before tau? Is that correct?
Soleimani-Meigooni: Yes and no. And that’s something complicated that we’re trying to tease out as well. Some people do have a little bit of tau in their brain, beginning in their 40s or 50s. It might be a little bit different than Alzheimer’s disease tau, but it might just stay in sort of the entorhinal cortex in very early temporal areas and not change at all over time. So sometimes people have tau, but it might not necessarily be Alzheimer’s tau early on.
But generally, when we think about the progression of these pathologies, we think of amyloid really setting the stage for tau to spread through the brain. And so you really need amyloid to facilitate that spread of tau.
Kan: So, Dr. Miller, can you clarify which dementias have the presence of plaque and tau and which ones don’t?
Miller: Just a comment on the amyloid first. This particular accumulation of plaques consisting of the amyloid beta protein, that’s pretty specific to Alzheimer’s. And in terms of the staging of this, this is a lot of amyloid. So pretty severe, extensive amyloid across the brain.
To the point of what causes symptoms, we think tau is much more important for the symptoms. The amyloid may be visible many years clinically before we actually see evidence of tau in the brain. Amyloid seems to accumulate in this case extensively, then as David said, it triggers the aggregation of a very specific form of tau in Alzheimer’s disease, the neurofibrillary tangle.
So the neurofibrillary tangle that we see in Alzheimer’s disease, which David has documented, spreads methodically. It starts in the memory systems in the brain, then spreads out into what we call the neocortex. And the symptoms that people have very strongly correlate with the neurofibrillary tangle, this form of tau.
Also specific to Alzheimer’s disease, but tau can aggregate in different ways in many other disorders. So this form of Alzheimer’s disease within a neurofibrillary tangle, which we are very good at characterizing now, is relatively specific to Alzheimer’s disease as well.
“We’re not going to be able to treat Alzheimer’s until we really are able to understand and treat TDP-43.”
Kan: Let’s talk a little bit about the vascular component of this. Our mom had hypertension.
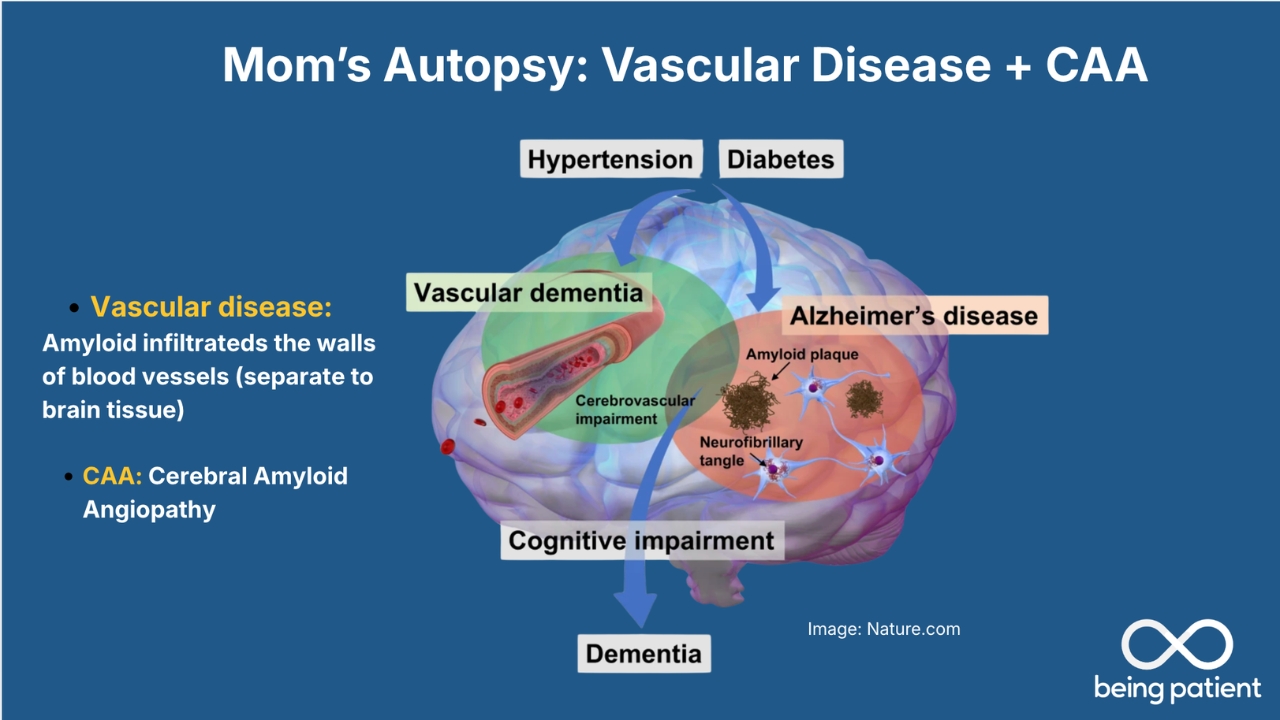
Soleimani-Meigooni: Your mom did have vascular risk factors. There is the hypertension that you mentioned, as well as a little bit of high cholesterol. And people who have that throughout life can get narrowing of small arteries that feed blood and oxygen to the brain, which can cause structural changes and also, to some degree, impact spread and change of animal pathology in the brain.
Another process that was seen on the autopsy, which was shown in the report earlier, is this cerebral amyloid angiopathy. So this is deposition of amyloid in vessels within the brain, which can make those vessels kind of friable and susceptible to things like small bleeds, microhemorrhages. And so this is another very common pathology that can present alongside Alzheimer’s disease.
As Dr. Miller mentioned, we often see TDP-43, specifically LATE. We often see Lewy body disease. We often see regular vascular disease. We also see a lot of cerebral amyloid angiopathy in people who have Alzheimer’s disease.
Kan: We’re hearing from people in our own community at Being Patient, they have now finally qualified for a PET scan because they want to go on one of the monoclonal antibodies that are out there, only to find out they actually have no plaque in their brains. Would that tend to make you both, as neurologists, say, “Oh, it might be LATE dementia,” or do we just not know?
Miller: There are amyloid-negative dementias, which we’re very good at diagnosing now. As you point out, Deborah, we have great blood tests and PET scans that can show us the amyloid associated with Alzheimer’s.
If you have a dementia, but you don’t have amyloid in the brain, then we’re pretty sure it’s not Alzheimer’s disease. And then it requires precision. David is an extraordinary clinician, and he can very precisely go through, well, is it Parkinson’s? Is it Lewy body dementia? Is it LATE? Is it a type of frontotemporal dementia? Is the vascular disease in the brain accounting for cognitive impairment? I think if we don’t see amyloid, that’s when our work really begins.
Soleimani-Meigooni: If there is no amyloid, that’s a big puzzle that we still have to solve,especially early in the course of symptoms. The symptoms for these different neurodegenerative diseases can look the same at an early stage. They can be very vague. This is pure clinical diagnosis, not using some of the neuromolecular biomarkers that we have. But this is relevant because a lot of the neurodegenerative diseases, we don’t have good molecular diagnostic biomarkers for them, so we’re still relying on clinical diagnostic criteria.
These criteria are designed to diagnose people based on the most quintessential features of the disease, which are usually very late symptoms of the disease. And so we become very certain of the diagnosis extremely late in the course of the disease for many non-Alzheimer’s neurodegenerative diseases. This is the big puzzle that we have.
In line with that and TDP-43 — limbic age-related TDP-43 encephalopathy,or LATE — this is a disorder that we’ve known about for a while. Although recently, in 2019, all of the pathologists and people who think about this disorder came to a consensus about naming and the pathological criteria so that we can all get on the same page and really understand this process.
Along with that broader recognition in the neuropathological community, as well as in the clinical community, clinicians started thinking about, well, can we develop criteria for trying to identify LATE during life? And so they looked at a bunch of brains of people who were found to have LATE and then went back and looked at their clinical histories and different tests like MRI or metabolic imaging of the brain. And based on that, they’ve come up with a potential research diagnostic pathway.
LATE can mimic Alzheimer’s disease. The symptoms are very similar: memory symptoms. Some of the things that help differentiate between the two — in LATE, we often see hippocampal atrophy — atrophy in those memory areas of the brain — on MRI that is severe and seems out of proportion to the level of symptoms that people have when they come in for assessment.
So severe atrophy in those memory centers, but very mild clinical symptoms — that’s one clue. And then if an amyloid PET is negative, then that would make you think a little bit more strongly that there’s LATE present rather than Alzheimer’s disease.
Kan: I want to talk a little bit about the symptoms my mom was experiencing at the time of diagnosis, which was in 2013. And then we’re going to look at some of her images to see what was visible then versus what we know now, because I think that will be interesting to look at. Sue, why don’t you talk about back in 2013 when Mom went to see UCSF with a problem, when she knew something was wrong.
Susan Whitaker: There were some very distinct memory issues happening. One was she would get lost quite easily driving around very familiar places.
She lived in San Francisco for 30-plus years and suddenly couldn’t find her way home from the airport, for instance. Or even going to UCSF, where our father worked, parking her car in the garage, not being able to find her car. It was mostly things like that.
She was a very competent, very independent person, and suddenly she started to sort of lose her way in ways that we had never seen before.
Kan: She lost her ability for numbers, which was unusual, like household things, simple things like the house alarm code, she could no longer remember. And we hadn’t changed it since we were kids. And when her ATM card got eaten three times in a week because she couldn’t remember the code. And that’s what signaled it was time to go in and get her checked.
What we just recently found out, which really shocked us, was that now that we have access to her medical records, she had memory concerns in 2008. So that was five years before it was time to see a doctor, which I guess — I see you shaking your head, Dr. Miller. Is that common?
Miller: Yes, I think delay of diagnosis is the rule, not the exception. And I think it’s very troubling to all of us in the field because the treatments that we have are much more likely to be effective when someone is early in their illness.
This is one of the challenges that we face, and having a podcast like this today can really, I think, alert the public about the importance of taking symptoms like trouble with navigation, very commonly associated with Alzheimer’s disease, to your physician.
Kan: Can you tell us what you saw in her brain in 2013?
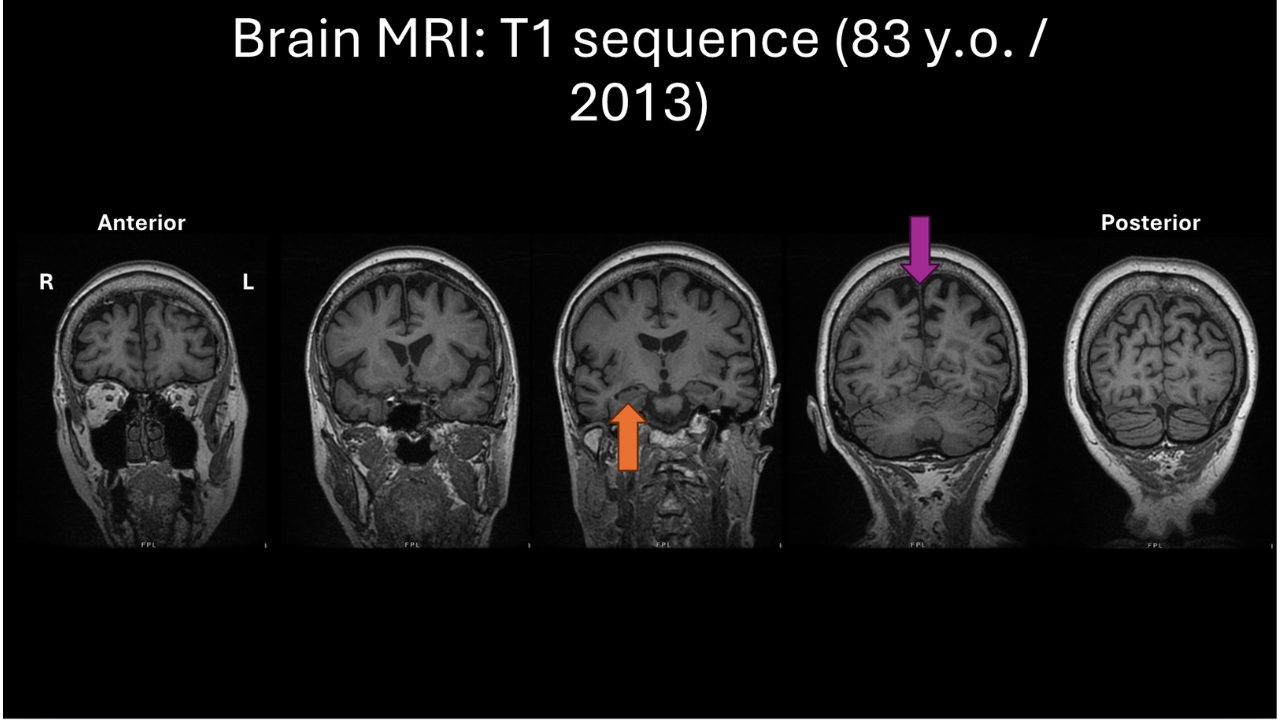
Soleimani-Meigooni: So this is an MRI sequence called T1, which helps us get a sense of the overall structure of the brain and assess structural changes.
Starting with the left side, you can see overall the frontal lobes generally look fairly full and healthy. On the next slide, we can see a little bit of the temporal lobes around the temporal poles, the very tips of the temporal lobes, and a little bit more of the frontal lobes. And maybe there are some mild changes, or volume loss, here.
In this middle slice, you can see the orange arrow is pointing to a main memory structure of the brain called the hippocampus. And there’s some definite thinning, or volume loss, of the hippocampus, and because of that, this adjacent fluid-filled space next to it that everyone has, called the ventricles, looks a little bit wide.
If we look at the rest of the temporal lobes, the tissue around the hippocampus, the folds of the brain there are a little thin, and the spaces between those folds are wider than what we’d like to see for normal healthy aging.
In the next-to-last slice, there’s a purple arrow pointing to the parietal lobes in an area called the precuneus. And this is an area that seems to be targeted a lot by Alzheimer’s disease pathology. And we can see a lot of brain volume changes and atrophy there. And that was certainly present in your mom’s MRI. This area probably stood out the most in terms of the level of atrophy, or volume loss, that was present.
And then further back on the far right-hand side in the occipital lobes, there are just some mild changes. So in neurological parlance, we just call this pattern a temporoparietal pattern of volume loss, or atrophy. And this is one of the patterns that can be associated with Alzheimer’s disease.
Miller: We really are very interested in the first symptoms. And Susan was very precise in telling us that the first symptoms were trouble getting lost. Well, what part of the brain is responsible for that? It’s the right — just exactly where the purple arrow is pointing — the parietal lobe. And the navigation difficulty is telling us that the parietal lobes are not working like they should.
The atrophy is a representation of loss of function back there. This is not 100 percent specific to Alzheimer’s, but very typical Alzheimer’s. So when the history is really good and the clinician takes time to take it properly, our diagnostic capacities dramatically increase.
Kan: Dr. Soleimani, at that time, I think she did off the bat receive a diagnosis of Alzheimer’s disease. From these images, because you can’t see plaque or tangles from an MRI, correct? So what is it about this that would make you suspect it’s probably Alzheimer’s?
Soleimani-Meigooni: At that time [I saw her,] in 2017, when biomarkers weren’t part of routine practice, what we try to do is what Dr. Miller had stated, is we’re looking to, number one, see: Is there a progressive course of cognitive symptoms? Does this progression in the type of symptoms fit a pattern of what we usually see in Alzheimer’s disease? Are these changes that we see in terms of the symptoms also present in cognitive testing over time?
And then we of course know the localization of these symptoms in the brain on an MRI. And so if we see progressive symptoms with objective evidence of change and a pattern that’s consistent with Alzheimer’s disease, and an MRI that’s supportive, that was what we were using to diagnose Alzheimer’s disease at that time.
Kan: Can you tell me what we’re looking at right now?
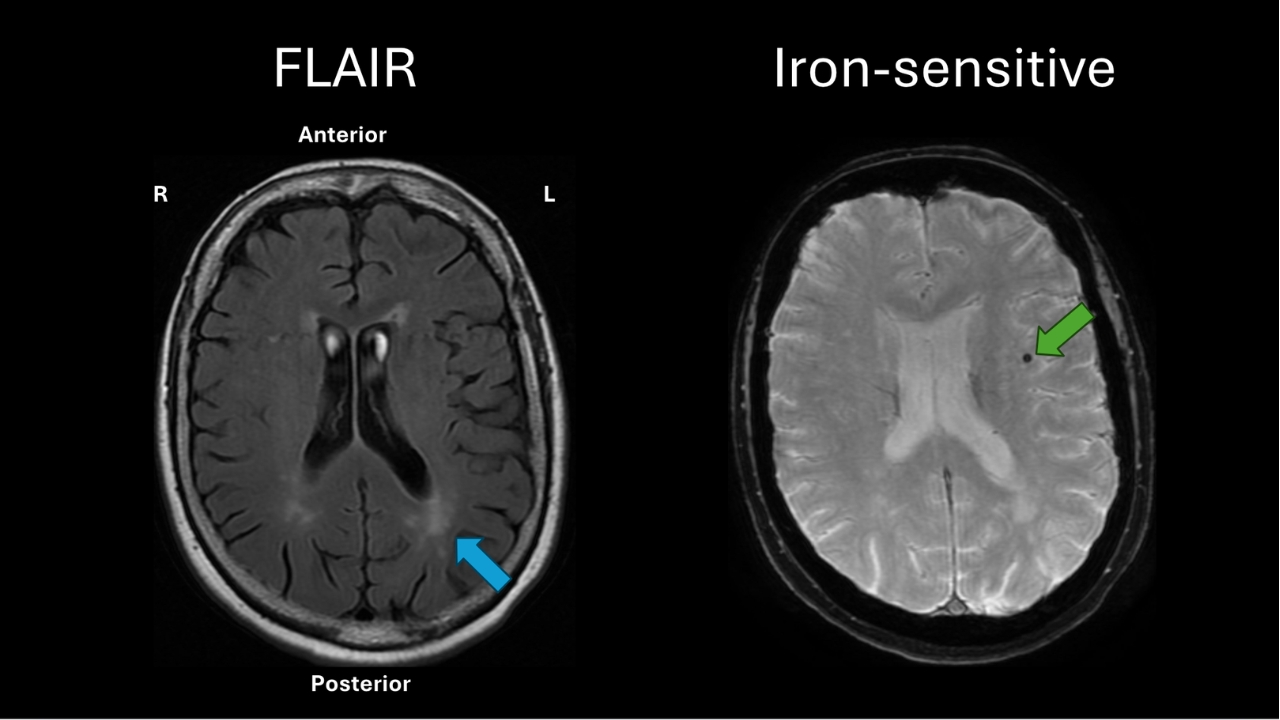
Soleimani-Meigooni: With MRI, there are other sequences that come with it where we’re looking at other structural changes in the brain. On the left is a sequence called the FLAIR sequence, and I’m just showing a section called an axial slice that goes right through the middle of the brain. And what this axial FLAIR slice is showing is that you can see these hazy, patchy, white-looking appearances. Right where the blue arrow is. And these changes are a sign that’s often associated with vascular changes in the brain, so narrowing of small arteries in deep parts of the brain.
And then on the right-hand side is one single axial section from an iron-sensitive sequence. And so our blood has hemoglobin, which is a molecule that’s made up of iron. And so if there’s ever been any small bleeds inside of the brain, it basically leaves an iron deposit that we can pick up with this sequence. And there was one iron deposit shown here with a green arrow in an area of the brain called the insula.
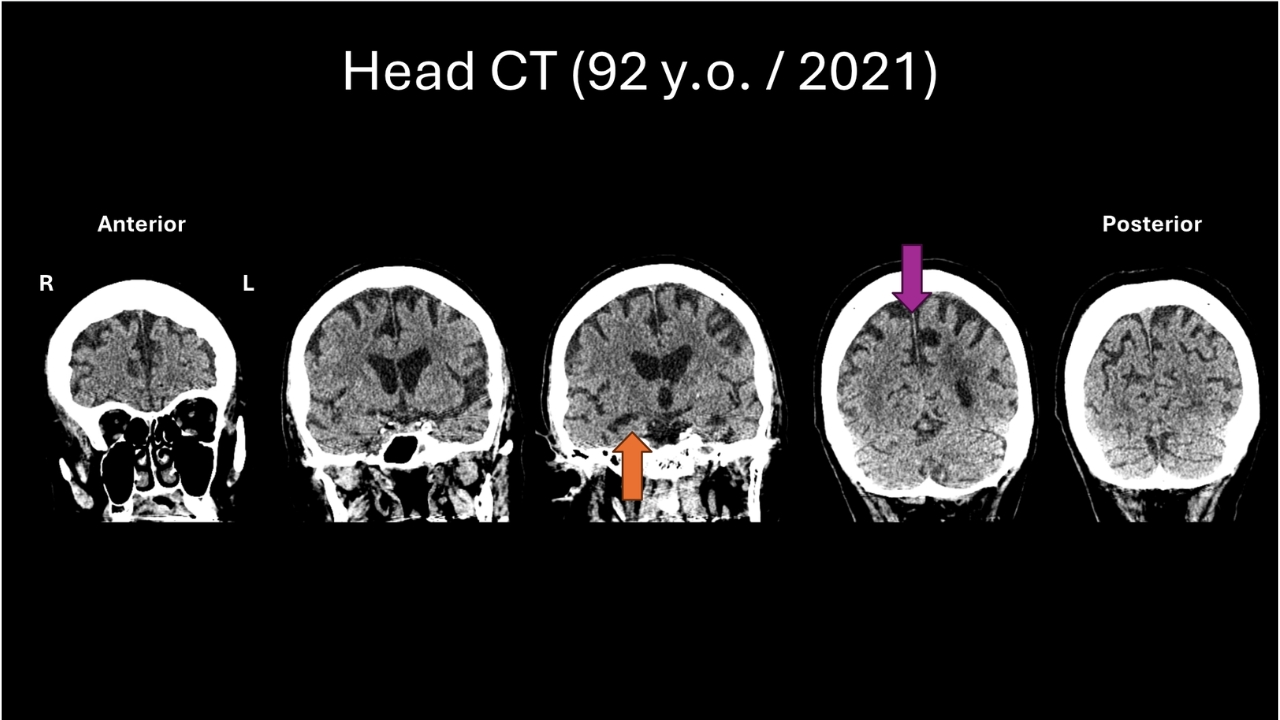
Deborah Kan: This was a CT scan that our mother got in 2021. So now that we have these images, what do you see? Because now this is several years after you first saw her.
Soleimani-Meigooni: Definitely. We can’t make a perfect apples-to-apples comparison between an MRI and CT. But despite that, we can still compare the scans. And what you notice, the orange arrow is pointing to the memory center, and you can see that it looks smaller than it did before. And then the purple arrow is pointing to those parietal regions, and you can see that they look smaller than before.
And so certainly comparing the previous scan to the most recent head CT, it does appear that there is progression of volume loss over time, especially in those areas that were affected most early on, and still consistent with the pattern that we usually see in Alzheimer’s disease.
Kan: Dr. Miller, is volume loss in the brain associated with cognitive decline?
Miller: Not necessarily. We sort of have a sliding scale, for lack of a better word, for aging. And we know that as people get older, there tends to be a loss of tissue, so the cortical thickness becomes less. But it doesn’t necessarily mean that someone is having cognitive impairment.
In the case of Alzheimer’s disease, the strongest correlate of cognitive impairment is whether there are neurofibrillary tangles. And those are within the neurons. They reflect marked disruption of the way our neurons are working. Atrophy can be benign, but it also, when it’s deviations outside of the norm, is much more likely to be associated with a neurodegenerative disease.
I’ve got to say, looking at this CT scan, would it have been possible for someone with this CT scan to have normal cognition? I would say yes. But on the other hand, as David points out, the progression of the atrophy and the more severe atrophy in the parietal lobes is very suggestive of Alzheimer’s.
Kan: Dr. Miller, tell me about the different dementias. If you’re diagnosing FTD, is that something on the scan that you can say, oh, that’s definitely FTD? Same thing with Lewy body. Tell me a little bit about how visible that is.
Miller: Two comments. One is, as you pointed out, our biomarkers, like the blood tests for Alzheimer’s, have gotten really good. So I think our diagnostic precision for ruling in Alzheimer’s or ruling it out has gotten extremely good.
We’re not at that place with frontotemporal dementia or the Parkinson’s-Lewy body dementias. Generally, I think clinically, the picture can guide you to the right diagnosis. We have an AI program that we’re developing, and it can differentiate FTD from Alzheimer’s about 90 percent of the time. So I think the atrophy is different. It’s more in the front, not in the back. The symptoms are different. They tend to be more behavioral changes, certainly not the case in your mom. Those are the first symptoms.
With Lewy body, there’s such a high co-occurrence with Alzheimer’s. I’m not surprised that some of your listeners are bringing this up. And sometimes it’s very subtle. Sometimes it’s a loss of smell. Sometimes the first manifestation is a visual hallucination. Sometimes the first manifestation is the handwriting getting smaller. We call this micrographia.
I think, because of the generosity of your mom and your family and other people in the audience, that we are going to be able to diagnose with biomarkers each one of these pathologies. I’d be surprised if in 10 years, we couldn’t tell you before an autopsy, during life, what the pathologies are. But right now, all we have are two things, really. It’s the clinical symptoms and the MRI pattern of atrophy. With Alzheimer’s, we’re better.
Deborah Kan: I want to know what’s going to happen with her brain. What does this mean for you? What is this going to enable you to do that maybe you couldn’t do? And how long will her brain be used for? What happens after we get this autopsy report?
Miller: I think our fellows at UCSF are extraordinary, and only in the last year have our fellows started to really push me about whether our patients have LATE or not. This autopsy will be incredibly important for the entire community.
Next on the docket of researchers is to try to develop a better biomarker, try to understand why it co-occurs with Alzheimer’s disease. Do the people who have TDP-43 along with Alzheimer’s pathology respond less to an amyloid-lowering antibody? These are some of the big questions that this autopsy reminds us of.
This tissue will be available for the best researchers in the world for many, many years. So I think as we solve the mystery of TDP-43, cerebral amyloid angiopathy, atherosclerosis of the brain, this will be a tissue that will help inform and help future generations.
Kan: So, Dr. Soleimani, maybe you could talk about it from the diagnostic perspective, because now you must see other cases like this. We donated our mom’s brain, and now you can compare, because you were with her at an earlier stage. So what does that mean for diagnostics?
David Soleimani-Meigooni: We need better diagnostics. That’s really the truth. And to build upon what Dr. Miller was saying, the extraordinary gift of your mom’s brain tissue can be used in hundreds of studies, studies that are going to hopefully help develop biomarkers for diagnosis of limbic age-related TDP-43, or LATE, cerebral amyloid angiopathy, these comorbid degenerative disorders.
And once we have reliable diagnoses — usually it starts with PET, and then PET serves as the basis for accelerating development of blood biomarkers — then hopefully we’ll have blood tests where during life we can be sure of the degenerative pathologies that a person has that are contributing to their symptoms, and then we can do targeted interventions for each of these pathologies. We really want to get to the point of individualized precision medicine for all the things that might be driving the cognitive symptoms. Once we get to that point, I think that’s where we’ll make the greatest impact in slowing or hopefully stopping the progression of these symptoms.