Snake bites are a global health crisis that rarely makes headlines. According to the World Health Organization and researchers in the field, between 80,000 and 100,000 people die from snake bites every year, while around 300,000 others are left with permanent disabilities. These numbers paint a grim picture of a danger that disproportionately affects rural and low-income populations who have limited access to antivenoms, treatments that are currently developed species by species, making broad protection both logistically and financially out of reach for much of the world.
Fear of snakes is widespread for good reason. An estimated one-third of adults worldwide suffer from ophidiophobia, the clinical term for that fear. And yet, Tim Friede, a self-described self-taught venom expert and now director of herpetology at the biotech company Centivax, is not among them. His two-decade experiment in deliberate envenomation, which began as a personal obsession, has evolved into what may be one of the most consequential contributions to toxinology in recent memory.
A Methodical, If Dangerous, Process
Friede’s approach was anything but reckless in its structure, even if the risks were enormous. He would first milk venomous snakes to extract their venom, then inject it into his own body, gradually increasing the dosage over time. The goal was to give his immune system the opportunity to develop antitoxin antibodies strong enough to neutralize future bites. Without that slow buildup, as he acknowledged, he would have died many times over.
 Tim Friede – © Centivax
Tim Friede – © Centivax
Once he judged himself sufficiently immune, Friede moved to direct snake bites, a live test of whether his body had truly adapted. It did not always go as planned. On one occasion, simultaneous bites from an Egyptian cobra and a monocled cobra sent him to the hospital, where he remained in a coma for four days. Reflecting on these moments, Friede was blunt in his assessment, telling Science News: “It always burns and it’s always, always painful. Was it a mistake? Yes. Was it stupid? Yes.”
Over twenty years, he built up immunity to some of the world’s most lethal species, including black mambas, king cobras, and tiger snakes.
A Once-in-a-Lifetime Immune History
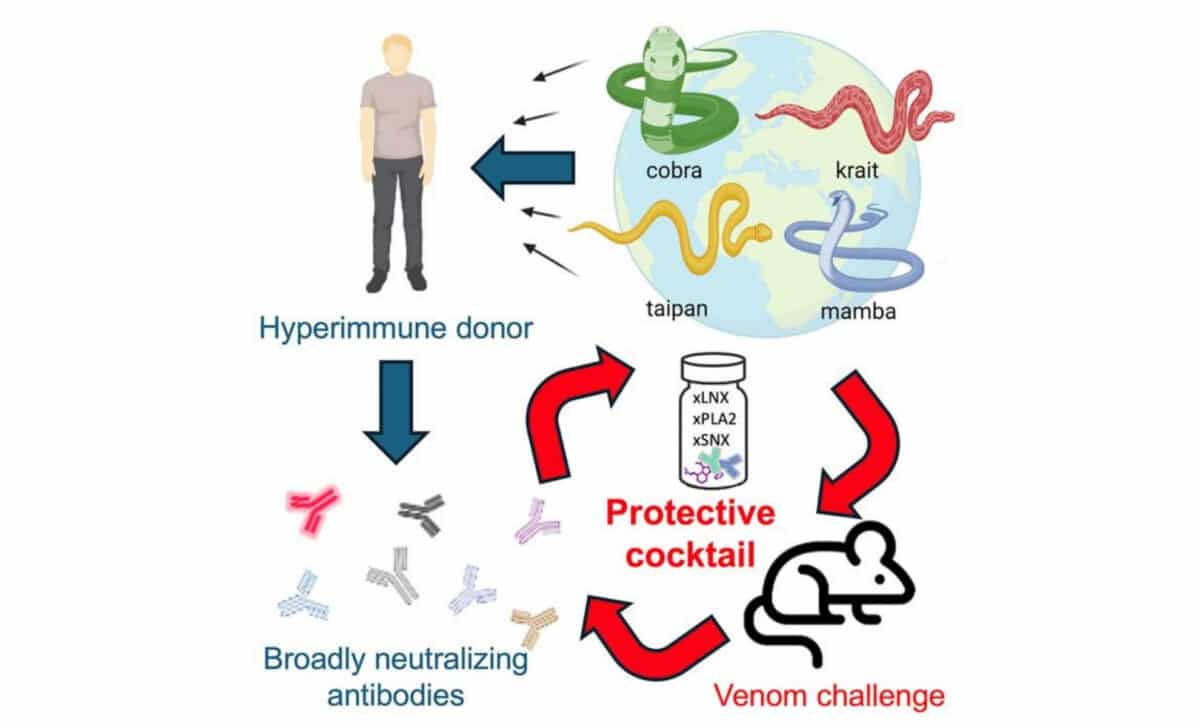
The scientific opportunity Friede represented did not go unnoticed. Jacob Glanville, lead author of the resulting study and CEO of Centivax, explained the significance of Friede’s biology in a news release. “What was exciting about the donor was his once-in-a-lifetime unique immune history,” Glanville said. “Not only did he potentially create these broadly neutralizing antibodies, in this case, it could give rise to a broad-spectrum or universal antivenom.”
 Graphical abstract – © Glanville et al. / Cell
Graphical abstract – © Glanville et al. / Cell
According to the study, published in 2025 in the journal Cell, Centivax developed an antivenom cocktail by isolating two of Friede’s antibodies, identified as LNX-D09 and SNX-B03, and combining them with varespladib, a toxin-blocking drug. When tested on mice, the cocktail provided complete protection against 13 snake species and partial protection against six more. The results represented a significant step toward what scientists had long considered a difficult goal: a single antivenom formulation capable of working across a wide range of venomous species, rather than requiring a separate treatment for each one.
What Comes Next
The research is now moving into the next phase of testing. As reported by Popular Mechanics, the Centivax team plans to begin clinical trials in Australia, working with dogs brought in for snake bite injuries in real-world settings. The researchers also have their sights set on expanding the treatment to cover vipers, a separate and equally dangerous family of venomous snakes not yet addressed by the current formulation.
Peter Kwong, a lead author of the study from Columbia University, described the ongoing work in a press statement: “We’re turning the crank now, setting up reagents to go through this iterative process of saying what’s the minimum sufficient cocktail to provide broad protection against venom from the viperids.”
The implications of this research extend well beyond one man’s unusual immune system. If a truly broad-spectrum antivenom can be developed and made accessible, it could transform how the global health community responds to one of its most overlooked emergencies, saving tens of thousands of lives annually, and sparing hundreds of thousands more from life-altering disabilities.

