Your support helps us to tell the story
From reproductive rights to climate change to Big Tech, The Independent is on the ground when the story is developing. Whether it’s investigating the financials of Elon Musk’s pro-Trump PAC or producing our latest documentary, ‘The A Word’, which shines a light on the American women fighting for reproductive rights, we know how important it is to parse out the facts from the messaging.
At such a critical moment in US history, we need reporters on the ground. Your donation allows us to keep sending journalists to speak to both sides of the story.
The Independent is trusted by Americans across the entire political spectrum. And unlike many other quality news outlets, we choose not to lock Americans out of our reporting and analysis with paywalls. We believe quality journalism should be available to everyone, paid for by those who can afford it.
Your support makes all the difference.Read more
A batch of blood pressure medication is being recalled after a potential manufacturing error meant some packets may contain the wrong dose.
The Medicines and Healthcare Products Regulatory Agency (MHRA) warned batches of Ramipril 10mg capsules, manufactured by Crescent Pharma Limited, may contain blister strips of a lower dose, specifically Ramipril 5mg, due to a potential manufacturing error.
It comes after a patient found one blister pack of Ramipril 5mg capsules inside a sealed carton of Ramipril 10mg capsules.
Both product batches were manufactured at the same site, and the error appears to have occurred during secondary packaging of the cartons.
The regulator has advised pharmacy and healthcare professionals to stop supplying the impacted batch of medication and return all remaining stock to their suppliers.
 Batches of a blood pressure medication are being recalled as some packs contain the wrong dose (PA)
Batches of a blood pressure medication are being recalled as some packs contain the wrong dose (PA)
However, the risk to patients taking the lower dose of this medicine for a short time is very low, the MHRA said.
Ramipril is taken by millions of people worldwide to treat high blood pressure and heart failure. It’s also prescribed after a heart attack.
The medication helps to prevent strokes, heart attacks and kidney problems by widening the blood vessels which makes it easier for the heart to pump blood around the body.
Dr Alison Cave, MHRA chief safety officer, said: “If you take Ramipril 10mg, check the packaging for batch number GR174091. The batch number and expiry date information can be found on the outer carton.
“If you have received this batch, check that the medication name on the carton matches the blister strips inside.
“If the 10 mg carton of Ramipril contains blister strips that are labelled as Ramipril 5mg capsules, contact your dispensing pharmacy.
“If the carton contains blister strips that are correctly labelled as Ramipril 10mg capsules, you do not need to take further action.”
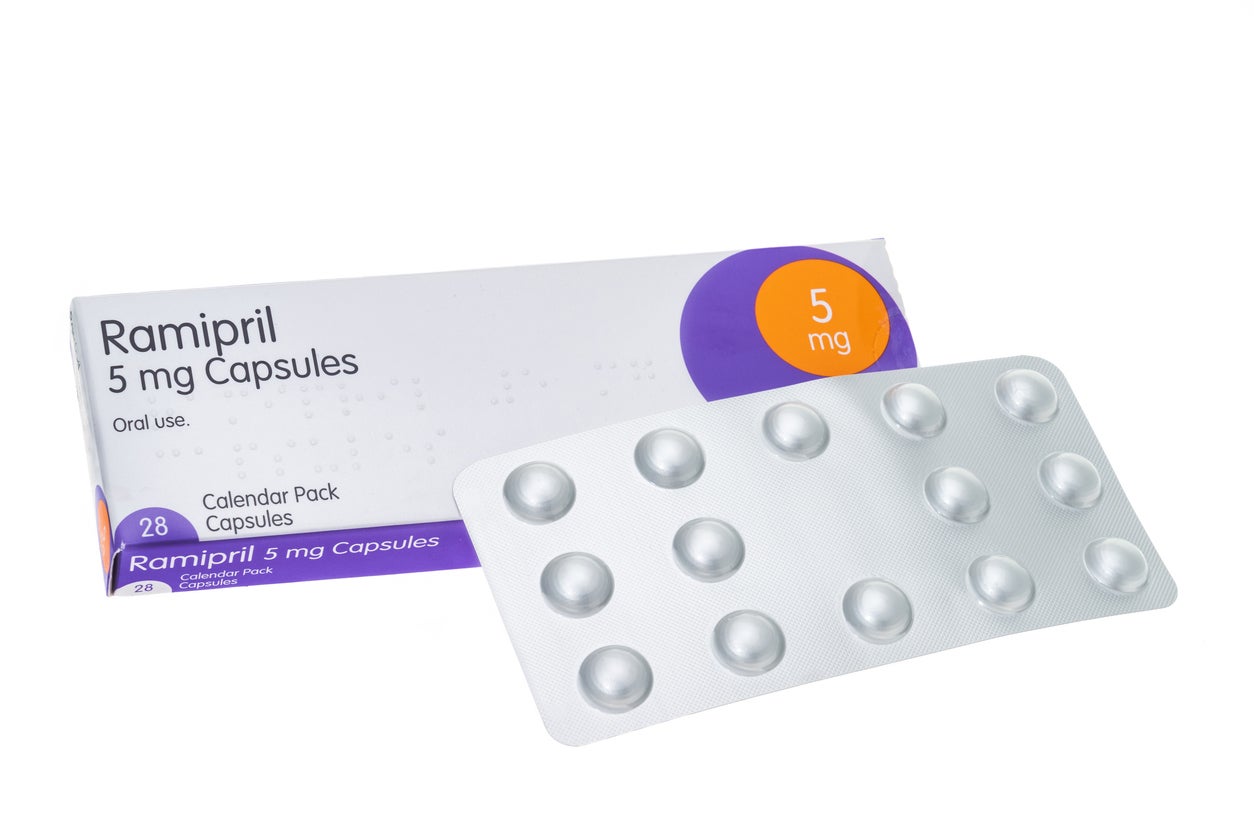 MHRA warned batches of Ramipril 10mg capsules, manufactured by Crescent Pharma Limited, may contain blister strips of a lower dose, specifically Ramipril 5mg (Getty/iStock)
MHRA warned batches of Ramipril 10mg capsules, manufactured by Crescent Pharma Limited, may contain blister strips of a lower dose, specifically Ramipril 5mg (Getty/iStock)
Those who are experiencing adverse effects after taking the lower doses that were packaged in error are being urged to seek medical advice.
But the MHRA reassured that there is a very low risk to health from taking a lower dose than prescribed.
Both strengths of the medication are used to treat high blood pressure, heart failure, and kidney disease.
Any possible impact of a lower dose of Ramipril is expected to be gradual rather than immediate or life threatening.
Depending on why a patient has been prescribed ramipril, the usual starting dose is between 1.25mg and 2.5mg once a day, to ensure it does not make the patient feel dizzy.
The dose is usually increased gradually over a few weeks with the maximum dose being 5mg twice a day or 10mg once a day.

