Thousands of patients have been given a “glimmer of hope” of accessing revolutionary Alzheimer’s drugs on the NHS after the medicine adviser said it would reconsider approving them.
The first two drugs to treat the underlying cause of Alzheimer’s disease, called lecanemab and donanemab, were rejected for use on the NHS in England and Wales in June.
At the time the National Institute for Health and Care Excellence (Nice) said the benefits of the drugs were “too small” to justify the cost. However, Nice has said it will now re-examine the evidence after the drugs’ manufacturers won an appeal.
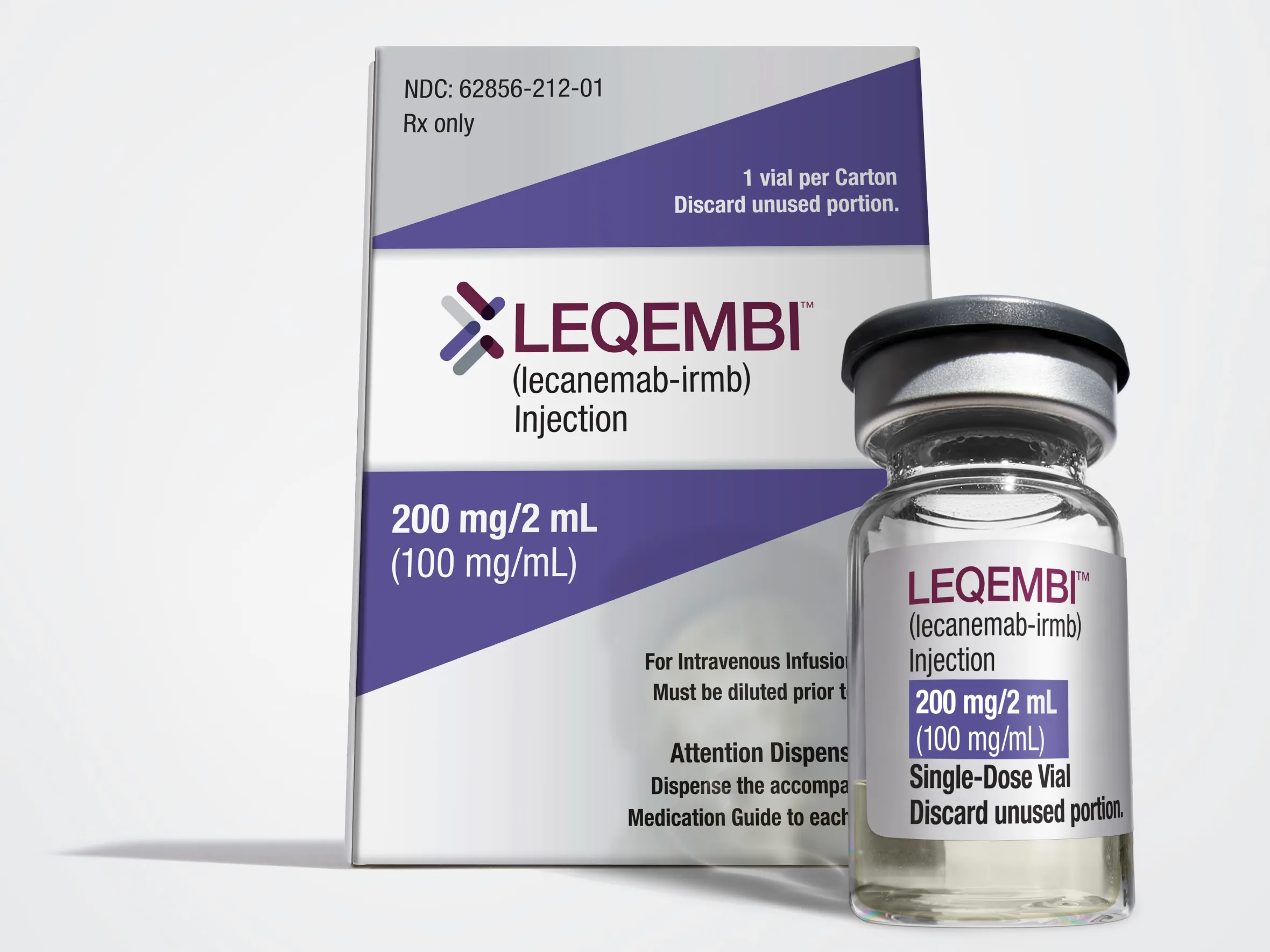 One of the drugs being reconsidered for NHS approval
One of the drugs being reconsidered for NHS approval
The appeal argued that Nice’s appraisal process had failed to take into account the “full value” of the drugs for the economy. This includes the benefits in relieving costs on caregivers and the wider NHS and care system, as well as improving the quality of life of patients and families.
About 70,000 patients in England in the early stages of Alzheimer’s could benefit from the drugs if they are approved on the NHS. The treatments, given as a drip, are available privately in the UK, costing about £60,000 a year.
Chris Stokes of Lilly, which makes donanemab, said: “This is a big moment in the battle against Alzheimer’s disease. Nice was right to look again at the evidence in front of them and it’s welcome that our appeal has been upheld. For people living with Alzheimer’s disease, and for the families and carers who support them, this matters — perhaps now more than ever.”
 Chris StokesJulian Benjamin for the times
Chris StokesJulian Benjamin for the times
Eisai, which makes lecanemab, said: “This news offers a welcome glimmer of hope for the Alzheimer’s disease community in England.
“We are keen to work quickly with Nice to assess this treatment effectively, because while we wait, Alzheimer’s disease does not.”
The two drugs are the first to treat the underlying cause of Alzheimer’s disease by clearing toxic amyloid protein from the brain, delaying cognitive decline by four to six months. However, they have dangerous side-effects, including a risk of brain swelling and bleeding.
More than 130 drugs are in development for Alzheimer’s around the world and the NHS is being urged to prepare for a new era of dementia treatment.
Michelle Dyson, the chief executive of the Alzheimer’s Society, said: “We welcome Nice’s decision to look again at the case for lecanemab and donanemab, particularly at the significant impact of dementia on unpaid carers. We know that carers are too often pushed to breaking point and this needs to be properly recognised.
“People living with dementia are desperate for new treatments that delay the progression of symptoms and help them stay independent for longer. The science is moving fast and globally more people are starting to access these drugs, but the UK is falling behind. With over 30 Alzheimer’s disease drugs in late-stage clinical trials, there are likely to be more treatments submitted for approval soon.
“The government must be ambitious about preparing for new treatments in their upcoming dementia plan. Time is ticking and it’s important that people with dementia in the UK don’t miss out.”
Helen Knight, director of medicines evaluation at Nice, said: “The independent appeal panel has ruled that some elements the committee used to inform its decision-making in relation to the cost-effectiveness of donanemab and lecanemab need further consideration.
“So today we have announced we will give stakeholders an opportunity to provide more information for the committee to help it address the areas of continuing uncertainty highlighted by the appeal panel.
“Throughout our assessment of donanemab and lecanemab we have done everything we possibly can to try and achieve a positive outcome, including providing an additional opportunity for evidence to be submitted.
“Today’s decision underlines our commitment to continuing that process.”