The warty comb jelly is not your typical ocean dweller. It can regenerate lost body parts, reproduce from a larval stage, and even fuse its body with other comb jellies to survive injury. It also lacks a permanent excretory opening, only forming one when strictly necessary. But these traits, extraordinary as they are, have been largely overshadowed by what researchers uncovered in 2024: the animal’s capacity to biologically de-age as a survival mechanism.
What sets this discovery apart from similar phenomena observed elsewhere in the natural world is the deliberate, reactive nature of the process. Unlike Turritopsis dohrnii , the so-called “immortal jellyfish,” which can restart its life cycle from scratch upon reaching maturity as a sort of default biological quirk, Mnemiopsis leidyi appears to use reverse development as an active response to extreme stress. It doesn’t simply reset. It retreats, strategically, when survival demands it.
A Disappearing Adult, the Accidental Spark of Discovery
The study, published in the journal Proceedings of the National Academy of Sciences, was led by Joan J. Soto-Angel, a marine biologist at the University of Bergen in Norway, alongside his colleague Pawel Burkhardt. But the research might never have happened were it not for a peculiar and puzzling moment in the lab.
One day, an adult comb jelly Soto-Angel had been keeping in a laboratory tank simply vanished. In its place was a specimen in its larval state. That observation, strange, almost unbelievable, was enough to convince him there was something worth investigating more rigorously.
It is worth noting that earlier research had already documented adult comb jellies shrinking in size and losing body mass in response to food scarcity. Those studies, however, had explicitly ruled out any genuine age reversal. Soto-Angel’s accidental observation upended that conclusion and set the stage for a controlled experiment.
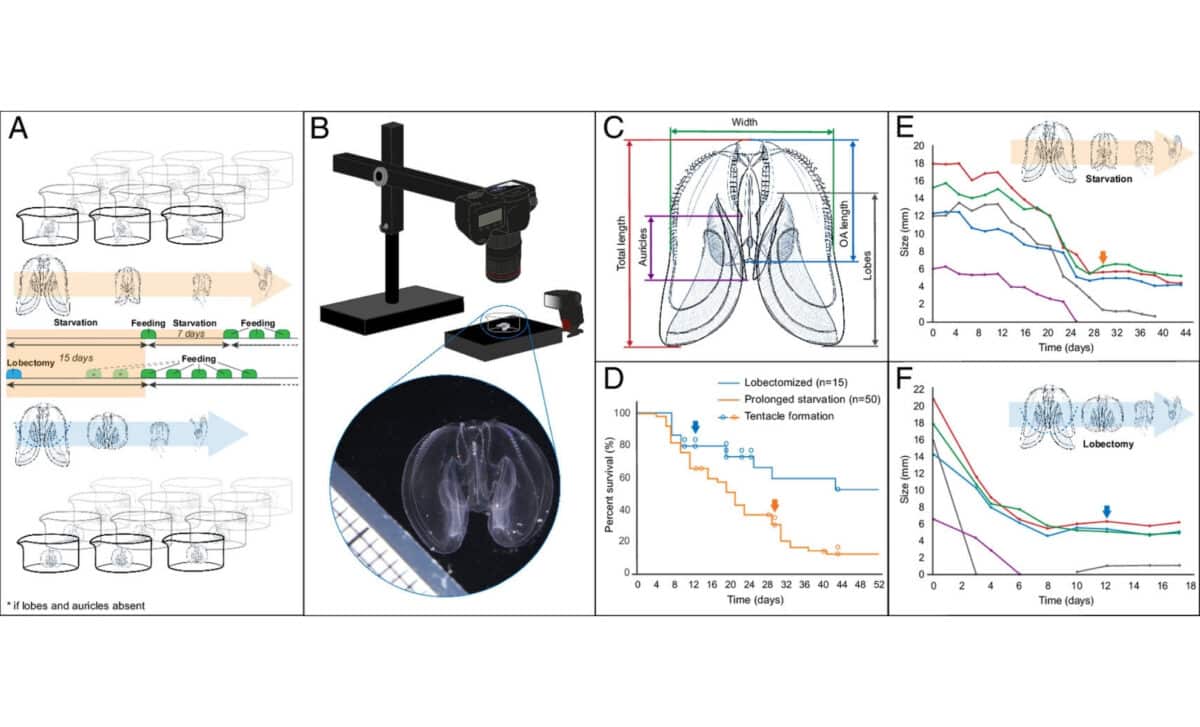 Reverse development in the ctenophore Mnemiopsis leidyi – © Proceedings of the National Academy of Sciences
Reverse development in the ctenophore Mnemiopsis leidyi – © Proceedings of the National Academy of Sciences
Starvation, Surgery, and a Slow Return to Childhood
Sixty-five adult Mnemiopsis leidyi specimens were isolated in tanks without food for 15 days. After that period, they were fed on a reduced diet of once per week. Researchers observed that the fleshy lobes these animals develop as markers of adulthood began to be reabsorbed into their bodies. Over several weeks, 13 of the 65 specimens had fully reverted to both the physical appearance and the feeding behaviors typical of larvae.
According to Soto-Angel, the experience was striking to witness firsthand. “Witnessing how they slowly transition to a typical cydippid larva, as if they were going back in time, was simply fascinating,” he said in a press release from the University of Bergen.
A separate subset of 15 specimens from the original 65 were subjected to even more invasive procedures: the adult lobes were surgically removed, adding a layer of physical trauma on top of the starvation. The results were telling. Six of those 15 subjects completed the reversal process in just 15 days, significantly faster than the roughly six weeks it took seven of the 50 non-operated specimens to achieve the same result. The more severe the stress, the faster the creature retreated to its juvenile form.
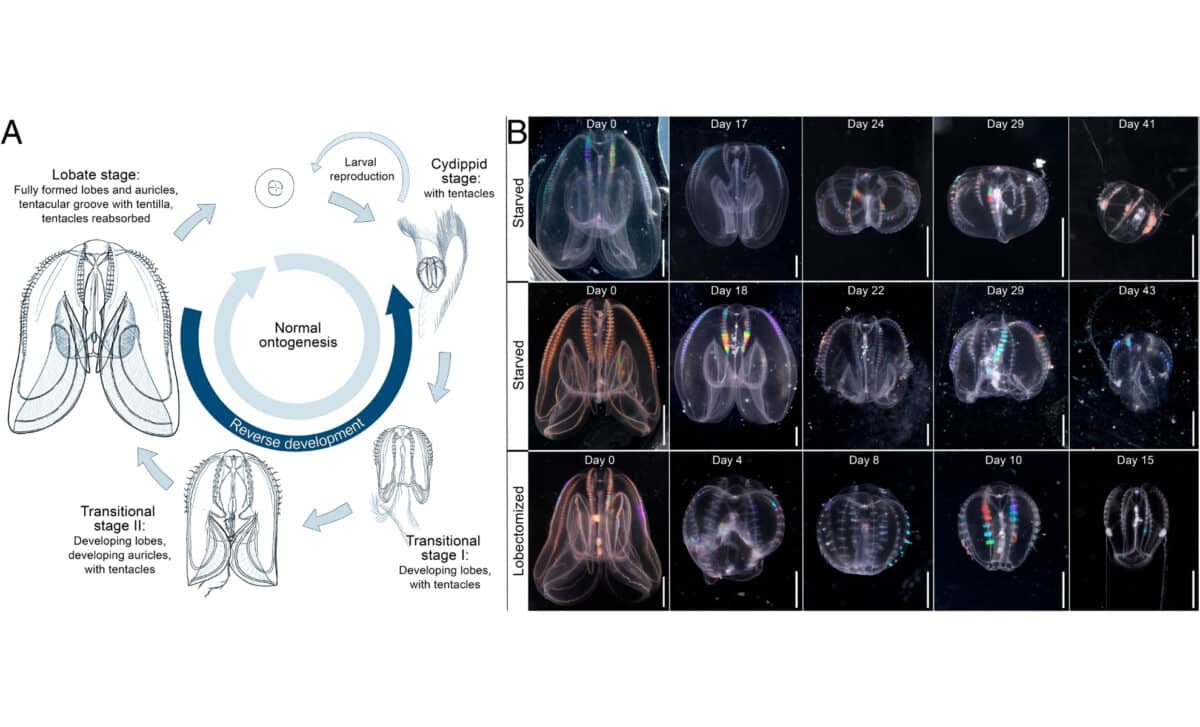 Life cycle and main morphological changes of the ctenophore Mnemiopsis leidyi – © Proceedings of the National Academy of Sciences
Life cycle and main morphological changes of the ctenophore Mnemiopsis leidyi – © Proceedings of the National Academy of Sciences
What the Science Still Doesn’t Know, and Why It Matters
The mechanisms behind this transformation remain unknown. According to Burkhardt, the molecular processes driving the reverse development have yet to be identified, as has the question of what happens to the animal’s nerve net during the transition.
“This fascinating finding will open the door for many important discoveries,” he said in the same university press release. “It will be interesting to reveal the molecular mechanism driving reverse development, and what happens to the animal’s nerve net during this process.“
The researchers also noted, as Popular Mechanics reported, that the study raises broader questions about whether other animal species might possess some version of this internal de-aging capacity, a possibility that, until recently, would have seemed far-fetched.

