Humans are not usually associated with strong regenerative powers, especially when compared to species like zebrafish or salamanders. These animals can restore complex structures such as eyes, limbs, or even parts of the brain. By contrast, mammals appear far more limited in their ability to regrow damaged body parts.
Yet this assumption does not fully hold. In certain cases, particularly in younger individuals, humans can regrow amputated fingertips, including not only skin but also bone, tissue, and nerves. This unusual capability has puzzled scientists for years and prompted closer investigation into the biological mechanisms involved.
A Rare Regenerative Ability in Mammals
Fingertip regeneration stands out because it contradicts the broader pattern observed in mammals. According to the study published in Science, regeneration occurs when the injury is located near the nail, while more proximal injuries typically result in scarring rather than regrowth.
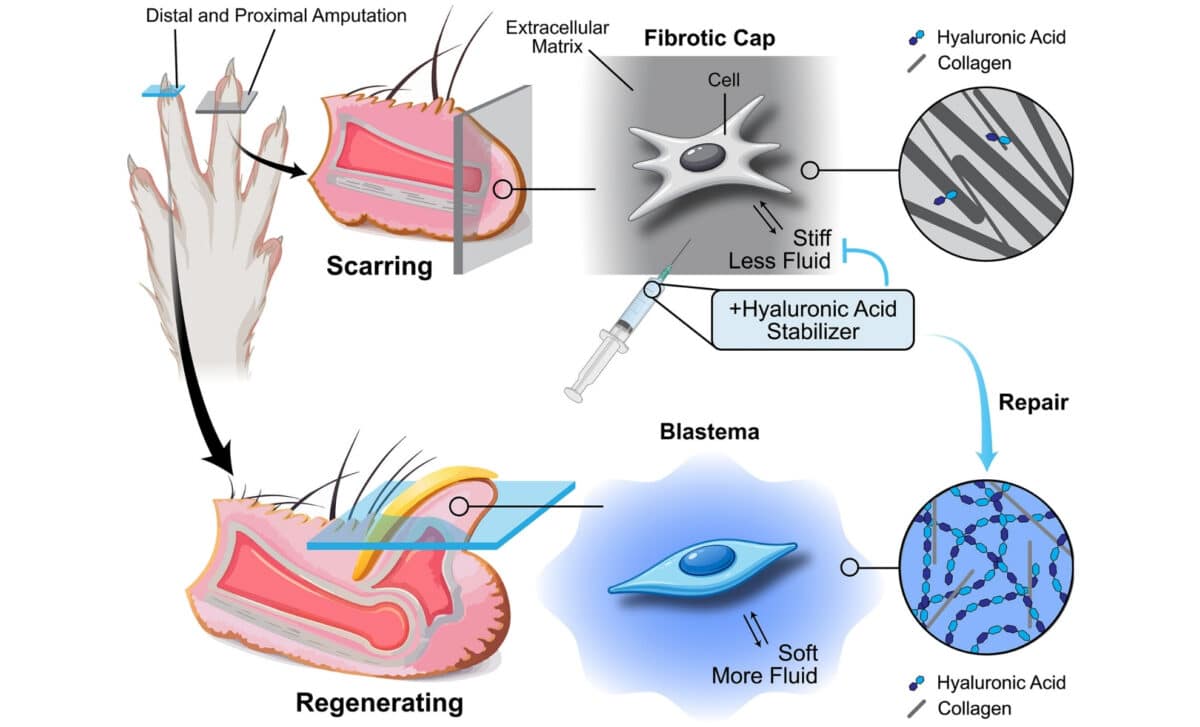 The ECM and tissue mechanics direct wound healing outcomes after digit amputations – © Science
The ECM and tissue mechanics direct wound healing outcomes after digit amputations – © Science
Researchers have long suspected that nail stem cells play a role in this process. Positioned near the cuticle, these cells may send signals that activate the regrowth of bone and nerve tissue after injury. Still, the exact mechanisms behind this phenomenon remained unclear, particularly regarding how the surrounding tissue environment influences the outcome.
The Role of the Extracellular Matrix
As reported by Popular Mechanics, the new research, conducted by scientists at Stanford University, shifts attention to the extracellular matrix (ECM), a non-cellular structure that surrounds and supports cells within tissues. According to the authors, the ECM acts as a key regulator of whether regeneration or scarring occurs.
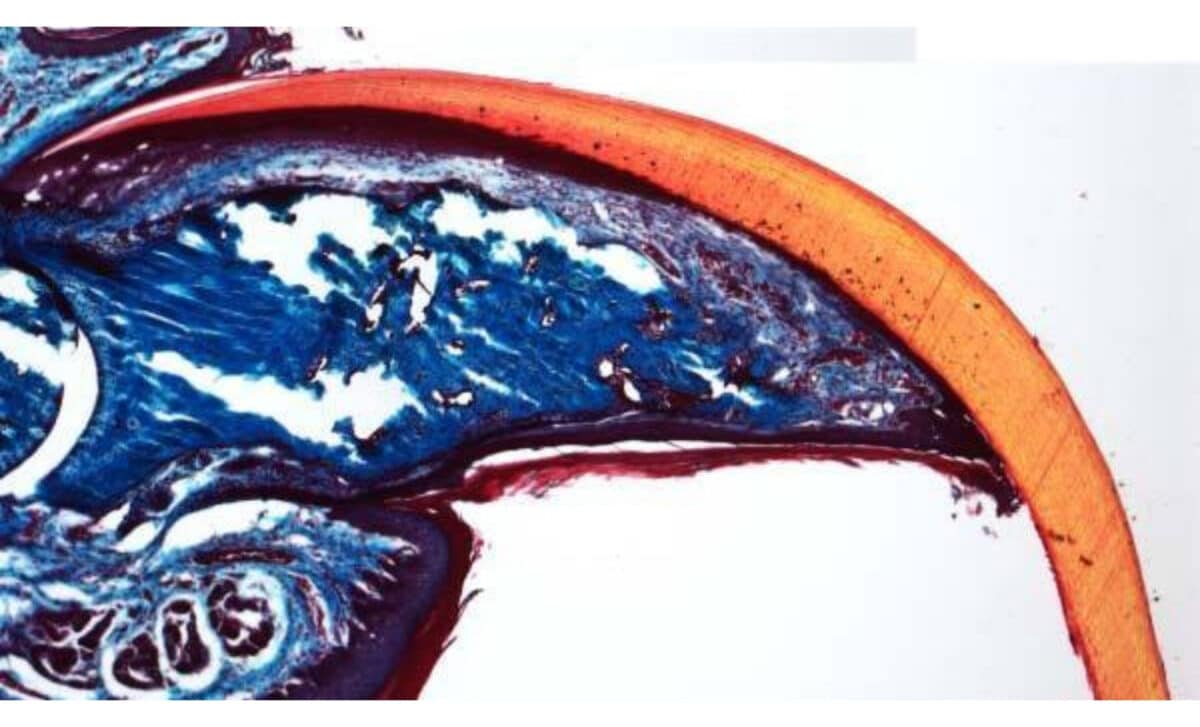 A mouse’s “fingertip” completely regenerated after amputation, growing back the bone and the claw, orange – © Ito Lab/New York University
A mouse’s “fingertip” completely regenerated after amputation, growing back the bone and the claw, orange – © Ito Lab/New York University
Their findings show a clear distinction between regenerative and nonregenerative regions. Areas capable of regrowth were softer and enriched in hyaluronic acid, while nonregenerative zones were stiffer and dominated by dense, organized collagen. As the researchers wrote, “nonregenerative regions were stiffer and contained dense, organized collagen, whereas regenerative regions were soft and enriched in hyaluronic acid.”
Using single-cell RNA sequencing, the team identified that osteo-lineage cells in regenerative areas produce hyaluronic acid along with molecules such as HAPLN1 and aggrecan. These components were largely absent in wounds that healed through scarring rather than regeneration.
Experimental Evidence from Mouse Models
To test the importance of hyaluronic acid, the researchers conducted experiments on mice. They suppressed the molecule using hyaluronidase and an additional inhibitor, observing a significant reduction in regenerative capacity. According to the study, this provided strong evidence of “an indispensable role” for hyaluronic acid in maintaining the soft, flexible tissue environment required for regrowth.
Further experiments focused on HAPLN1, a protein associated with the extracellular matrix. When applied to fibroblasts in nonregenerative digit stumps, HAPLN1 softened the tissue, reduced scarring, and even triggered bone regeneration. These results reinforced the idea that the mechanical properties of the tissue environment are central to the process.
The researchers concluded that the extracellular matrix functions as a “mechanochemical signalling hub.” According to their findings, shifting tissue conditions from a stiff, collagen-rich state to a softer, hyaluronic acid-rich environment can unlock regenerative potential, offering a new direction for regenerative medicine research.

