SR
McDermott Will & Schulte
More
As we look to 2026, exits aren’t just about timing. Dealmakers are utilizing process, planning, and creativity to find new paths to liquidity.
United States
Food, Drugs, Healthcare, Life Sciences
To print this article, all you need is to be registered or login on Mondaq.com.
As we look to 2026, exits aren’t just about timing.
Dealmakers are utilizing process, planning, and creativity to find
new paths to liquidity. Investors across healthcare sectors are
looking to dynamic opportunities while integrating flexible exit
pathways and regulatory readiness to maximize values.
At McDermott Will & Schulte’s HPE NYC 2025 conference,
industry leaders explored new sectors alongside investors’
shifting focus to innovative and deliberate exits. Here, we
continue the conference conversations with a review of emerging
opportunities and strategic exits amid regulatory developments.
In Depth
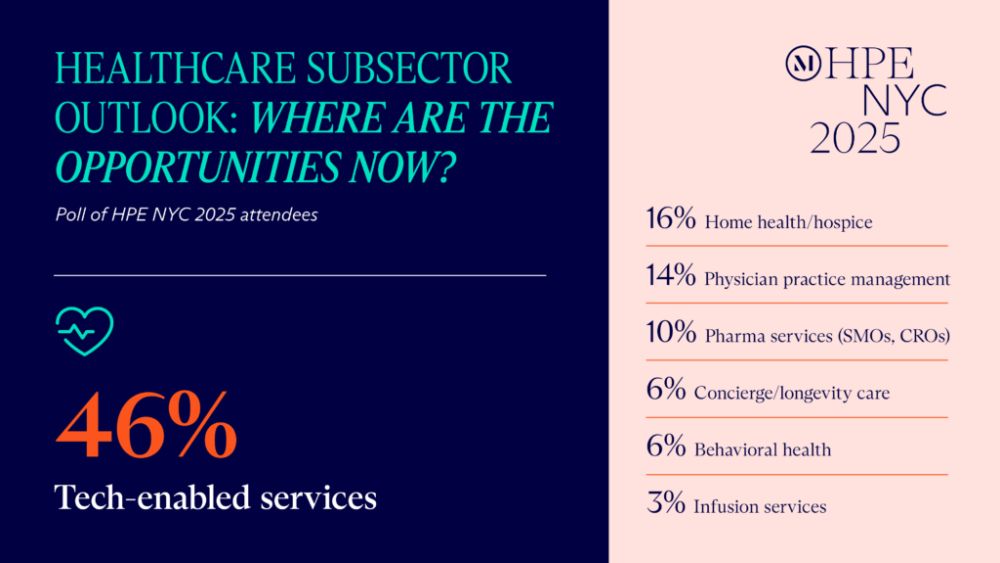
Emerging opportunities
Development and commercialization
partnerships. Strategic acquirers are increasingly
targeting tech-enabled contract research organizations and
commercialization platforms as initial public offerings (IPOs) and
cross-border activity lag. To illustrate, our event panelists noted
investments in drug manufacturing, supply chains, and drug
administration (including pharmacies and infusion centers).
Liquidity is consolidating around those strategic acquirers, which
are historically slower to get to closing and do not pay a private
equity premium.
Pharmaceutical industry. HPE NYC panelists
expect increased mergers and acquisitions activity in the
pharmaceutical industry. Specifically, looming patent cliffs and
the resulting need to replace lost revenue make late-stage,
de-risked assets strong targets with strategic advantages.
Additionally, the panelists pointed to potential for partnerships
in artificial intelligence (AI) and other technologies, which would
enable the development of increasingly comprehensive data
collection and maintenance via life sciences services.
Behavioral health. Demand and competition
continued to grow in the behavioral health sector, with valuation
rates tracking the realities of contract structures, operational
stability, and reimbursements. Notably, the increasing role of
technology is helping enhance success rates and implement
evidence-based care while boosting efficiency in the sector.
Investor-payor collaboration. Our panelists
pointed to the desirability of pursuing potential partnerships to
mitigate current pressures facing physician groups. Collaboration
with payors would help reduce reimbursement lags and offer
opportunities to launch lab services.
Creative exits
Flexible pathways. Private equity is turning
to continuation vehicles and sponsor-to-sponsor trades for flexible
exit pathways, maximizing value as the market’s appetite for
IPOs is diminished.
Trial execution. Platforms that enhance trial
execution, including in the oncology and central nervous system
spheres, offer another framework for creating exits. HPE NYC
panelists pointed out that, given the financial risks in the trial
arena, venture capital investment is common.
Physician practice management (PPM). The
panelists further highlighted the stability and potential for tech
enablement in the PPM sector, predicting an uptick in exits over
the next few years. PPM organizations are less impacted by the
macro dynamics and regulatory issues affecting other industry
businesses, and the improvements driven by technology should
solidify exit opportunities.
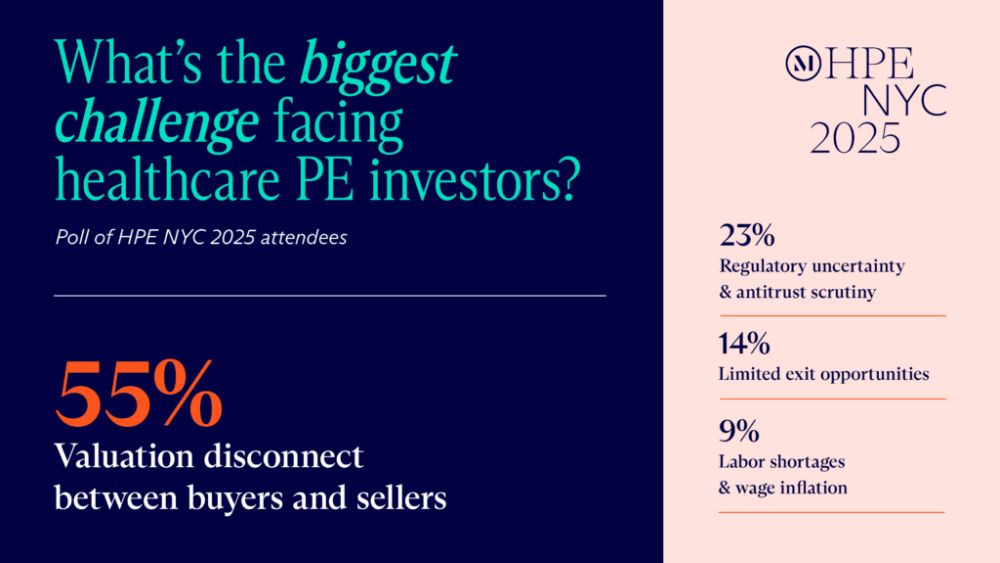
Regulatory readiness
Investors face growing risks from fragmented state privacy laws,
HIPAA and Federal Trade Commission enforcement, and US Food and
Drug Administration scrutiny of AI. In fact, 50% of HPE NYC 2025
attendees believe that navigating complex regulatory and compliance
landscapes is the most significant nonfinancial challenge faced by
private equity firms investing in healthcare. Federal and state
regulators are imposing new notice and approval requirements that
have significant transaction implications for healthcare providers,
industry operators, dealmakers, and investors.
Baby HSR. Modeled on the federal
Hart-Scott-Rodino (HSR) Act, “Baby HSR” or
“mini-HSR” laws are state laws that impose notice and
approval requirements on parties to certain healthcare
transactions. While the timing and details vary by state, event
panelists noted concern that these relatively new regulations will
delay deals. As an initial matter, parties should monitor Baby HSR
laws in the states in which they operate (or plan to operate) and
consider the impact that doing business in a state may have on the
timing of future exit transactions.
Work with lawmakers. When new laws are
proposed, it may be wise to turn to lobbyists or public relations
firms in an effort to minimize current deal disruptions or future
exit delays. Building relationships with lawmakers is key, as is
communicating the positive results healthcare investors have
achieved – and continue to achieve – in their
communities.
Readiness audits. A review of the relevant
state’s filing requirements is essential before going to
market. Readiness audits completed in conjunction with regulatory
preparedness help effectively position assets in both strategic and
sponsor exits. In addition to managing the deal timeline, that
analysis may provide alternative structures to minimize filing
requirements.
Conclusion
Looking toward increased activity in dynamic sectors and
creative exit opportunities, stakeholders should consider the
impact of regulatory reforms across the healthcare industry.
As always, we look forward to continuing the conversation with
our clients and partners. To explore these topics further, please
reach out to a member of the firm’s Health & Life Sciences Practice Group.
The content of this article is intended to provide a general
guide to the subject matter. Specialist advice should be sought
about your specific circumstances.

