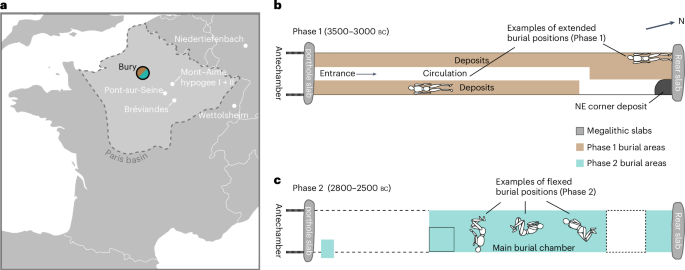
We sampled the cementum layer of 182 teeth excavated from Bury (Supplementary Table 1) and sequenced these to a depth of coverage ranging from ×0.001 to ×4.6 (median ×0.126) with 132 genomes above ×0.01, which we term sufficient coverage for further analysis (Supplementary Table 2). The remains were categorized into two chronological phases based upon the stratigraphy and the location of the remains, burial behaviours and the radiocarbon program made in parallel with the excavation18 (Fig. 1 and Supplementary Table 3). These results were then completed with data from the European Research Council-funded advanced grant ‘The times of their lives’. Phase assignments were later confirmed and refined, in particular for isolated mandibles, using the results of genetic kinship analysis. With this combined procedure, we assigned 74 and 51 samples with coverage over ×0.01 to Phases 1 and 2, respectively, while 7 samples could not be assigned to a phase (Fig. 2b).
Fig. 2: Genetic results from Bury.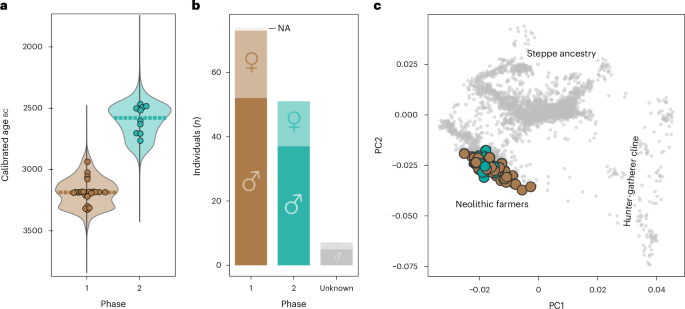
a, Kernel density calibration curves for all radiocarbon-dated individuals included in the genetic program in Phase 1 (brown) and Phase 2 (teal). The stippled line represents the median of the group. b, Number of males and females buried in the different use phases with coverage >×0.01. NA represents individuals where sex could not be reliably determined. c, PCA showing the genetic ancestry of the samples from Bury with coverage >×0.01 (in colour) in relation to published west Eurasian ancient genomes (grey).
Genetic sexing confirmed the high predominance of males in both phases20 (Fig. 2b and Supplementary Note 1), similar to reports from other Neolithic sites from present-day France and Germany21,22,23,24. Of the 131 individuals where genetic sex could be reliably determined, 71% and 73% males were observed in Phases 1 and 2, respectively. This imbalance concerns both adult and non-adult individuals, and is incompatible with a natural population, thus suggesting differential burial treatment between males and females at Bury—for some reason, more than half of the females in the community were excluded from being buried in the grave.
Mitochondrial DNA haplogroup analysis revealed a diverse set of maternal lineages over both phases (Supplementary Table 2), whereas Y chromosome haplogroups showed lower diversity among males, but with distinct patterns between phases (Supplementary Table 4 and Extended Data Fig. 1). While men from Phase 1 generally carried haplogroups H2a1 or I2a1a2, men from Phase 2 carried haplogroup I2a1a1 almost exclusively.
Genetic discontinuity between Phases 1 and 2
To determine genetic ancestry of the individuals at Bury, we analysed them within the context of a reference panel of ~4,700 previously published ancient genomes (Supplementary Table 5). From our principal component analysis (PCA), we found that all individuals from Bury fell within the broader diversity of ‘Neolithic farmers’ from western Eurasia (Fig. 2c). Furthermore, we note that the Phase 1 individuals generally appear more diverse, with a wider distribution on the PCA, as opposed to the Phase 2 individuals, which are more concentrated on the plot (Extended Data Fig. 2). The higher diversity during Phase 1 is also reflected by the proportion of hunter-gatherer ancestry, which is elevated in a handful of samples from Phase 1, resulting in a slight shift towards the hunter-gatherer cline of the PCA (Fig. 2c and Extended Data Fig. 3a,b).
To further investigate how the populations of Phases 1 and 2 in Bury relate to each other and to other Neolithic groups of Europe, we analysed identity-by-descent (IBD) segments shared between pairs of individuals. Using hierarchical graph clustering on the resulting IBD-sharing network between individuals, we found that the individuals from Bury generally clustered based on their phase. While individuals from Phase 1 cluster on their own or with individuals from the Paris Basin and western Germany, the Phase 2 individuals display strong ties to southern France and Iberia (Supplementary Fig. 2.3).
Next, we investigated total pairwise IBD sharing between the Bury populations and all other clusters to determine how genetically different the two populations at Bury were (Supplementary Fig. 2.4). We found that the two Bury populations did not share the most genetic segments with each other; instead, each phase appeared to have more total IBD sharing with several other Neolithic groups. For the population in Phase 1, 12 other groups share a higher mean total IBD length than the Phase 2 population. For Phase 2, ten groups are more similar to that population than the Phase 1 population. We also estimated the effective population sizes for each phase projected back in time using this IBD data (Extended Data Fig. 4c and Supplementary Note 2.4). From this analysis, we found that the two phases followed separate population size trajectories, and that the effective population size of Phase 1 was markedly smaller than that of Phase 2 at the point of sampling. Importantly, these estimates also revealed a very recent population size contraction in Phase 1. Taken together, these IBD-based findings provide further support for considering the individuals from the two phases as separate populations.
Having established that the two phases at Bury form two separate populations, we decided to explore whether these findings are compatible with either genetic continuity or discontinuity between the two phases (Methods). To do this, we simulated the following two different scenarios: discontinuity (blue; Extended Data Fig. 4a,b) and continuity (pink; Extended Data Fig. 4a,b). We compared patterns of IBD sharing between the simulated populations and the actual data, and found no overlap between the real data and the continuity scenario. Accordingly, these simulations rule out the scenario of complete continuity between the two populations at Bury. However, the simulation does not exclude the possibility of some amount of gene flow between the two burial phases.
Neolithic ancestry from the south could explain the shift at Bury
To put our findings from Bury into context with other Neolithic people from western Europe, we modelled the proportion of various ancestries in all individuals from Bronze Age and Neolithic Europe (Methods and Supplementary Tables 6 and 7). In agreement with our PCA analysis, we found high diversity in the Phase 1 individuals with varying proportions of modelled ancestries from Early Neolithic France and from a group of Neolithic Iberians dated to the fourth millennium BC (Supplementary Note 2.1 and Extended Data Fig. 5). This pattern is reflected in other contemporaneous individuals from the Paris Basin, from the sites Mont Aimé hypogee (I + II)25, Wettolsheim26 and Pont-sur-Seine26. For Phase 2, on the other hand, the modelling revealed a more homogenous population, with over 80% (mean 83.8% ± 0.1% s.d.) ancestry from Iberia.
When visualizing major ancestry groups on a map (Fig. 3 and Extended Data Fig. 4), the mixture modelling reveals a stepwise northwards spread of this Neolithic Iberian ancestry (Supplementary Note 2.1). By 2900 BC, populations across southern France and Iberia all constituted a large fraction of Iberian ancestry, while people in the Paris Basin still comprised mixed ancestry proportion, as represented by the Phase 1 individuals. At some point after 2900 BC, a final northwards push of the Iberian ancestry partially replaces the existing local ancestry in the Paris Basin, resulting in the homogenous population we observe in Phase 2. After the end of Phase 2, around 2500 BC, people with steppe ancestry first appear in the Paris Basin27, where they mix with the local population to form the genetic profile typically associated with Bell Beakers (Fig. 3). As such, these results readily explain the difference between the populations of Phases 1 and 2, and could suggest an event that facilitated the northwards expansion of Neolithic Iberian ancestry at around 2900 BC.
Fig. 3: The spread of Neolithic ancestry from Iberia.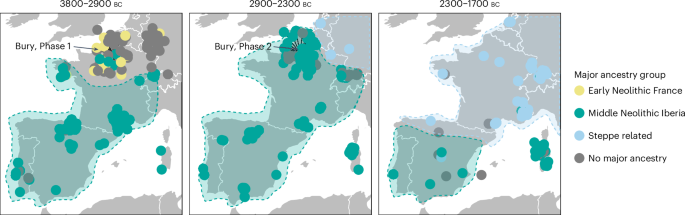
Map of genomes from western Europe coloured by the major modelled ancestry group in each individual, split by time period. Grey (‘no major ancestry’) represents individuals where no ancestry group makes up over 60% of the total ancestry. Shaded areas represent our interpretation of the geographical spread of steppe ancestry and Neolithic Iberians. Basemap data from Natural Earth (https://www.naturalearthdata.com/).
The scenario outlined above represents our interpretation of this mixture modelling data. An alternative explanation could be that the few individuals who already had high proportions of ‘Middle Neolithic Iberia’ DNA in Phase 1 proliferated and came to dominate the population in Phase 2. However, if the descendants of a few Phase 1 individuals had come to dominate in Phase 2, we would expect to see a strong bottleneck in Phase 2, more similarity in the population size trajectories for both phases (Extended Data Fig. 4c) and simulation results indicative of higher levels of population continuity. As none of these patterns are present in our data, we find this explanation less likely. Instead, we view the individuals with Iberian ancestry in Phase 1 as early arrivals originating from outside the local region. This interpretation is supported by the observation that half of the unrelated Phase 1 individuals carried high proportions of ‘Middle Neolithic Iberia’ ancestry, while only three individuals within the pedigrees exhibited this genetic profile.
A shift in societal structure inferred by genetic links
To examine differences in the social organization between the two phases at Bury, we investigated genetic relatedness with NgsRelate28,29 and KIN30 (Supplementary Table 8). We found that three quarters of individuals in Phase 1 (55 out of 72 (76%); Fig. 4c) have at least one first- or second-degree relative also buried at the site, whereas Phase 2 displays a larger fraction of unrelated individuals (only 21 related among 53 individuals (40%); Fig. 4c). Furthermore, we found no trace of relatedness between individuals across the two phases. To investigate whether there were differences in societal structure between the two phases, we combined these results with Y chromosome and mitochondrial haplogroups to reconstruct a total of 14 pedigrees. From each phase, seven pedigrees could be reconstructed varying in size, from large networks spanning up to five generations to small pedigrees consisting of only two individuals (Fig. 4 and Extended Data Figs. 6 and 7).
Fig. 4: Familial relations in Phases 1 and 2.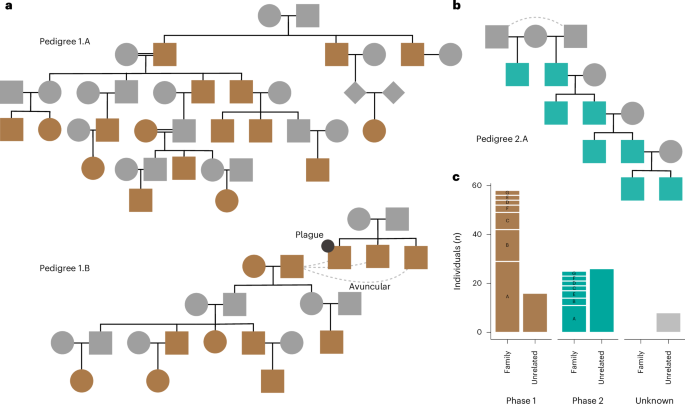
a, Pedigree 1.A and 1.B from Phase 1. Double black lines indicate mating between related individuals. The stippled lines in a and b represent unknown second degree relationships (avuncular: relationship between uncle/aunt and nephew/niece). b, Pedigree 2.A from Phase 2. c, Bar plot depicting the number of individuals within each pedigree group compared to unrelated individuals, stratified into phases. For visual clarity, each pedigree group is abbreviated with a single letter (A, B, C and so on), where, for example, ‘A’ in Phase 1 corresponds to ‘pedigree 1.A’, while ‘B’ in Phase 2 represents ‘pedigree 2.B’, and so on (Supplementary Note 2.2, Extended Data Figs. 6 and 7 and Supplementary Table 2).
In the first phase of the burial, the sampled material is dominated by large biological groups spanning several generations, with several cases of three or four full siblings (for example, siblings sharing both parents, as opposed to half siblings) or their offspring being buried in the grave (Fig. 4a and Extended Data Fig. 6). We term the seven groups in Phase 1 ‘1.A–1.G’. We observe no cases where males from outside the family line entered it by having children with its members. Aside from the founding generation of each pedigree, all subsequent males are genetically descended from earlier members. On the other hand, all females except one are related exclusively to their offspring, suggesting a high level of female exogamy for the females buried in the tomb. Interestingly, the male sex bias observed within families is not as pronounced for unrelated individuals. This observation aligns well with reports from sites with high levels of female exogamy, where unrelated individuals tend to be dominated by females, perhaps representing females that never produced offspring or whose offspring were buried outside of the tomb13.
Pedigree 1.A is the largest of the Phase 1 pedigrees (Fig. 4a and Extended Data Fig. 6a), containing 29 sequenced individuals and a further 19 inferred individuals who were either not sampled or buried outside the grave. All males in this group have Y chromosome haplogroup H2a1. Three brothers (BUR222, BUR174 and BUR343) form the first generation. Of these, BUR343 does not have offspring that were buried in the grave, while BUR174 only has a single granddaughter. However, BUR222 fathers four children (three sons, two of which sequenced, and one daughter), and all of these children have children, grandchildren and, in one case, great-grandchildren who were buried within the grave.
The two sequenced sons of BUR222, BUR291 and BUR275, both show long runs of homozygosity, with similar length profiles as expected from parents that are first cousins (Extended Data Fig. 8a), suggesting that BUR222 and the unsampled mother were third-degree relatives. One of these sons, BUR291, has a further son (unsampled), who conceived three children with one female, BUR262, who is a third- or fourth-degree relative to BUR291, her reproductive partner’s biological father. The only sequenced son of this union (BUR316) also exhibits long runs-of-homozygosity segments. Incidentally, BUR262 is also the only female buried in the grave who has both ancestors and children also buried at Bury.
One of the three brothers, BUR174, in the first generation of Phase 1 is also of some note. He was found as an almost complete skeleton, in a seated position in the northeastern corner of the grave. The skull had three perimortem lesions, each of which was caused by a blow with a heavy and sharp instrument, theorized to be an axe19. Around BUR174, three children and one perinatal individual were deposited in a seemingly deliberate pattern. One of these children, BUR189, was successfully sequenced and found to be an avuncular relation to the three brothers. As they share the same mitochondrial haplogroup, it is likely that BUR189 is the offspring of a sister to the three-brother group. It is not known if the other children in the burial arrangement are relatives, but given the high predominance of related individuals in Phase 1, it is likely that these individuals were related to others. This arrangement was previously hypothesized to be part of a ‘founding act’ of the monument because of the position and characteristics of BUR174 in the corner of the rear end of the grave19 (Fig. 1a). Given our genetic results and BUR174’s position in the first generation of pedigree group 1.A, this hypothesis seems likely.
The other large pedigree in the first phase, pedigree 1.B, spans four generations, with all individuals descending from a ‘founder’ reproductive couple, BUR257 and BUR266. Their children, an unsampled female and male, went on to have a total of at least five children and three grandchildren. All of these individuals but one son were included in our analysis. The remaining son was either not buried at Bury or not sampled. One of the children, the only offspring of the unsampled daughter, BUR185, is a juvenile male who shares the same chromosome Y haplogroup H2a1 as pedigree group 1.A. This is remarkable, as the rest of pedigree group 1.B males have the I2a1 haplogroup. The second phase lacks the large pedigrees that dominated the first phase (Fig. 4c and Extended Data Fig. 7). There are seven distinct pedigrees, which we term pedigree 2.A–2.G, the largest of which is pedigree group 2.A. Pedigree group 2.A spans four generations in a patrilineal line, with each of the four generations constituting exactly two brothers (or half-brothers), only one of which in turn has offspring buried in the grave. This consistent pattern is suggestive of a hereditary network.
There are only two females in the second phase with first-degree relatives—one mother of a son buried in the grave (pedigree 2.F) and one female with a full brother (pedigree 2.C). On the one hand, as the second phase lacks the wide pedigrees that characterize the first phase, it seems as though the burials were more selective. However, the higher fraction of totally unrelated individuals could speak to the monument following a different kind of organization with a much higher degree of non-biological relatives included in the grave. Altogether, the differences in the pedigrees and the distribution of related and unrelated individuals between the two phases of Bury imply that the grave was used differently through time—a finding which is corroborated by demographic analysis12; in the first phase it was used for wider families and in the second phase it was used for smaller patrilineal lines and unrelated individuals.
There is no correlation between burial position within the grave and genetic relationship, except in the founding deposit (Supplementary Note 7). Without comparable studies of similar collective burials, it is difficult to interpret such a result. However, it confirms the community character of the tomb. Slightly older, the two chambers of the long barrow of Hazleton North31, with fewer individuals, nevertheless show that the internal distribution of the individuals is not guided by genetic links; conversely, these links decide the distribution of individuals in either chamber. The predominance of a large genetic group during the first phase, probably related to the other small groups, suggests a strong association between this group and the collective grave, or to the links that could exist between the inhabitants of the same place. Phase 2, on the other hand, is characterized by a single paternal family line, and a handful of small pedigrees scattered throughout the different spaces of the tomb. As this phase is also represented by a substantially higher proportion of unrelated individuals, this could imply that social or cultural ties were more important during this time. It should also be noted that the longer duration of Phase 2, compared to Phase 1 (ref. 19), may have contributed to the different selection of the dead.
Evidence for the earliest diverging lineage of Yersinia pestis at Bury
While no published record on the general health of the populations in Bury exist, an analysis of the oral-dental area did not find evidence of poor conditions20. To investigate if this pattern could be corroborated by genetic data, we screened all samples for pathogen DNA (Methods). In total, we detected four different microbes that most likely stem from a disease in the host: Yersinia enterolitica (n = 8), Yersinia pestis (n = 4), Borrelia recurrentis (n = 2) and Human alphaherpesvirus 1 (that is, Herpes simplex virus 1, n = 1; Supplementary Table 9). Of these four pathogens, only Yersinia pestis, the aetiological agent of plague32,33, displays an uneven distribution between the two burial phases: three individuals are from Phase 1, whereas only one individual with plague is detected in Phase 2. The plague-positive individuals do not appear to be related; one male comes from pedigree 1.E (Extended Data Fig. 6e), another male comes from pedigree 1.B (Fig. 4a), while one female from Phase 1 and a male from Phase 2 are both unrelated (Extended Data Figs. 6h and 7h). Of the other pathogens detected, Yersinia enterocolitica, the cause of yersiniosis, and Borrelia recurrentis, the causative agent of louse-borne relapsing fever, are both noteworthy. While the mortality of yersiniosis today is low, almost a third of cases need hospitalization. Furthermore, the mortality of untreated louse-borne relapsing fever ranges from 15% to 40%, and has been associated with poor hygiene and, in historic times, with catastrophic events such as war or famine.
The coverage of Yersinia pestis in the four plague-positive individuals is generally low. With a coverage of ×0.43, BUR218 is our best sample, while the remaining samples are significantly lower (×0.010, ×0.019 and ×0.002; Supplementary Table 10). Using phylogenetic placement analyses, we found that the three plague-positive samples over ×0.01 were placed in the pre-Late Neolithic/Bronze Age (preLNBA) cluster (Extended Data Fig. 9). Furthermore, due to the higher coverage of our best sample, we were able to place it in the phylogeny with higher confidence. We found that the plague form from BUR218 (3339–3042 BC) was placed at the branch basal to the genome RV203934 (3350–3100 BC; Fig. 5b). This finding suggests that the plague form from Phase 1 of Bury is similar, and perhaps slightly older, than that of the RV2039 genome isolated from a hunter-gatherer in present-day Latvia.
Fig. 5: Phylogenetic placement of the Bury plague.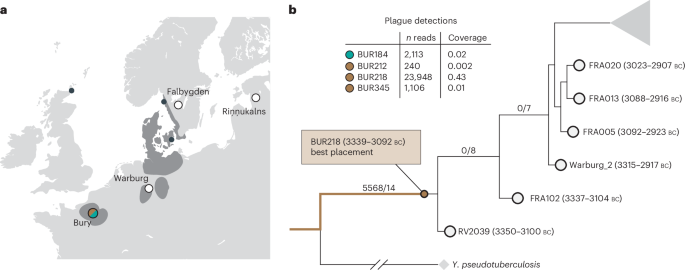
a, Map of pre-Late Neolithic/Bronze Age plague cases. Areas in dark grey correspond to the main concentrations of collective graves in the second half of the fourth millennium, with bones preserved. b, Phylogenetic placement of the highest coverage plague sample (BUR218, depth of coverage: ×0.43). Basemap data from Natural Earth (https://www.naturalearthdata.com/).
Given that we find three plague cases in Phase 1 (3 out of 74, 4% of Phase 1 samples with coverage ≥×0.01) and only a single case in Phase 2 (1 out of 51, 2% of Phase 2 samples with coverage ≥×0.01), one could hypothesize that an outbreak of the plague caused the end of Phase 1, leading to the cessation of burials. However, two hallmark features that would be expected for a deadly plague outbreak are missing at Bury: (1) at 4% the plague prevalence in Phase 1 is still relatively low (at the Swedish site Frälsegården, for example, a prevalence of 28% was reported13) and (2) the plague-positive cases do not appear to be distributed in the last generations of the pedigree, although they are among the last deposits of Phase 1 in the anterior part of the grave. However, the grave only represents a subset of the population and it is perfectly possible that a severe plague epidemic would leave very little evidence behind, if the entire population perished without being buried or if they were buried differently, presumably elsewhere, as was the case for many plague victims during the second pandemic35,36.
Environmental data on the Neolithic decline
Lastly, we investigated pollen data from the Paris Basin to assess if the population collapse observed between Phase 1 and Phase 2 could be linked to any vegetational changes. Of the seven Neolithic temporal windows analysed by David et al.37, the interval from 2900 to 2500 BC shows evidence of forest regeneration, which is typically linked to a decrease in human activity (Supplementary Note 3). A similar pattern was observed, both in Scania, Sweden (Supplementary Note 3), where forest regrowth reached a climax around 3100 BC, and in Zealand, Denmark, showing a climax between 3000 and 2800 BC (Supplementary Note 4). Similar results have previously been documented in northern Germany38, and in central Europe where the decline period could be dated to between 3300 and 2950 BC39 based on a combination of summed probability distributions of radiocarbon dates and palaeoecological proxies. As such, these observations from a number of well-documented regions can be interpreted as resulting from abandonment of grazing lands and fields, implying settlements were given up. Accordingly, they describe a significant decline in human activity, and are in agreement with similar observations after the Justinian plague40 and the Black Death41.